Revolutionizing Alpha-Carbonyl Amide Production with Green Metal-Free Oxidative Ring Opening Technology
Introduction to Advanced Alpha-Carbonyl Amide Synthesis
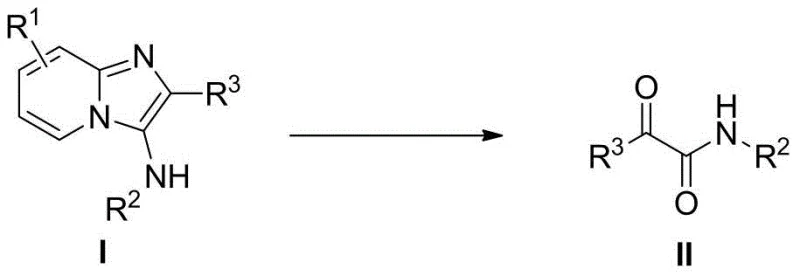
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN109096139B, which discloses a novel preparation method for alpha-carbonyl amide derivatives. This technology represents a paradigm shift by utilizing a pyridoimidazole amine compound as a precursor, which undergoes an oxidative ring-opening reaction in the presence of water and an oxidant. Unlike traditional methods that often rely on harsh conditions or expensive metal catalysts, this invention achieves the insertion of an oxygen atom directly from water sources under mild conditions. For R&D directors and procurement specialists seeking reliable suppliers of high-purity intermediates, this metal-free approach offers a compelling value proposition, combining operational simplicity with exceptional substrate universality. The ability to recycle byproducts back into raw materials further enhances the atom economy, positioning this method as a cornerstone for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-carbonyl amide derivatives has been fraught with challenges that impact both cost and environmental compliance. Traditional strategies often involve small molecule coupling reactions or oxidation processes catalyzed by transition metals such as copper. For instance, prior art describes methods using substituted aromatic acetamide oxidized under copper catalysis or coupling arynes with amines using related auxiliary agents. These conventional pathways frequently necessitate stringent reaction conditions, including anhydrous and oxygen-free environments, which significantly increase operational complexity and infrastructure costs. Furthermore, the reliance on transition metal catalysts introduces a critical bottleneck in the purification process; removing trace heavy metals to meet pharmaceutical grade specifications often requires additional, costly steps like chelation or extensive chromatography. The generation of hazardous waste associated with metal salts and the difficulty in scaling these sensitive reactions further limit their commercial viability for large-scale production of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN109096139B offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic workflow. By employing a pyridoimidazole amine compound as the starting material, the process achieves selective ring opening and simultaneous oxygen insertion without the need for any metal catalyst. The reaction utilizes water—either added directly or sourced from crystalline hydrates like TsOH·H2O—as the oxygen donor, a feature that drastically reduces reagent costs and improves safety profiles. As illustrated in the general reaction scheme below, the transformation proceeds smoothly in common organic solvents such as 1,2-dichloroethane at temperatures ranging from room temperature to 100°C. This robustness eliminates the need for specialized inert atmosphere equipment, allowing for simpler reactor setups and reduced energy consumption. The broad substrate scope, accommodating various substituted phenyl and heterocyclic groups, ensures that this technology can be adapted for a wide array of high-value drug intermediates.
Mechanistic Insights into Metal-Free Oxidative Ring Opening
The core innovation of this technology lies in its unique mechanistic pathway, which bypasses the need for transition metal mediation. The reaction initiates with the interaction between the pyridoimidazole amine substrate and the hypervalent iodine oxidant, typically iodobenzene diacetate (PhI(OAc)2). This oxidant activates the substrate towards nucleophilic attack by water molecules. The water source, which can be as simple as the water of crystallization from p-toluenesulfonic acid monohydrate (TsOH·H2O), provides the oxygen atom that is eventually incorporated into the alpha-carbonyl position of the final amide product. This direct utilization of water as an oxygen source is a remarkable feat of atom economy, avoiding the use of pre-functionalized oxygen donors that generate stoichiometric waste. The ring-opening step is highly selective, cleaving the imidazole ring specifically to yield the desired alpha-keto amide structure while releasing a pyridylamine byproduct. Crucially, this byproduct is not merely waste; it can be recycled and reused as a raw material for synthesizing the starting pyridoimidazole amine, creating a closed-loop system that maximizes resource efficiency and minimizes environmental impact.
From an impurity control perspective, the absence of metal catalysts inherently reduces the risk of metal-related impurities, which are strictly regulated in pharmaceutical products. The reaction profile is clean, with the primary side reactions being minimal due to the specificity of the oxidative ring opening. The use of mild acidic conditions provided by TsOH helps in protonating intermediates, facilitating the ring cleavage without promoting excessive degradation of sensitive functional groups like nitro or halogen substituents. This tolerance allows for the synthesis of diverse derivatives, including those with electron-withdrawing groups that might be unstable under harsher basic or high-temperature conditions typical of metal-catalyzed couplings. The result is a high-purity crude product that requires less intensive purification, directly translating to higher overall yields and lower production costs for the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize Alpha-Carbonyl Amide Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward, leveraging standard organic synthesis equipment without the need for exotic hardware. The process begins by charging a reaction vessel with the specific pyridoimidazole amine precursor, followed by the addition of the water source and the organic solvent. The choice of solvent is flexible, with 1,2-dichloroethane (DCE) being preferred for its optimal solubility and reaction rate characteristics, though alternatives like dichloromethane or acetonitrile are also viable. Once the mixture is homogenized, the oxidant is introduced, and the reaction is allowed to proceed with stirring. Monitoring is easily achieved via thin-layer chromatography (TLC), with most reactions reaching completion within 1 to 2 hours at ambient or slightly elevated temperatures. For detailed standardized operating procedures and specific stoichiometric ratios tailored to your specific substrate, please refer to the guide below.
- Combine the pyridoimidazole amine starting material with a water source (such as TsOH·H2O) and an organic solvent like 1,2-dichloroethane in a reaction vessel.
- Add the oxidant, typically iodobenzene diacetate (PhI(OAc)2), to the mixture and stir at temperatures ranging from room temperature to 100°C.
- Monitor the reaction progress via TLC, and upon completion, separate the organic phase and purify the target alpha-carbonyl amide derivative using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free oxidative ring-opening technology offers substantial strategic advantages beyond mere technical novelty. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive metals like palladium or copper and the subsequent removal processes required to meet regulatory standards. This simplification of the downstream processing workflow leads to a drastic reduction in manufacturing lead times and operational expenditures. Furthermore, the use of inexpensive and readily available reagents such as TsOH·H2O and PhI(OAc)2 ensures a stable and resilient supply chain, mitigating the risks associated with the volatility of rare metal markets. The robustness of the reaction conditions, which do not require strict anhydrous or oxygen-free environments, lowers the barrier for scale-up, allowing for seamless transition from kilogram-scale development to multi-ton commercial production without the need for specialized reactor modifications.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of costly metal catalysts and the simplification of purification steps. By avoiding the need for heavy metal scavengers or complex chromatographic separations to remove metal residues, manufacturers can achieve significant cost savings in raw materials and labor. Additionally, the high atom economy achieved by recycling the pyridylamine byproduct back into the synthesis of the starting material further reduces the net cost of goods sold (COGS). The mild reaction conditions also translate to lower energy consumption, as there is no need for extreme heating or cooling, contributing to a leaner and more cost-effective manufacturing profile for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are widely available from multiple global suppliers. Unlike specialized ligands or sensitive organometallic catalysts that may have long lead times or single-source dependencies, reagents like iodobenzene diacetate and tosic acid are standard inventory items in the fine chemical industry. This ubiquity ensures that production schedules are not disrupted by raw material shortages. Moreover, the stability of the reaction to ambient moisture and oxygen means that storage and handling requirements for reagents are less stringent, reducing the risk of spoilage and waste during logistics and warehousing, thereby enhancing overall supply chain reliability for complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by its inherent safety and simplicity. The absence of pyrophoric reagents or high-pressure hydrogenation steps reduces the safety hazards typically associated with scale-up, allowing for larger batch sizes in standard glass-lined or stainless steel reactors. From an environmental standpoint, the metal-free nature of the reaction significantly reduces the load of heavy metals in wastewater streams, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The ability to recycle byproducts aligns with green chemistry principles, minimizing waste generation and supporting corporate sustainability goals while maintaining high production throughput for alpha-carbonyl amide derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative ring-opening technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating route feasibility and procurement teams assessing supplier qualifications. The answers highlight the versatility of the method across different substrates and its alignment with modern manufacturing standards.
Q: What is the source of the oxygen atom in this synthesis?
A: According to patent CN109096139B, the oxygen atom inserted into the structure is derived directly from water molecules, which can be introduced via crystalline hydrates like TsOH·H2O or undried solvents, eliminating the need for specialized oxygen donors.
Q: Does this process require transition metal catalysts?
A: No, this method is distinctively metal-free. It avoids the use of copper or other transition metal catalysts commonly found in traditional alpha-carbonyl amide synthesis, thereby simplifying downstream purification and reducing heavy metal contamination risks.
Q: What are the typical reaction conditions?
A: The reaction proceeds under mild conditions, typically between room temperature and 100°C, without the need for strict anhydrous or oxygen-free environments, making it highly suitable for scalable industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative ring-opening technology described in CN109096139B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in practical manufacturing. Our state-of-the-art facilities are equipped to handle metal-free syntheses with rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch of alpha-carbonyl amide derivative meets the highest international quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this metal-free method can optimize your budget without compromising quality. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring a seamless transition from development to commercial supply for your critical drug intermediates.
