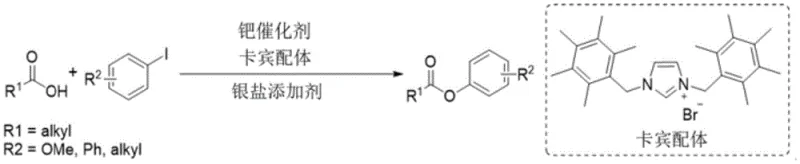
Advanced Palladium-Catalyzed Synthesis of Carboxylic Acid Aryl Esters for Pharmaceutical Applications
The landscape of pharmaceutical synthesis is constantly evolving, driven by the need for more efficient and versatile methods to construct complex molecular architectures. A significant breakthrough in this domain is documented in Chinese Patent CN110683926B, which details a novel preparation method for carboxylic acid aryl ester compounds. This technology addresses a long-standing challenge in organic chemistry: the direct and selective formation of carbon-oxygen (C-O) bonds between carboxylic acids and halogenated aromatic hydrocarbons. Unlike traditional esterification techniques that often require harsh conditions or multiple activation steps, this patented approach utilizes a sophisticated catalytic system comprising an organopalladium complex and a silver salt additive. The result is a robust synthetic route capable of accommodating a wide variety of substrates while achieving exceptional yields, reportedly reaching as high as 95% in optimized examples. For R&D directors and procurement managers in the fine chemical industry, this represents a pivotal shift towards more streamlined and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl esters has been fraught with limitations regarding substrate scope and reaction efficiency. Prior art, such as the work by Professor Itomi's group, relied on electron-rich dibenzyl azacarbene ligands with palladium catalysts. While effective for specific scenarios, this methodology was severely restricted to electron-rich iodobenzenes and bulky aliphatic carboxylic acids, failing to deliver consistent results across a broader chemical space. Similarly, recent advancements by Professor MacMillan's group introduced photocatalytic energy transfer pathways using nickel catalysis. Although this expanded the scope to include electron-deficient bromobenzenes, the system demonstrated poor compatibility with electron-donating coupling reagents. These conventional methods often suffer from narrow applicability, requiring specific electronic properties on the aromatic ring to proceed, which limits their utility in the diverse synthesis of drug candidates where functional group tolerance is paramount.
The Novel Approach
The method disclosed in patent CN110683926B overcomes these historical bottlenecks by employing a dual-component catalytic system that facilitates direct selective C-O coupling. By combining an organopalladium complex formed from a palladium compound and a nitrogen-containing ligand with a silver salt, the reaction achieves remarkable versatility. This system is not constrained by the electronic nature of the aryl halide, successfully coupling both electron-rich and electron-deficient substrates. Furthermore, it accommodates a vast array of carboxylic acid derivatives, including those with significant steric hindrance. The integration of the silver salt acts as a crucial halogen scavenger, driving the equilibrium forward and ensuring high conversion rates. This broad compatibility makes the process an ideal solution for cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the need for multiple synthetic routes to access different structural analogs.
Mechanistic Insights into Pd-Catalyzed Direct C-O Coupling
The core of this technological advancement lies in the precise engineering of the catalytic cycle. The organopalladium complex is generated in situ by compounding a palladium source, such as palladium acetate or palladium trifluoroacetate, with rigid nitrogen-containing ligands like o-phenanthroline or bipyridine derivatives. These ligands play a critical role in stabilizing high-valent palladium intermediates that are essential for the oxidative addition and reductive elimination steps of the catalytic cycle. The rigidity and electron-rich nature of ligands such as 4,7-dimethoxy-1,10-phenanthroline enhance the stability of the active catalyst species, preventing premature decomposition and ensuring sustained turnover numbers. This stabilization is key to maintaining high reaction rates even with sterically demanding substrates, a common failure point in less optimized systems.
Simultaneously, the inclusion of a silver salt additive serves a dual purpose in the reaction mechanism. Primarily, it acts as a halogen scavenger, precipitating the halide ion released during the oxidative addition of the aryl halide to the palladium center. This removal of the halide byproduct shifts the reaction equilibrium towards the formation of the desired ester product, effectively suppressing reverse reactions and side products. Additionally, the silver species may participate in transmetallation steps or assist in the activation of the carboxylic acid substrate. The synergy between the stabilized palladium catalyst and the halogen-scavenging silver salt creates a highly efficient pathway for C-O bond formation. This mechanistic robustness ensures that impurity profiles remain clean, which is a critical factor for regulatory compliance in the production of high-purity OLED materials or API intermediates.
How to Synthesize Carboxylic Acid Aryl Esters Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The process begins with the selection of appropriate starting materials, where the carboxylic acid and aryl halide are mixed with the pre-formed or in-situ generated catalyst system. The patent specifies optimal molar ratios, typically ranging from 1:1.1 to 1:3 for the substrates, ensuring that the limiting reagent is fully consumed. The reaction is conducted in polar or non-polar aprotic solvents such as trifluorotoluene or toluene, which provide the necessary solubility for all components while remaining inert under the reaction conditions. Maintaining an inert atmosphere is non-negotiable to prevent catalyst oxidation.
- Prepare the reaction mixture by combining the carboxylic acid substrate, aryl halide substrate, organopalladium complex catalyst, and silver salt additive in a suitable reaction solvent.
- Ensure the reaction vessel is purged with an inert gas such as argon or nitrogen to maintain an oxygen-free environment throughout the process.
- Heat the mixture to a temperature between 100°C and 150°C and stir for 8 to 20 hours to facilitate the direct C-O coupling reaction.
Commercial Advantages for Procurement and Supply Chain Teams
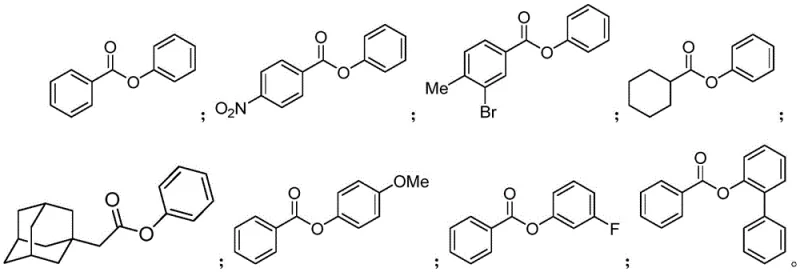
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the simplification of the supply chain for raw materials. Since the method accepts a wide range of commercially available carboxylic acids and aryl halides without requiring specialized pre-functionalization, sourcing becomes significantly more straightforward and resilient. This flexibility reduces the risk of supply disruptions caused by the unavailability of niche activated intermediates. Furthermore, the high yields reported, often exceeding 90% for standard substrates, translate directly into reduced material waste and lower overall consumption of starting materials per kilogram of final product. This efficiency is a key driver for cost reduction in fine chemical manufacturing, allowing for more competitive pricing structures without compromising on quality margins.
- Cost Reduction in Manufacturing: The elimination of pre-activation steps for carboxylic acids removes entire unit operations from the production workflow, significantly lowering energy consumption and labor costs. By avoiding the use of expensive coupling reagents or harsh activating agents typically required in traditional esterification, the process inherently reduces the bill of materials. The high atom economy of the direct coupling reaction ensures that a greater proportion of the input mass ends up in the final product, minimizing waste disposal costs. Additionally, the use of relatively common nitrogen ligands and silver salts, which can potentially be recovered or optimized for loading, prevents the cost escalation often associated with exotic catalyst systems.
- Enhanced Supply Chain Reliability: The broad substrate scope means that manufacturers are not locked into a single source for highly specific precursors. If a particular aryl halide becomes scarce, the robustness of the catalytic system often allows for the use of alternative halogenated analogs (e.g., switching from iodide to bromide with minor adjustments) without a complete process redesign. This adaptability enhances the resilience of the supply chain against market volatility. Moreover, the reaction conditions, while requiring elevated temperatures, utilize standard reactor equipment found in most multipurpose chemical plants, reducing the need for capital investment in specialized photochemical or cryogenic infrastructure.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the catalytic system and the use of standard organic solvents. The reaction does not generate hazardous gaseous byproducts, simplifying off-gas treatment requirements. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, which reduces the solvent load and energy intensity of downstream purification processes like column chromatography or recrystallization. This aligns well with modern green chemistry initiatives, helping companies meet stringent environmental regulations while maintaining high throughput for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this palladium-catalyzed esterification technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction parameters and scope. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: What are the key advantages of this Pd-catalyzed method over traditional esterification?
A: This method enables direct C-O coupling between carboxylic acids and aryl halides without requiring pre-activation of the acid, offering broader substrate compatibility and higher yields up to 95%.
Q: Which ligands are most effective for stabilizing the palladium catalyst in this process?
A: Nitrogen-containing ligands such as o-phenanthroline compounds (e.g., 4,7-dimethoxy-1,10-phenanthroline) and bipyridine compounds are highly effective at stabilizing high-valent palladium intermediates.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard solvents and commercially available reagents under inert gas conditions, making it highly suitable for scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Aryl Ester Supplier
The technological potential of this direct C-O coupling method is immense, offering a pathway to synthesize complex ester motifs that are prevalent in modern medicinal chemistry. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of carboxylic acid aryl ester meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply continuity and quality consistency in the global drug supply chain.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments for your desired intermediates. Let us partner with you to drive innovation and efficiency in your chemical manufacturing operations.
