Advanced Palladium-Catalyzed Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer enhanced biological profiles and synthetic accessibility. Patent CN114163436A introduces a significant advancement in this domain by disclosing a series of diarylmethane derivatives containing an indolizine core. These compounds are not merely structural variants but represent a strategic evolution in drug design, combining the pharmacophore of indolizine with benzofuran or indole moieties to achieve dual antibacterial and anticancer activities. Unlike previous iterations which often suffered from limited biological scope, such as the single-activity sPLA2 inhibition seen in earlier analogs, this new class of molecules offers a broader therapeutic potential. As a reliable pharmaceutical intermediate supplier, understanding the structural nuances defined in this patent is crucial for developing next-generation therapeutics. The general structural formulas (I) and (II) presented in the patent highlight the versatility of the scaffold, allowing for diverse substitutions at the R1 through R4 positions to fine-tune pharmacokinetic properties.
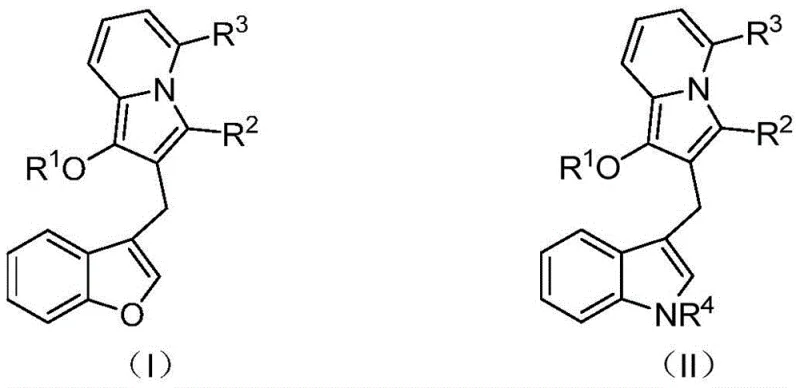
The significance of this invention extends beyond mere biological activity; it addresses a critical gap in the availability of high-purity intermediates for complex drug synthesis. The ability to access these fused heterocyclic systems efficiently opens new avenues for medicinal chemists aiming to optimize lead compounds. For R&D directors focusing on oncology and infectious disease pipelines, these derivatives provide a robust starting point for structure-activity relationship (SAR) studies. The presence of both indolizine and benzofuran/indole rings creates a rigid, planar architecture that is often favorable for intercalating into DNA or binding to specific enzyme active sites, thereby enhancing potency. Furthermore, the patent explicitly mentions their utility as fluorescent probes and organic synthesis intermediates, indicating a multifunctional application profile that adds value to the supply chain for specialty chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diarylmethane structures containing fused nitrogen heterocycles has been fraught with challenges. Conventional routes often rely on multi-step sequences that require harsh reaction conditions, protecting group manipulations, and the use of stoichiometric amounts of toxic reagents. These traditional methods frequently suffer from poor regioselectivity, leading to difficult-to-separate isomeric mixtures that compromise the overall purity of the final API intermediate. Moreover, the construction of the indolizine ring system typically involves cyclization reactions that are sensitive to substrate electronics, limiting the scope of compatible functional groups. This lack of generality forces process chemists to develop bespoke routes for each analog, drastically increasing the time and cost associated with drug discovery. Additionally, many older protocols utilize heavy metal promoters that are difficult to remove to the stringent levels required by regulatory bodies, creating a bottleneck in the commercialization of these candidates.
The Novel Approach
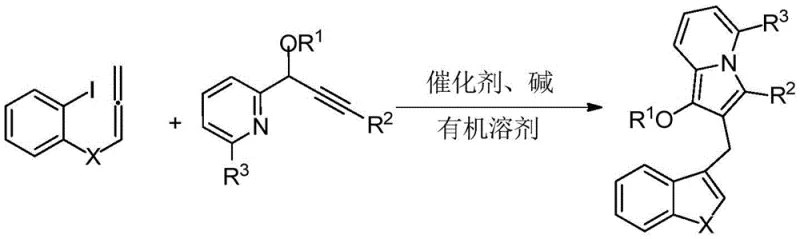
In stark contrast, the methodology described in CN114163436A offers a streamlined, one-pot solution that elegantly bypasses these historical hurdles. The novel approach employs a palladium-catalyzed serial cyclization reaction that constructs the complex indolizine-diarylmethane skeleton in a single operational step. By utilizing 2-iodophenyl allene compounds and propargyl pyridine derivatives as building blocks, the reaction achieves high atom economy and excellent functional group tolerance. This transition metal-catalyzed cascade allows for the simultaneous formation of multiple bonds and rings, significantly reducing the number of isolation steps and solvent consumption. The mild reaction conditions, typically ranging from 60°C to 90°C, ensure that sensitive functional groups remain intact, thereby expanding the chemical space accessible to researchers. This efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it minimizes waste generation and maximizes throughput.
Mechanistic Insights into Pd-Catalyzed Serial Cyclization
The core of this technological breakthrough lies in the sophisticated palladium-catalyzed mechanism that drives the formation of the indolizine core. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the 2-iodophenyl allene substrate. This generates a reactive aryl-palladium intermediate which subsequently undergoes migratory insertion into the allene moiety. The resulting organopalladium species then engages in an intramolecular nucleophilic attack or further insertion with the alkyne component of the propargyl pyridine. This cascade sequence is meticulously orchestrated to ensure the correct regiochemistry, favoring the formation of the desired five-membered indolizine ring over potential six-membered byproducts. The presence of the base, such as potassium carbonate, plays a pivotal role in facilitating the final elimination or deprotonation steps that restore aromaticity and release the active catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise approaches. Because the cyclization occurs in a concerted or rapid sequential manner within the coordination sphere of the metal, there is less opportunity for intermolecular side reactions or polymerization of the reactive allene and alkyne components. The choice of ligand on the palladium center, such as triphenylphosphine in tetrakis(triphenylphosphine)palladium, further tunes the electronic and steric environment to suppress off-cycle pathways. This inherent selectivity results in a cleaner crude reaction profile, which simplifies downstream purification. For quality control teams, this means that achieving the stringent purity specifications required for clinical grade materials becomes more predictable and less resource-intensive. The robustness of this catalytic cycle against variations in substrate substitution patterns ensures consistent quality across different batches of production.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The patent outlines a generalized protocol that serves as a robust foundation for process development. Typically, the reaction is conducted in polar aprotic solvents like N,N-dimethylformamide (DMF), which effectively solubilizes both the organic substrates and the inorganic base. The molar ratio of the palladium catalyst to the substrate is kept low, often between 0.02:1 and 0.2:1, demonstrating the high turnover number of the catalytic system. Maintaining an inert atmosphere, preferably nitrogen, is critical to prevent oxidation of the palladium(0) species and degradation of the sensitive allene starting material. Following the reaction period of 4 to 12 hours, the workup involves standard aqueous extraction techniques followed by chromatographic purification.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst (e.g., Pd(PPh3)4), alkali (e.g., K2CO3), and organic solvent (e.g., DMF) in a reaction vessel.
- Stir and react the mixture for 4-12 hours at a temperature range of 60-90°C under an inert nitrogen atmosphere to facilitate the cascade cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying over anhydrous sodium sulfate, filtration, solvent removal, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits. The primary advantage stems from the utilization of commercially available and relatively inexpensive starting materials. 2-iodophenyl allene compounds and propargyl pyridines are accessible from bulk chemical suppliers, reducing the risk of raw material shortages that often plague specialized synthetic routes. Furthermore, the one-pot nature of the reaction eliminates the need for intermediate isolation, which significantly reduces solvent usage, labor costs, and processing time. This streamlining of the manufacturing process leads to substantial cost savings without compromising the quality of the final product. The ability to produce these high-value intermediates more efficiently allows for better margin management in the production of downstream APIs.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the use of a catalytic amount of palladium rather than stoichiometric reagents drastically lowers the bill of materials. By avoiding expensive protecting group strategies and harsh reagents, the overall cost of goods sold (COGS) is significantly optimized. The simplified workup procedure, which relies on common extraction and distillation techniques rather than specialized equipment, further contributes to operational expenditure reductions. This economic efficiency makes the technology attractive for large-scale production where even marginal savings per kilogram translate into significant financial gains.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents like DMF and ethyl acetate ensures a stable supply chain that is less vulnerable to geopolitical disruptions or niche vendor dependencies. The robustness of the reaction conditions, which tolerate a wide range of temperatures and concentrations, allows for flexible manufacturing scheduling. This flexibility is crucial for meeting tight delivery deadlines and adapting to fluctuating market demands. Additionally, the high yields reported in the patent examples suggest a reliable process that minimizes batch failures, ensuring a consistent flow of material to downstream customers.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is straightforward due to the absence of hazardous reagents or extreme conditions. The use of a heterogeneous base like potassium carbonate simplifies filtration and waste handling compared to soluble strong bases. The reduced solvent intensity and higher atom economy align with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. This sustainability profile enhances the corporate social responsibility (CSR) standing of the manufacturing entity and future-proofs the supply chain against tightening regulatory frameworks regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine derivatives. These answers are derived directly from the technical disclosures in patent CN114163436A and represent the current state of the art in this specific chemical domain. Understanding these details is essential for partners looking to integrate these intermediates into their own development pipelines or commercial manufacturing processes.
Q: What are the key biological activities of these indolizine derivatives?
A: According to patent CN114163436A, these derivatives exhibit dual antibacterial and anticancer activities, overcoming the single-activity limitation of prior art compounds like sPLA2 inhibitors.
Q: What catalysts are suitable for this synthesis?
A: The process utilizes palladium salt catalysts such as tetrakis(triphenylphosphine)palladium, tris(dibenzylideneacetone)dipalladium, or bis(dibenzylideneacetone)palladium.
Q: Is this process scalable for industrial production?
A: Yes, the method uses cheap, readily available raw materials and standard organic solvents like DMF, with a simple workup procedure involving extraction and column chromatography, making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Diarylmethane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN114163436A for the development of novel anticancer and antibacterial agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to global market supply. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of indolizine intermediate meets the highest standards of quality and consistency required by international regulatory agencies. We are committed to supporting your R&D efforts with reliable, high-quality materials that accelerate your time to market.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce costs while securing a reliable source of these critical pharmaceutical intermediates.
