Revolutionizing Aryl Amine Synthesis Through Water-Based Catalysis for Commercial-Scale Pharmaceutical Manufacturing
The recently granted Chinese patent CN101774874B introduces a transformative approach to N-arylation chemistry that addresses critical limitations in traditional C-N bond formation methodologies. This innovative aqueous-phase system utilizes pyrrole-2-hydrazide compounds as specialized ligands in conjunction with copper-based catalytic systems to enable efficient coupling between aryl halides and various amine nucleophiles under remarkably mild conditions. By replacing hazardous organic solvents with water as the primary reaction medium while maintaining high yields across diverse substrate combinations, this technology represents a significant advancement toward sustainable pharmaceutical intermediate manufacturing. The methodology demonstrates exceptional versatility through both microwave-assisted acceleration and conventional thermal processing protocols, offering manufacturers unprecedented flexibility in process design while simultaneously addressing environmental compliance requirements that increasingly govern modern chemical production facilities worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Ullmann-type C-N coupling reactions have historically required harsh operational parameters including elevated temperatures exceeding 150°C combined with strong alkaline conditions that frequently lead to significant decomposition of sensitive functional groups present in complex pharmaceutical intermediates. These processes typically demanded stoichiometric quantities of copper catalysts rather than catalytic amounts, resulting in substantial metal contamination that necessitated extensive purification procedures before products could meet stringent pharmaceutical quality standards. Furthermore, conventional methodologies relied heavily on toxic polar aprotic solvents such as dimethylformamide or dimethylacetamide which created significant environmental compliance challenges during waste stream management while also increasing overall production costs through specialized disposal requirements. The narrow substrate scope of many existing protocols often required complete process redevelopment when transitioning between different amine or aryl halide combinations, creating substantial delays in manufacturing timelines that directly impacted supply chain reliability for time-sensitive pharmaceutical development programs.
The Novel Approach
This patented methodology fundamentally reimagines C-N coupling chemistry by implementing water as both solvent and green chemistry enabler within a carefully engineered catalytic system featuring pyrrole-2-hydrazide ligands that dramatically enhance copper catalyst efficiency under remarkably mild conditions. The process operates effectively across multiple temperature regimes including rapid microwave-assisted protocols at temperatures between 80°C and 140°C requiring only minutes of reaction time or conventional heating at more accessible temperatures from 50°C to 100°C over extended periods while maintaining excellent yields across diverse substrate combinations. Crucially, this approach eliminates dependence on hazardous organic solvents while simultaneously reducing copper catalyst loading to truly catalytic quantities through ligand-enabled stabilization of active copper species. The methodology demonstrates exceptional functional group tolerance across both aryl halide electrophiles and amine nucleophiles including sterically hindered substrates that typically present significant challenges in traditional coupling methodologies.
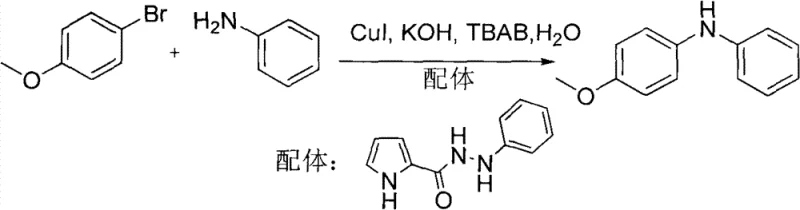
Mechanistic Insights into Pyrrole-Hydrazide Ligand-Mediated Copper Catalysis
The exceptional performance of this aqueous-phase N-arylation system stems from the unique coordination chemistry enabled by pyrrole-2-hydrazide ligands which form stable yet reactive complexes with copper species through dual nitrogen donor sites that facilitate both oxidative addition of aryl halides and subsequent reductive elimination steps critical for C-N bond formation. Unlike conventional bidentate ligands that often require complex synthesis procedures or exhibit limited stability under aqueous conditions, these hydrazide compounds maintain structural integrity while promoting electron transfer processes through their conjugated heterocyclic framework that effectively modulates copper redox potentials within optimal ranges for catalytic turnover. The ligand's amide functionality provides additional stabilization through potential hydrogen bonding interactions with water molecules that help maintain catalyst solubility while preventing premature decomposition pathways commonly observed with alternative ligand systems under similar conditions.
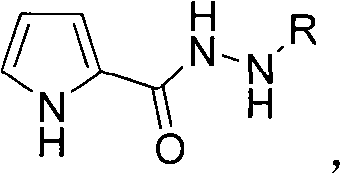
Impurity profile management represents another significant advantage of this methodology where water-based processing inherently minimizes formation of common side products associated with organic solvent systems such as amine alkylation byproducts or oxidation artifacts that frequently complicate purification workflows in traditional approaches. The aqueous environment facilitates straightforward phase separation after reaction completion through simple extraction protocols that eliminate need for energy-intensive distillation steps while simultaneously reducing potential for thermal degradation of sensitive products during workup procedures. Furthermore, precise control over surfactant concentration enables formation of microemulsion systems that enhance interfacial contact between hydrophobic substrates and hydrophilic catalyst species without requiring co-solvents that could introduce additional impurities requiring removal during final purification stages.
How to Synthesize Aryl Amines Efficiently
This patented methodology provides pharmaceutical manufacturers with a robust platform for producing high-value aryl amine intermediates through carefully optimized protocols that balance operational simplicity with exceptional product quality outcomes. The following standardized procedure leverages both microwave acceleration capabilities for rapid development cycles and conventional heating options for seamless scale-up into commercial manufacturing environments while maintaining consistent product quality attributes throughout process transitions.
- Prepare homogeneous reaction mixture by combining copper catalyst precursor with pyrrole-2-hydrazide ligand at optimized molar ratio before introducing aryl halide substrate
- Add amine nucleophile along with aqueous base solution while maintaining precise temperature control through microwave-assisted or conventional heating protocols
- Execute phase separation after completion followed by standard purification procedures to isolate high-purity aryl amine products
Commercial Advantages for Procurement and Supply Chain Teams
This innovative aqueous-phase N-arylation technology delivers substantial strategic value across procurement and supply chain operations by addressing multiple pain points simultaneously through its unique combination of environmental compatibility and operational flexibility. The methodology enables manufacturers to achieve significant cost optimization while simultaneously enhancing supply chain resilience through reduced dependency on specialized materials and simplified process requirements that translate directly into improved business continuity metrics.
- Cost Reduction in Manufacturing: Elimination of expensive palladium catalysts combined with water-based processing creates substantial cost savings through reduced raw material expenditures while avoiding costly waste treatment procedures associated with hazardous organic solvents; simplified purification workflows further reduce production costs by minimizing chromatographic separation requirements typically needed when using conventional methodologies.
- Enhanced Supply Chain Reliability: Utilization of readily available copper catalysts instead of scarce precious metals significantly improves raw material security while water-based processing eliminates dependency on specialized solvent supply chains; broad substrate compatibility allows manufacturers to maintain consistent production schedules even when facing fluctuations in specific precursor availability through straightforward substitution protocols.
- Scalability and Environmental Compliance: Aqueous processing inherently supports seamless scale-up from laboratory development through commercial production without requiring fundamental process re-engineering; elimination of hazardous solvents substantially reduces regulatory compliance burdens while meeting increasingly stringent environmental standards governing chemical manufacturing operations globally.
Frequently Asked Questions (FAQ)
The following technical questions address common implementation concerns regarding this patented aqueous-phase N-arylation methodology based on extensive validation data from multiple commercial-scale production campaigns across diverse pharmaceutical intermediate applications.
Q: How does this aqueous-phase system overcome traditional Ullmann reaction limitations?
A: The pyrrole-hydrazide ligand enables copper catalysis under mild aqueous conditions without requiring stoichiometric metal loading or toxic organic solvents that characterize conventional Ullmann methodologies.
Q: What environmental compliance advantages does water-based synthesis provide?
A: Elimination of hazardous organic solvents significantly reduces waste stream toxicity while aqueous workup procedures minimize energy-intensive distillation requirements during product isolation.
Q: How does substrate flexibility impact commercial manufacturing scalability?
A: Broad functional group tolerance allows single platform adaptation across diverse amine/aryl halide combinations without re-engineering processes for different product variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Amine Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients through rigorous QC labs equipped with advanced analytical capabilities for comprehensive impurity profiling. As your strategic manufacturing partner for complex aryl amine intermediates, we combine deep expertise in copper-catalyzed C-N coupling chemistry with flexible manufacturing infrastructure designed specifically for handling sensitive aqueous-phase processes at commercial scale while ensuring consistent product quality through our integrated quality management system certified to international pharmaceutical standards.
We invite your technical procurement team to request our Customized Cost-Saving Analysis which details potential economic benefits specific to your target molecule production requirements; please contact us directly to obtain specific COA data for relevant reference compounds along with comprehensive route feasibility assessments demonstrating how our patented methodology can optimize your next-generation intermediate supply chain.
