Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery and development. A recent technological breakthrough documented in patent CN113045489B introduces a highly efficient preparation method for 3-arylquinolin-2(1H)one derivatives, a class of compounds renowned for their presence in various bioactive molecules and natural products. This novel approach leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm of how these valuable intermediates are constructed. By utilizing benzisoxazole as a dual-purpose reagent acting simultaneously as a nitrogen source and a formyl source, the process eliminates the complexity associated with traditional multi-step syntheses. The significance of this innovation cannot be overstated for R&D directors and procurement specialists alike, as it promises to streamline the supply chain for high-purity pharmaceutical intermediates while reducing the overall environmental footprint of the manufacturing process.
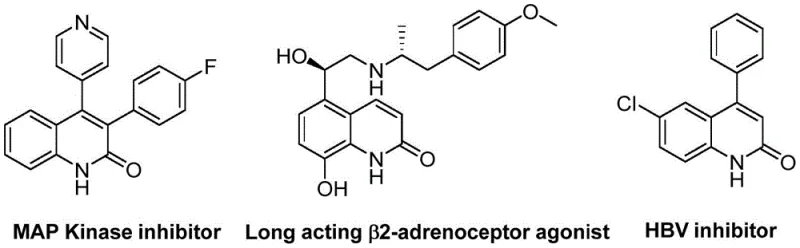
The structural versatility of the quinolin-2(1H)one core is evident in its application across a wide spectrum of therapeutic areas, ranging from antibiotics and antiplatelet agents to antitumor drugs and receptor antagonists. As illustrated in the provided chemical structures, this scaffold is integral to potent MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, underscoring its immense value in modern medicinal chemistry. The ability to access diverse derivatives of this core structure efficiently is therefore a strategic priority for any organization aiming to maintain a competitive edge in drug development. The methodology described in the patent not only addresses the synthetic challenges but also aligns with the growing demand for sustainable and atom-economical processes in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions, which, while effective, often suffer from significant drawbacks when applied to complex molecule synthesis. These traditional routes frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to poor functional group tolerance and the formation of undesirable by-products. Furthermore, many conventional methods necessitate the use of specialized reagents that are either expensive, hazardous, or difficult to source in bulk quantities, thereby creating bottlenecks in the supply chain. Transition metal-catalyzed improvements and ring-closing metathesis have been explored to mitigate some of these issues, yet they often introduce their own complexities, such as the need for rigorous exclusion of moisture or oxygen, and the challenge of removing trace metal residues from the final product to meet stringent pharmaceutical purity standards.
The Novel Approach
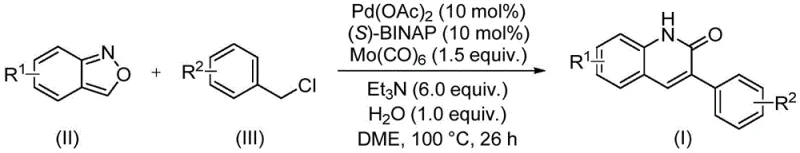
In stark contrast to these legacy methods, the novel palladium-catalyzed aminocarbonylation reaction described in the patent offers a streamlined and operationally simple alternative that overcomes many of the aforementioned limitations. This innovative route utilizes readily available benzisoxazole and benzyl chloride compounds as starting materials, reacting them under relatively mild conditions in the presence of a palladium catalyst system comprising Pd(OAc)2, (S)-BINAP, and Mo(CO)6. The reaction proceeds efficiently in DME solvent with triethylamine as a base and water as an additive, typically at a temperature of 100°C for about 26 hours. What sets this approach apart is the unique role of benzisoxazole, which undergoes ring-opening to provide both the nitrogen atom and the carbonyl carbon required for the quinolinone framework, thereby simplifying the reaction stoichiometry and reducing waste. This method exhibits exceptional substrate scope, tolerating a wide array of functional groups including halogens, alkoxy, cyano, and trifluoromethyl substituents, which is crucial for the late-stage functionalization of drug candidates.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The mechanistic pathway of this transformation involves a sophisticated interplay between the palladium catalyst, the molybdenum carbonyl source, and the benzisoxazole substrate, resulting in the construction of the quinolinone ring with high regioselectivity. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ, forming a benzyl-palladium(II) intermediate. Concurrently, the benzisoxazole undergoes a ring-opening process, likely facilitated by the basic conditions and the coordination to the metal center, releasing the necessary nitrogen and carbon fragments. The molybdenum hexacarbonyl serves as a safe and convenient source of carbon monoxide, which inserts into the palladium-carbon bond to form an acyl-palladium species. Subsequent nucleophilic attack by the nitrogen moiety derived from the benzisoxazole, followed by reductive elimination, yields the final 3-arylquinolin-2(1H)one product and regenerates the palladium catalyst for the next cycle.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of side products that are common in acid-catalyzed cyclizations. The use of a well-defined chiral ligand like (S)-BINAP, although primarily known for asymmetric catalysis, in this context helps to stabilize the palladium center and prevent the aggregation of palladium black, which can lead to catalyst deactivation and heterogeneous impurities. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups on the aromatic rings, ensuring that the impurity profile of the crude product is clean and manageable. This high level of chemoselectivity is paramount for R&D teams who need to ensure that the synthetic route is robust enough to handle diverse substrates without requiring extensive optimization for each new analog, thereby accelerating the lead optimization phase in drug discovery programs.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction parameters to maximize yield and purity. The general procedure involves charging a sealed tube or reactor with the palladium catalyst, ligand, molybdenum source, base, water, and the two primary organic substrates in the appropriate solvent. The mixture is then heated to the specified temperature and maintained for the duration of the reaction, after which standard workup procedures including filtration and chromatography are employed to isolate the pure product. For detailed operational specifics and safety considerations regarding the handling of organometallic reagents, please refer to the standardized synthesis steps provided below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route presents a compelling value proposition driven by significant reductions in raw material costs and process complexity. The reliance on benzisoxazole and benzyl chlorides as starting materials is particularly advantageous because these chemicals are commodity items that are widely available from multiple global suppliers, reducing the risk of supply chain disruptions associated with bespoke or proprietary reagents. Moreover, the elimination of hazardous carbonyl gas sources in favor of solid Mo(CO)6 enhances workplace safety and simplifies regulatory compliance, which translates directly into lower operational overheads and insurance costs. The high yields reported across a broad range of substrates indicate that the process is robust and reliable, minimizing the loss of valuable intermediates and ensuring consistent output quality.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily stemming from the dual functionality of benzisoxazole which acts as both a nitrogen and carbon source, effectively reducing the number of reagents required per mole of product. By avoiding the use of expensive and toxic gaseous carbon monoxide cylinders, the facility saves on both the direct cost of gases and the infrastructure needed for their safe storage and handling. Additionally, the high atom economy of the reaction means that less waste is generated, leading to lower disposal costs and a more sustainable manufacturing profile that aligns with modern green chemistry principles. The use of a recyclable palladium catalyst system further contributes to cost efficiency, as the precious metal can potentially be recovered and reused in subsequent batches.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical intermediates is a top priority for pharmaceutical manufacturers, and this method significantly de-risks the supply chain by utilizing off-the-shelf starting materials. Benzisoxazoles and substituted benzyl chlorides are produced on a large scale for various industries, ensuring that there is no single point of failure in the sourcing strategy. The robustness of the reaction conditions, which tolerate moisture and air to a reasonable extent compared to other organometallic processes, also means that the manufacturing process is less susceptible to minor fluctuations in environmental controls, leading to more predictable production schedules. This reliability allows supply chain planners to optimize inventory levels and reduce the need for safety stock, freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or even tonne scale is straightforward due to the use of standard solvents like DME and common heating protocols that do not require cryogenic conditions or ultra-high pressures. The post-processing steps involve simple filtration and column chromatography, techniques that are easily adapted to continuous processing or large-batch operations without the need for specialized equipment. From an environmental standpoint, the process generates fewer hazardous by-products compared to traditional methods, facilitating easier waste treatment and helping the company meet increasingly stringent environmental regulations. The overall simplicity and safety of the protocol make it an ideal candidate for technology transfer to contract manufacturing organizations (CMOs) or internal production facilities looking to expand their capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of adopting this technology for your specific applications. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, eliminating the need for separate carbonylating agents and simplifying the reaction stoichiometry significantly.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating substituents such as halogens, alkoxy groups, cyano, and trifluoromethyl groups without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: The method utilizes commercially available starting materials and standard purification techniques like column chromatography, making it highly adaptable for scaling from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can deliver high-quality intermediates with consistent purity and on-time delivery. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met whether you are in the early stages of research or full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-arylquinolin-2(1H)one derivatives meets the highest industry standards, giving you confidence in the integrity of your supply chain.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis tailored to your project volume. By partnering with us, you gain access to our deep technical expertise and flexible manufacturing capabilities, allowing you to focus on your core competencies while we handle the complexities of chemical synthesis. Reach out today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with our reliable and efficient solutions.
