Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The landscape of modern pharmaceutical development is increasingly defined by the demand for complex heterocyclic scaffolds that offer superior metabolic stability and bioavailability. Among these, the 1,2,4-triazole core stands out as a privileged structure, embedded in the molecular architecture of blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox. As illustrated in the structural diversity of these active pharmaceutical ingredients, the incorporation of trifluoromethyl groups into these heterocycles further enhances their physicochemical properties, including lipophilicity and electronegativity. 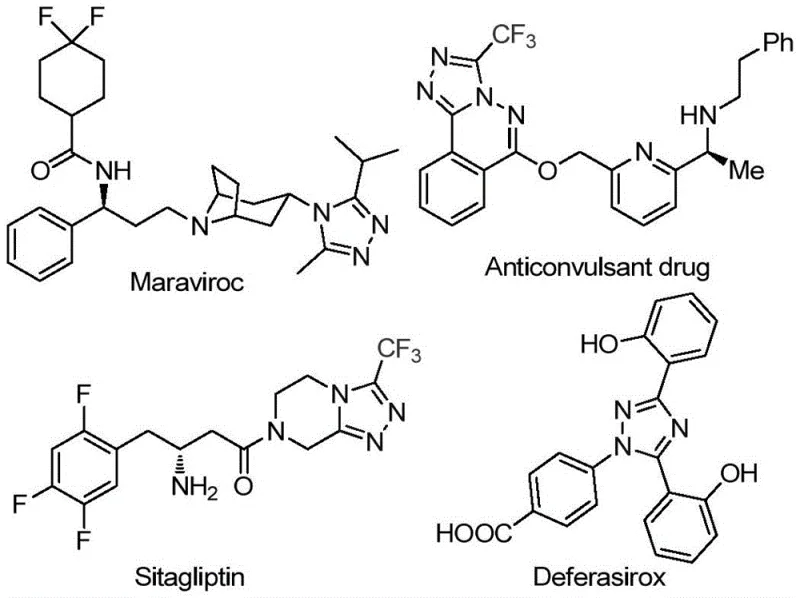 . Addressing the critical need for efficient access to these motifs, the technology disclosed in patent CN113105402B presents a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. This innovation offers a streamlined, metal-free pathway that bypasses the limitations of traditional synthesis, providing a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines.
. Addressing the critical need for efficient access to these motifs, the technology disclosed in patent CN113105402B presents a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. This innovation offers a streamlined, metal-free pathway that bypasses the limitations of traditional synthesis, providing a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, particularly those bearing both acyl and trifluoromethyl functionalities, has been fraught with synthetic challenges. Conventional methodologies often rely heavily on the use of transition metal catalysts, such as copper or palladium complexes, which introduce significant cost burdens and complicate downstream purification due to stringent regulatory limits on residual heavy metals in drug substances. Furthermore, many existing protocols necessitate rigorous anhydrous and oxygen-free environments, requiring specialized equipment like gloveboxes or Schlenk lines that are difficult to justify in large-scale commercial manufacturing. The reliance on exotic reagents and harsh conditions not only inflates the cost reduction in API manufacturing but also poses safety risks and environmental hazards associated with toxic waste disposal. Additionally, the substrate scope in older methods is frequently narrow, failing to tolerate diverse functional groups, which limits the medicinal chemist's ability to explore structure-activity relationships efficiently.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a simple yet highly effective iodine-promoted strategy that fundamentally shifts the paradigm of triazole synthesis. By utilizing cheap and readily available aryl ethanones and trifluoroethylimide hydrazides as starting materials, this method eliminates the dependency on precious metal catalysts entirely. The reaction proceeds in dimethyl sulfoxide (DMSO), which serves a dual role as both the solvent and an oxygen source for the initial oxidation step, thereby simplifying the reagent list. Crucially, the process operates under ambient atmospheric conditions without the need for inert gas protection, drastically lowering the barrier to entry for commercial scale-up of complex pharmaceutical intermediates. The operational simplicity extends to the workup procedure, which involves basic filtration and standard column chromatography, ensuring high purity specifications are met with minimal effort. This accessibility makes the technology particularly attractive for high-purity OLED material and pharmaceutical sectors alike.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The elegance of this synthesis lies in its tandem reaction mechanism, which seamlessly integrates oxidation and cyclization into a single pot. The process initiates with the iodination and subsequent Kornblum oxidation of the aryl ethanone substrate. In the presence of iodine and DMSO at elevated temperatures (90-110°C), the methyl ketone is transformed into an reactive aryl diketone intermediate. This oxidative transformation is critical, as it generates the necessary electrophilic carbonyl centers required for the subsequent ring closure. Following this initial phase, the introduction of trifluoroethylimide hydrazide triggers a condensation reaction, forming a hydrazone intermediate. The reaction mixture is then heated to 110-130°C in the presence of sodium dihydrogen phosphate and pyridine, which act as mild bases to facilitate the final intramolecular cyclization. 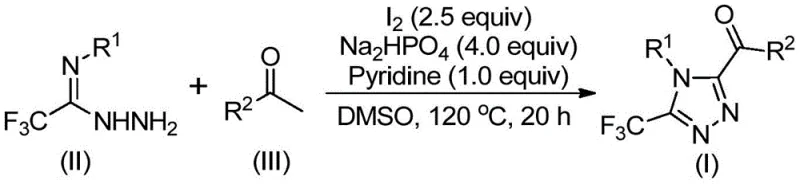 . This cascade effectively constructs the 1,2,4-triazole ring while simultaneously installing the trifluoromethyl group at the 3-position and the acyl group at the 5-position, demonstrating remarkable atom economy and step efficiency.
. This cascade effectively constructs the 1,2,4-triazole ring while simultaneously installing the trifluoromethyl group at the 3-position and the acyl group at the 5-position, demonstrating remarkable atom economy and step efficiency.
From an impurity control perspective, the use of elemental iodine and inorganic phosphate buffers ensures a clean reaction profile. Unlike transition metal catalysis, which can lead to complex organometallic side products that are difficult to separate, the byproducts of this iodine-mediated process are generally inorganic salts and unreacted starting materials that are easily removed during the aqueous workup or silica gel treatment. The tolerance of the reaction to various substituents on the aryl rings (R1 and R2), including electron-donating groups like methoxy and electron-withdrawing groups like chloro or trifluoromethyl, indicates a robust mechanistic pathway that is not easily perturbed by steric or electronic variations. This broad functional group tolerance is essential for generating diverse libraries of high-purity pharmaceutical intermediates, allowing R&D teams to rapidly iterate on lead compounds without being constrained by synthetic feasibility.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible even to laboratories with standard equipment. The protocol begins by combining the aryl ketone and a stoichiometric amount of iodine in DMSO, followed by a controlled heating period to effect the oxidation. Once the intermediate is formed, the hydrazide component and the base system are introduced directly into the same vessel, eliminating the need for intermediate isolation and reducing solvent waste.
- Oxidation Phase: Add aryl ethanone and iodine to DMSO, heat to 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroethylimide hydrazide, additional iodine, sodium dihydrogen phosphate, and pyridine. Heat to 110-130°C for 12-20 hours to complete the ring closure.
- Purification: Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-promoted synthesis represents a strategic opportunity to optimize sourcing strategies and reduce overall production costs. The shift away from noble metal catalysts to abundant elemental iodine translates directly into significant raw material savings, as iodine is orders of magnitude cheaper than palladium or rhodium complexes. Moreover, the elimination of heavy metals simplifies the regulatory compliance landscape, removing the need for expensive scavenging resins and extensive testing for metal residues, which are common bottlenecks in API release. The robustness of the reaction conditions, specifically the lack of requirement for anhydrous solvents or inert atmospheres, reduces the capital expenditure required for specialized reactor setups, allowing for more flexible manufacturing scheduling.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, driven primarily by the substitution of expensive catalytic systems with inexpensive iodine and the use of commodity chemicals like aryl ketones. By avoiding the use of transition metals, manufacturers can bypass the costly and time-consuming steps associated with metal removal and validation, leading to substantial cost savings in the overall production budget. Furthermore, the high conversion rates and yields reported in the patent examples suggest that raw material utilization is highly efficient, minimizing waste generation and maximizing the output per batch. This efficiency is critical for maintaining competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials. Aryl ethanones and hydrazides are commodity chemicals produced by numerous suppliers globally, reducing the risk of single-source dependency that often plagues projects relying on bespoke ligands or catalysts. The simplified operational requirements mean that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, ensuring continuity of supply even in the face of logistical disruptions. This flexibility allows for better inventory management and faster response times to fluctuating market demands.
- Scalability and Environmental Compliance: The potential for commercial scale-up is a key advantage, as the reaction has been demonstrated to proceed smoothly on the gram scale with consistent results. The absence of toxic heavy metals aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations, facilitating easier permitting and waste disposal. The use of DMSO, a solvent with a well-understood safety profile and recycling potential, further enhances the environmental sustainability of the process. This alignment with eco-friendly manufacturing standards not only reduces liability but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the patent specifications. These insights are intended to clarify the operational parameters and potential applications for technical teams evaluating this route for their own portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method utilizes elemental iodine as a non-metal promoter, significantly reducing raw material costs and eliminating the need for complex heavy metal removal processes.
Q: What are the reaction conditions regarding moisture and oxygen?
A: The process is robust and does not require strict anhydrous or oxygen-free conditions, simplifying operational requirements and reducing equipment costs for industrial scale-up.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the patent explicitly demonstrates that the reaction can be easily expanded to the gram level and beyond, utilizing cheap and readily available starting materials like aryl ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted synthesis in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical procurement team to explore how this innovative methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for speed, cost, and reliability in the competitive pharmaceutical landscape.
