Transforming Pharmaceutical Intermediate Production: High-Yield C-O Coupling Process for Commercial Scale Manufacturing
The Chinese patent CN110683926B, granted on November 4, 2022, introduces a groundbreaking methodology for synthesizing carboxylic acid aryl ester compounds through a direct C-O coupling process. This innovative approach represents a significant advancement in pharmaceutical intermediate manufacturing by enabling the direct coupling of carboxylic acids with halogenated aromatic compounds without requiring pre-activation of the carboxyl group. The patented technology utilizes a sophisticated catalyst system comprising palladium complexes with nitrogen-containing ligands (specifically phenanthroline or bipyridine derivatives) in combination with silver salts, achieving remarkable substrate versatility and high yields up to 95%. This development addresses critical challenges in traditional ester synthesis methods, offering pharmaceutical manufacturers a more efficient and reliable pathway for producing complex aryl ester intermediates essential for drug development and production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing carboxylic acid aryl esters have been plagued by significant limitations that hinder their practical application in pharmaceutical manufacturing. Conventional approaches typically require pre-activation of carboxylic acids into more reactive species such as acid chlorides or anhydrides, adding extra synthetic steps and generating additional waste streams. The Itomi group's method using electron-rich dibenzyl azacarbene as a ligand demonstrated restricted substrate scope, being applicable only to methoxy or alkyl-substituted iodobenzenes with bulky aliphatic carboxylic acids. Similarly, MacMillan's photocatalytic approach, while addressing electron-deficient substrates, showed poor compatibility with electron-donating coupling reagents. These limitations resulted in narrow applicability, inconsistent yields, and complex purification requirements that increased production costs and extended manufacturing timelines for pharmaceutical intermediates.

The Novel Approach
The patented methodology overcomes these longstanding challenges through an innovative dual-component catalyst system that simultaneously employs organopalladium complexes and silver salts. This synergistic combination enables direct selective C-O coupling between carboxylic acids and halogenated aromatic compounds without requiring pre-activation of the carboxyl group. The process demonstrates exceptional substrate versatility, accommodating a wide range of carboxylic acids (including aliphatic, aromatic, and complex cyclic structures) and halogenated aromatics (with various electron-donating or electron-withdrawing substituents). The reaction proceeds efficiently under mild conditions with high yields up to 95%, significantly reducing the number of synthetic steps while improving overall process efficiency. This breakthrough represents a paradigm shift in aryl ester synthesis, offering pharmaceutical manufacturers a more sustainable and economically viable production pathway.
Mechanistic Insights into Palladium-Catalyzed C-O Coupling
The core innovation lies in the sophisticated catalytic system that enables direct C-O bond formation through a well-defined mechanism. The organopalladium complex, formed by combining palladium compounds (such as palladium acetate or trifluoroacetate) with nitrogen-containing ligands (specifically phenanthroline or bipyridine derivatives), creates an active catalytic species that facilitates oxidative addition into the carbon-halogen bond of the aromatic substrate. The silver salt acts as a halogen scavenger, promoting the reaction equilibrium toward product formation while preventing catalyst deactivation. This dual-component system stabilizes high-valent palladium intermediates through the rigid nitrogen ligands, enabling selective C-O bond formation without competing side reactions that typically plague traditional coupling methodologies.
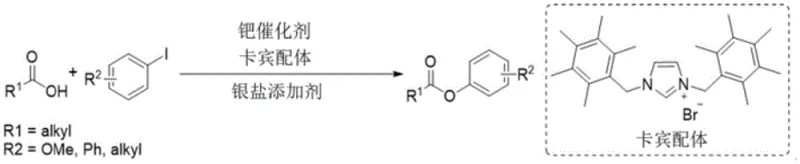
The process achieves exceptional impurity control through several key mechanisms inherent to the catalytic system design. The carefully selected nitrogen ligands (such as 4,7-dimethoxy-1,10-phenanthroline) provide optimal steric and electronic properties that direct the reaction pathway toward selective C-O bond formation while suppressing unwanted decarboxylation or homocoupling side reactions. The silver salt additive not only scavenges halide ions but also modulates the catalyst's reactivity profile, ensuring consistent performance across diverse substrate combinations. This precise control over reaction selectivity results in significantly cleaner reaction profiles with minimal byproduct formation, translating directly to reduced purification requirements and higher final product purity—critical factors for pharmaceutical intermediate manufacturing where stringent quality specifications must be met.
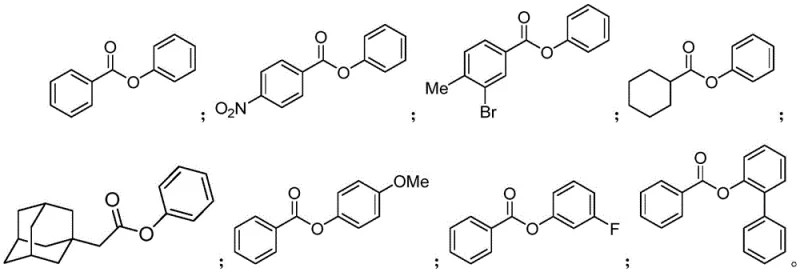
How to Synthesize Carboxylic Acid Aryl Ester Efficiently
This patented methodology provides a robust and scalable approach for synthesizing carboxylic acid aryl esters with exceptional yield and purity characteristics. The process represents a significant improvement over conventional methods by eliminating pre-activation steps and enabling direct coupling between readily available starting materials. The following standardized procedure has been optimized through extensive experimentation to ensure consistent high performance across various substrate combinations while maintaining excellent scalability from laboratory to commercial production environments. Detailed operational parameters and safety considerations are provided in the subsequent step-by-step guide.
- Prepare the reaction mixture by combining carboxylic acid substrate, halogenated aromatic compound, palladium catalyst complex, and silver salt in appropriate molar ratios within a suitable reaction solvent.
- Conduct the reaction under inert gas atmosphere at elevated temperature (100-150°C) for extended duration (8-20 hours) to ensure complete conversion and high yield.
- Purify the crude product through standard workup procedures followed by column chromatography to obtain high-purity aryl ester compound with minimal impurities.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional aryl ester synthesis methods. The elimination of pre-activation steps reduces raw material complexity while improving overall process efficiency, directly impacting cost structures and production timelines. The broad substrate compatibility ensures reliable access to diverse intermediate compounds without requiring significant process revalidation when switching between different product variants—a crucial advantage in dynamic pharmaceutical manufacturing environments where flexibility is paramount.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates multiple processing steps required by conventional methods, significantly reducing both raw material consumption and processing time. By avoiding pre-activation reagents and simplifying purification requirements through superior reaction selectivity, this process achieves substantial cost savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The use of readily available starting materials combined with robust reaction conditions ensures consistent production performance across varying batch sizes and manufacturing sites. This reliability translates to reduced risk of supply disruptions while providing procurement teams with greater flexibility in sourcing raw materials from multiple suppliers without impacting final product quality.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes while maintaining consistent yield and purity profiles. The simplified reaction pathway generates fewer byproducts and waste streams compared to traditional methods, aligning with increasingly stringent environmental regulations while reducing waste treatment costs associated with pharmaceutical intermediate manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the implementation and benefits of this innovative manufacturing process. These inquiries have been compiled based on extensive technical discussions with pharmaceutical manufacturing professionals and reflect the most critical considerations for adopting this technology in commercial production environments.
Q: How does this palladium-catalyzed process achieve higher substrate compatibility compared to conventional methods?
A: The innovative combination of palladium complexes with nitrogen ligands (phenanthroline or bipyridine derivatives) and silver salts enables selective C-O coupling across diverse carboxylic acid and halogenated aromatic substrates, overcoming limitations of previous methods that were restricted to specific substrate types.
Q: What are the key advantages of this process for pharmaceutical manufacturing supply chains?
A: This process eliminates the need for pre-activated carboxylate species, simplifies the synthetic route, and provides consistent high yields (up to 95%) across various substrates, significantly enhancing supply chain reliability and reducing production lead times for critical pharmaceutical intermediates.
Q: How does the catalyst system contribute to impurity profile control in the final product?
A: The carefully designed palladium-nitrogen ligand complex stabilizes high-valent palladium intermediates, promoting selective C-O bond formation while minimizing side reactions that typically generate impurities in traditional coupling processes, resulting in superior product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Aryl Ester Supplier
Our patented C-O coupling technology represents a significant advancement in the synthesis of complex aryl ester intermediates essential for pharmaceutical development and production. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless technology transfer from laboratory to full-scale manufacturing environments. Our rigorous QC labs maintain stringent purity specifications through advanced analytical capabilities, guaranteeing consistent product quality that meets or exceeds pharmaceutical industry requirements for critical intermediates.
We invite you to explore how our innovative manufacturing capabilities can enhance your supply chain resilience and reduce production costs for complex aryl ester intermediates. Request a Customized Cost-Saving Analysis from our technical procurement team to evaluate specific benefits for your manufacturing needs, including detailed COA data and route feasibility assessments tailored to your production requirements.
