Revolutionizing Brivaracetam Synthesis: A Deep Dive into High-Efficiency Chiral Lactam Manufacturing
Revolutionizing Brivaracetam Synthesis: A Deep Dive into High-Efficiency Chiral Lactam Manufacturing
The global demand for next-generation antiepileptic drugs has placed immense pressure on the supply chains of key pharmaceutical intermediates, specifically requiring unprecedented levels of chiral purity and process efficiency. Patent CN109516943B introduces a groundbreaking preparation method for a lactam intermediate with high chiral purity, which serves as a critical precursor for Brivaracetam. This technology represents a paradigm shift from traditional resolution-based methods to a direct, highly selective catalytic hydrogenation process. By leveraging a unique combination of heavy metal catalysts and specific chiral inducers, this innovation allows manufacturers to bypass complex separation steps that have historically bottlenecked production capacity. For R&D directors and procurement strategists alike, understanding the nuances of this patent is essential for securing a competitive edge in the generic and branded API markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral lactam intermediates for antiepileptic agents has been plagued by significant inefficiencies related to stereochemical control. Traditional routes often rely on late-stage resolution techniques, such as chiral chromatography or enzymatic kinetic resolution, which inherently limit the maximum theoretical yield to fifty percent unless dynamic processes are employed. These methods not only consume vast amounts of expensive chiral stationary phases but also generate substantial solvent waste, driving up the environmental footprint and operational expenditure. Furthermore, conventional reduction methods frequently suffer from poor diastereoselectivity, resulting in complex mixtures of isomers that require multiple recrystallization cycles to meet the stringent purity specifications required for clinical applications. The reliance on harsh reaction conditions in older methodologies also poses safety risks and complicates the scale-up process, making it difficult to ensure batch-to-batch consistency in commercial manufacturing environments.
The Novel Approach
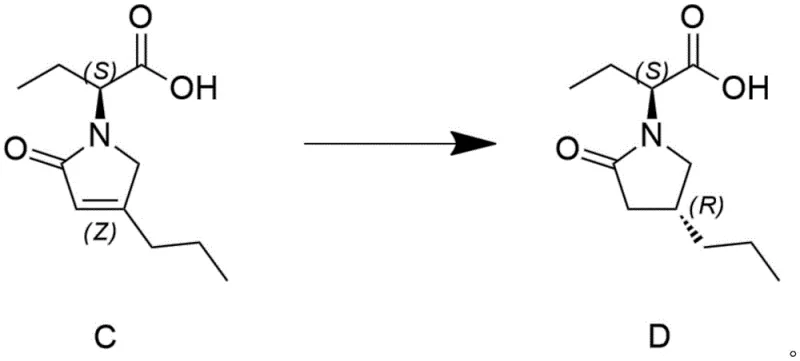
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated asymmetric hydrogenation strategy that installs the necessary stereocenters with exceptional precision during the reduction phase itself. By employing a heavy metal catalyst, such as palladium on carbon, in conjunction with a carefully selected chiral inducer like hydrobromic acid or citric acid, the reaction directs the hydrogen addition to favor the desired diastereomer overwhelmingly. This method achieves a conversion rate of Compound C reaching up to eighty-one percent and a de value exceeding ninety-nine percent, effectively eliminating the need for costly chiral column separation. The process operates under mild conditions, with temperatures ranging from minus twenty degrees Celsius to fifty degrees Celsius and hydrogen pressures not exceeding five bar, which significantly enhances operational safety. This streamlined workflow transforms a multi-step purification nightmare into a simple filtration and recrystallization sequence, drastically reducing the overall production timeline and resource consumption.
Mechanistic Insights into Pd/C-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the synergistic interaction between the heterogeneous metal catalyst and the chiral inducing agent within the reaction medium. When Compound C, an unsaturated lactam derivative, is introduced to the palladium surface in the presence of a chiral acid, the acid molecules likely adsorb onto the catalyst or form transient ion-pair complexes with the substrate. This interaction creates a chiral environment around the active sites of the metal, sterically hindering the approach of hydrogen from one face of the double bond while facilitating attack from the other. The result is a highly diastereoselective reduction that locks the stereochemistry at the newly formed chiral center with remarkable fidelity. The choice of solvent, particularly protic solvents like ethanol or isopropanol mixed with water, further stabilizes these transition states and ensures efficient mass transfer of hydrogen gas to the catalyst surface. This mechanistic elegance allows for the use of relatively inexpensive, commercially available catalysts like Pd/C instead of exotic homogeneous noble metal complexes, making the process economically viable for large-scale industrial application.
Controlling impurities in this synthesis is achieved through the inherent selectivity of the catalytic system combined with a robust workup procedure. Because the reaction generates minimal amounts of the unwanted diastereomer, the burden on downstream purification is significantly lightened. The patent highlights that the solid state of the reaction intermediate allows for easy further purification via recrystallization using solvents like methyl tert-butyl ether. This physical property is crucial for removing trace metal residues and any minor organic byproducts that may form during the hydrogenation. Additionally, the ability to filter and recycle the palladium catalyst not only reduces raw material costs but also minimizes the risk of heavy metal contamination in the final product, a critical quality attribute for pharmaceutical intermediates. The process avoids high-temperature and high-pressure extremes, thereby preventing thermal degradation pathways that often lead to complex, hard-to-remove impurity profiles in traditional synthesis routes.
How to Synthesize Chiral Lactam Intermediate D Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and stereoselectivity while maintaining safety standards. The process begins with the dissolution of the starting material in a optimized solvent system, followed by the sequential addition of the catalyst and the chiral promoter. It is imperative to maintain an inert atmosphere, typically using nitrogen protection, to prevent catalyst poisoning and ensure safe handling of hydrogen gas. The reaction temperature must be strictly regulated, often requiring cooling to sub-zero temperatures to achieve the highest de values, as demonstrated in the patent examples where minus ten to zero degrees Celsius yielded optimal results. Once the hydrogenation is complete, indicated by the disappearance of starting material on TLC or HPLC, the mixture undergoes a straightforward filtration to recover the catalyst, followed by pH adjustment and crystallization to isolate the pure product.
- Dissolve Compound C in a suitable solvent system such as ethanol or isopropanol mixed with water, ensuring complete solubilization prior to catalysis.
- Introduce the heavy metal catalyst, preferably palladium on carbon, along with a specific chiral inducer like hydrobromic acid or citric acid to the reaction mixture.
- Conduct hydrogenation under mild pressure (less than 5 bar) and controlled low temperatures ranging from -20°C to 50°C to achieve high diastereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. The most significant economic driver is the complete elimination of chiral chromatography, a unit operation known for its exorbitant capital and operational costs due to the expense of chiral resins and the large volumes of solvents required. By achieving high chiral purity directly through catalysis, manufacturers can drastically simplify their production infrastructure, reducing the need for specialized separation equipment and lowering the overall cost of goods sold. Furthermore, the ability to recycle the palladium catalyst multiple times without significant loss of activity provides a sustainable avenue for cost reduction in API manufacturing, insulating the supply chain from volatility in precious metal prices. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a greener and more cost-effective production profile that aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The removal of expensive chiral separation columns and the associated solvent recovery systems leads to substantial capital expenditure savings and reduced operating costs. The use of common, inexpensive acids as chiral inducers instead of proprietary chiral ligands further drives down the raw material bill, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The simplicity of the post-treatment process, involving basic filtration and crystallization, reduces the risk of batch failures and delays often associated with complex purification steps. The robustness of the catalyst system allows for consistent production schedules, ensuring a steady flow of high-purity intermediates to downstream API synthesis units without the bottlenecks of resolution processes.
- Scalability and Environmental Compliance: Operating at low hydrogen pressures and moderate temperatures simplifies the engineering requirements for reactor vessels, making the scale-up from pilot plant to commercial tonnage significantly safer and more straightforward. The reduction in solvent usage and the elimination of hazardous waste streams associated with chiral columns enhance the environmental compliance profile, facilitating easier regulatory approval and permitting for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What is the primary advantage of this hydrogenation method over traditional resolution?
A: The primary advantage is the elimination of chiral chromatographic separation. By introducing a chiral inducer during the hydrogenation step, the process achieves greater than 99 percent de value directly, significantly reducing downstream processing costs and time.
Q: Can the catalyst be recycled in this specific synthetic route?
A: Yes, the patent data indicates that the palladium carbon catalyst can be filtered and separated after the reaction for recycling and reuse, which contributes to substantial cost optimization and waste reduction in large-scale production.
Q: What are the critical reaction parameters for maintaining high optical purity?
A: Maintaining the reaction temperature between -20°C and 0°C is critical, alongside controlling the hydrogen pressure below 5 bar. The ratio of the chiral inducer to the substrate also plays a vital role, with lower ratios often yielding excellent selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics seen in patent literature are faithfully reproduced on an industrial scale. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific requirements of catalytic hydrogenation, including specialized high-pressure reactors and advanced filtration systems necessary for catalyst recovery and product isolation.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis route for their Brivaracetam supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this more efficient manufacturing process. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless and cost-effective path to market for your final drug products.
