Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
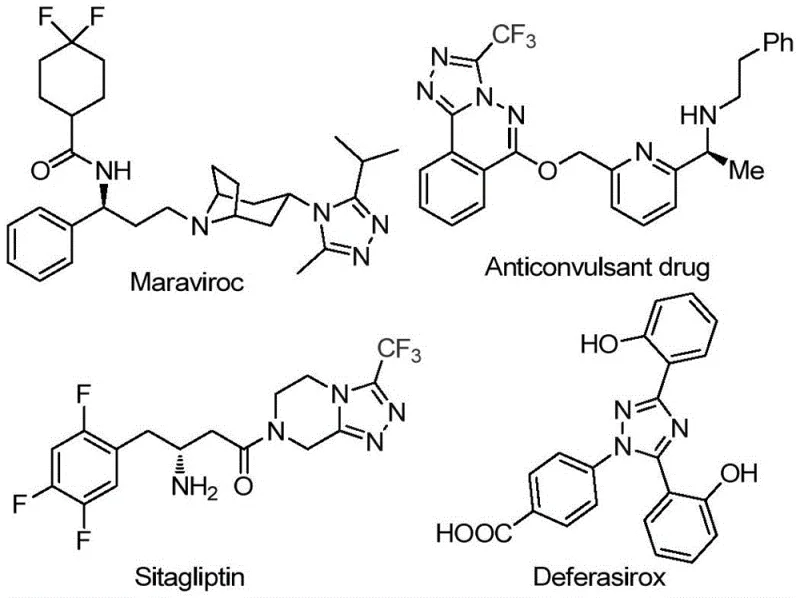
The rapid evolution of medicinal chemistry continues to drive demand for robust, scalable synthetic routes to nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. This technology addresses critical bottlenecks in the production of key pharmaceutical scaffolds found in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity of bioactive molecules below.
The patented methodology leverages a metal-free, iodine-promoted cascade reaction that transforms readily available aryl ethanones and trifluoroethylimide hydrazides into high-value intermediates. For R&D directors and procurement specialists alike, this represents a paradigm shift away from traditional transition-metal catalysis towards more sustainable, cost-efficient organic synthesis. By eliminating the reliance on precious metals and complex ligand systems, this process not only reduces the environmental footprint but also drastically simplifies the impurity profile, ensuring that the resulting high-purity pharmaceutical intermediates meet the stringent quality standards required for global regulatory submission.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole cores has relied heavily on transition-metal catalyzed cross-coupling reactions or multi-step condensation sequences that often suffer from poor atom economy and harsh reaction conditions. Traditional protocols frequently necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce significant challenges in downstream processing, specifically regarding the removal of trace heavy metal residues to parts-per-million levels. Furthermore, many existing synthetic routes require strictly anhydrous and oxygen-free environments, demanding specialized equipment and inert gas manifolds that increase capital expenditure and operational complexity. These factors collectively contribute to extended lead times and higher manufacturing costs, creating supply chain vulnerabilities for companies seeking reliable sources of complex heterocyclic building blocks.
The Novel Approach
In stark contrast, the innovation presented in patent CN113105402B introduces a streamlined, one-pot strategy that utilizes elemental iodine and dimethyl sulfoxide (DMSO) to drive the reaction forward under relatively mild conditions. This approach capitalizes on the dual functionality of iodine as both an oxidant and a promoter, facilitating a Kornblum oxidation of the aryl ethanone followed by a tandem cyclization with the hydrazide component. The elimination of sensitive metal catalysts means that the reaction does not require rigorous exclusion of moisture or oxygen, significantly lowering the barrier to entry for manufacturing facilities. Moreover, the use of cheap, commodity-grade starting materials such as aryl ethanones ensures that the cost reduction in pharmaceutical intermediate manufacturing is substantial, while the operational simplicity allows for seamless translation from laboratory discovery to commercial-scale production without the need for exotic reagents.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated example of cascade reactivity, beginning with the iodine-mediated activation of the aryl ethanone in DMSO. As depicted in the general reaction scheme below, the initial phase involves the formation of an aryl diketone intermediate through an iodination and subsequent Kornblum oxidation sequence. This electrophilic species then undergoes a dehydration condensation with the trifluoroethylimide hydrazide to generate a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine serves to buffer the reaction medium and facilitate the final intramolecular cyclization step, driven by the oxidative power of iodine, to yield the target 3,4,5-trisubstituted 1,2,4-triazole scaffold.
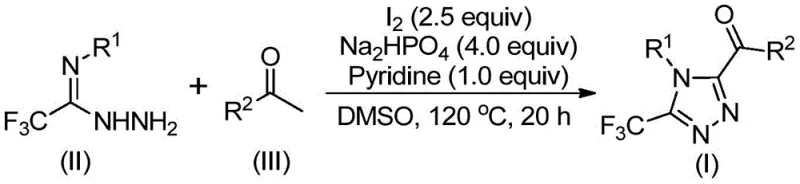
From a purity perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed alternatives. The stepwise nature of the oxidation and cyclization, controlled by temperature staging (initially 90-110°C, then 110-130°C), minimizes the formation of polymeric byproducts and regio-isomers that often plague triazole synthesis. The specific stoichiometry employed—typically a molar ratio of hydrazide to ketone of 1:2, with 2.5 equivalents of iodine—ensures complete conversion of the limiting reagent while suppressing side reactions. For process chemists, understanding this kinetic profile is crucial for optimizing residence times and maximizing yield, with experimental data demonstrating isolated yields ranging from 37% to 86% across a diverse array of substrates, confirming the robustness of the catalytic cycle.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
Implementing this synthesis requires precise control over thermal parameters and reagent addition sequences to ensure optimal conversion and selectivity. The process is designed to be operationally simple, utilizing standard glassware or stainless steel reactors without the need for high-pressure equipment. The following guide outlines the standardized protocol derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this efficient route. Detailed standard operating procedures and safety guidelines for handling iodine and DMSO at elevated temperatures should be consulted prior to execution.
- Oxidation Phase: React aryl ethanone with iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine. Heat to 110-130°C for 12-20 hours.
- Purification: Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted methodology translates directly into tangible strategic benefits, primarily driven by the simplification of the bill of materials and the reduction of processing steps. By removing the dependency on volatile precious metal markets and complex ligand supply chains, manufacturers can achieve greater predictability in raw material sourcing and pricing. The ability to run the reaction without stringent anhydrous conditions further reduces utility costs associated with solvent drying and inert gas consumption, contributing to a leaner, more agile manufacturing model that is resilient to external market shocks.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with commodity-grade elemental iodine results in a drastic decrease in direct material costs. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of silica gel and purification solvents, leading to substantial overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Since the starting materials, including aryl ethanones and trifluoroethylimide hydrazides, are commercially available and synthetically accessible from common precursors, the risk of supply disruption is minimized. This accessibility ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and enabling just-in-time delivery models for downstream API synthesis.
- Scalability and Environmental Compliance: The process has been validated for scalability from gram to multi-kilogram levels, demonstrating excellent heat transfer characteristics in DMSO. Furthermore, the absence of heavy metals simplifies waste stream treatment and disposal, aligning with increasingly stringent environmental regulations and supporting corporate sustainability goals without compromising on output volume or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on operational parameters and substrate compatibility for potential licensees and manufacturing partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metallic promoter, completely avoiding the need for costly palladium or copper catalysts and simplifying downstream purification.
Q: What are the optimal reaction conditions for scale-up?
A: The process operates effectively in DMSO solvent with a two-stage heating profile: initial oxidation at 90-110°C followed by cyclization at 110-130°C, making it highly suitable for industrial thermal management.
Q: Is the method tolerant to various functional groups on the aryl rings?
A: Yes, the protocol demonstrates excellent substrate scope, successfully accommodating substituents such as methyl, methoxy, chloro, and trifluoromethyl groups on both the hydrazide and ketone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocycle synthesis plays in accelerating drug development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall cost of goods sold.
