Scalable Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial API Production
Scalable Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical cores in bioactive molecules. Patent CN113045489B introduces a significant advancement in the preparation of 3-arylquinolin-2(1H)one derivatives, a class of compounds renowned for their diverse biological activities ranging from antibiotic to antitumor properties. This innovative methodology leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally alters the traditional approach to constructing the quinolinone ring system. By utilizing benzisoxazole as a unique dual-purpose reagent acting as both a nitrogen and formyl source, the process circumvents the handling hazards associated with gaseous carbon monoxide while maintaining high reaction efficiency. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more streamlined manufacturing protocols that do not compromise on yield or purity standards.
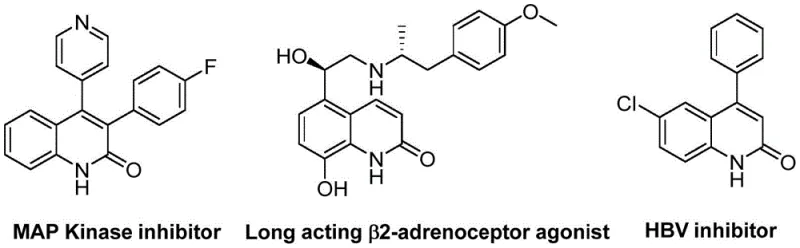
The structural versatility of these derivatives is evident in their application across various therapeutic areas, including their use as MAP Kinase inhibitors and HBV inhibitors, highlighting the commercial value of mastering this synthesis. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies allows us to offer superior supply chain solutions that align with the rigorous demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-documented in academic literature, they often present substantial challenges when translated to an industrial setting. Traditional routes frequently require harsh reaction conditions, including the use of strong acids or bases at elevated temperatures, which can lead to poor functional group tolerance and significant formation of by-products. Furthermore, many conventional carbonylation strategies necessitate the direct use of carbon monoxide gas, a highly toxic and hazardous reagent that demands specialized high-pressure equipment and stringent safety protocols. These factors collectively contribute to increased operational costs, complex waste management issues, and extended lead times for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for critical API precursors.
The Novel Approach
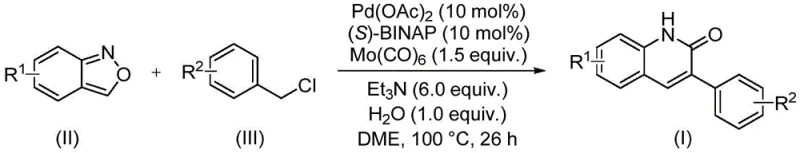
In stark contrast, the method disclosed in patent CN113045489B offers a transformative solution by employing a palladium-catalyzed system that operates under much milder and safer conditions. The core innovation lies in the use of benzisoxazole, which undergoes cleavage to provide the necessary nitrogen and carbonyl components in situ, effectively replacing the need for external CO gas. This reaction proceeds smoothly in ethylene glycol dimethyl ether (DME) at 100°C over a period of 26 hours, utilizing a catalyst system composed of palladium acetate and (S)-BINAP. This approach not only simplifies the operational procedure but also significantly enhances the safety profile of the manufacturing process. The ability to tolerate a wide range of functional groups on both the benzisoxazole and benzyl chloride substrates means that a diverse library of derivatives can be accessed from a single, unified platform, thereby reducing the complexity of process development for new drug candidates.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The mechanistic pathway of this transformation is a sophisticated interplay of organometallic steps that ensure high selectivity and yield. The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from the palladium acetate precursor and the chiral phosphine ligand. Following this, the benzisoxazole ring undergoes a crucial cleavage event, likely facilitated by the metal center or the basic conditions provided by triethylamine, releasing the nitrogen nucleophile and a carbonyl equivalent. This carbonyl species then inserts into the palladium-carbon bond, forming an acyl-palladium intermediate. Subsequent intramolecular cyclization involving the nitrogen atom closes the quinolinone ring, followed by reductive elimination to release the final 3-arylquinolin-2(1H)one product and regenerate the active catalyst. The inclusion of molybdenum hexacarbonyl serves as an additional carbonyl source or promoter, ensuring that the carbonylation step proceeds to completion even without high pressures of CO gas.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-mediated cyclizations. The mild basic conditions and the specific nature of the palladium cycle minimize side reactions such as polymerization or over-alkylation, which are common pitfalls in Friedlander-type syntheses. The use of water as an additive further aids in the hydrolysis steps required for the benzisoxazole ring opening, ensuring clean conversion. For quality control teams, this translates to a cleaner crude reaction profile, which simplifies downstream purification. The ability to achieve high purity specifications through standard column chromatography indicates that the impurity spectrum is manageable and predictable, a critical factor for regulatory compliance in API manufacturing where genotoxic impurities must be strictly controlled.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and reproducibility. The patent outlines a robust protocol where the molar ratio of benzisoxazole to benzyl chloride is optimized to ensure complete consumption of the limiting reagent while minimizing homocoupling side products. The detailed standardized synthesis steps below provide a clear roadmap for laboratory and pilot-scale execution, ensuring that the benefits of this novel chemistry can be realized immediately in your process development workflows.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift towards this palladium-catalyzed route addresses several critical pain points associated with the sourcing and production of complex heterocyclic intermediates. By eliminating the dependency on hazardous gaseous reagents and reducing the number of unit operations required for purification, the overall cost structure of the manufacturing process is significantly optimized. This efficiency gain allows for more competitive pricing models without sacrificing the stringent quality standards required by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and readily available starting materials. Benzisoxazoles and substituted benzyl chlorides are commodity chemicals that can be sourced from multiple suppliers, mitigating the risk of raw material shortages. Furthermore, the elimination of high-pressure carbon monoxide infrastructure reduces capital expenditure requirements for reactor setups. The simplified workup procedure, which involves filtration and standard chromatography rather than complex distillation or crystallization sequences, lowers labor and utility costs. These factors combine to deliver substantial cost savings in API manufacturing, allowing for better margin management in volatile market conditions.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this synthesis route enhances reliability by relying on stable, shelf-stable reagents. Unlike processes that require cryogenic conditions or unstable intermediates, this method operates at a moderate temperature of 100°C using common solvents like DME. The robustness of the catalyst system ensures consistent batch-to-batch performance, reducing the likelihood of failed runs that can disrupt delivery schedules. Additionally, the broad substrate scope means that the same production line can be adapted to synthesize various analogues with minimal changeover time, providing flexibility to respond to changing client demands rapidly.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen challenges, but this methodology is inherently designed for scalability. The reaction conditions are homogeneous and do not involve exothermic hazards typical of nitration or strong acid reactions. From an environmental standpoint, the avoidance of toxic CO gas and the use of catalytic amounts of palladium align with green chemistry principles. Waste generation is minimized due to higher atom economy and cleaner reaction profiles, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. This makes the commercial scale-up of complex pharmaceutical intermediates not only feasible but also environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their specific projects.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for external toxic carbon monoxide gas and simplifying the reagent profile significantly.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and operates under relatively standard heating conditions (100°C), making it highly amenable to commercial scale-up compared to harsh traditional methods.
Q: What is the functional group tolerance of this reaction?
A: The method demonstrates broad substrate scope, tolerating various substituents such as halogens, alkoxy groups, cyano, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate seamlessly into industrial reality. We are committed to delivering high-purity 3-arylquinolin-2(1H)one derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt patented processes like the one described in CN113045489B allows us to offer customized manufacturing solutions that optimize both cost and quality for our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can enhance your supply chain efficiency and drive your product success in the competitive pharmaceutical marketplace.
