Advancing Alpha-Carbonyl Amide Production: A Green CuI-Catalyzed Oxidative Amidation Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex molecular scaffolds. A significant breakthrough in this domain is detailed in patent CN113248458B, which discloses a novel preparation method for alpha-carbonyl amide compounds. These compounds are critical structural motifs found in numerous bioactive molecules, natural products, and active pharmaceutical ingredients (APIs). The traditional reliance on hazardous reagents and harsh conditions has long been a bottleneck for manufacturers aiming to improve their environmental footprint and operational safety. This patent introduces a transformative approach utilizing cuprous iodide (CuI) catalysis under mild conditions, representing a paradigm shift towards greener chemistry in the production of high-value intermediates.
The significance of this technology extends beyond mere academic interest; it addresses practical pain points faced by reliable pharmaceutical intermediate suppliers and global procurement teams. By leveraging oxygen as the sole oxidant and generating nitrogen gas as the only byproduct, the process eliminates the generation of toxic waste streams associated with conventional stoichiometric oxidants. This innovation not only aligns with stringent global environmental regulations but also streamlines the downstream purification processes, thereby enhancing the overall economic viability of producing these essential chemical building blocks for the life sciences sector.
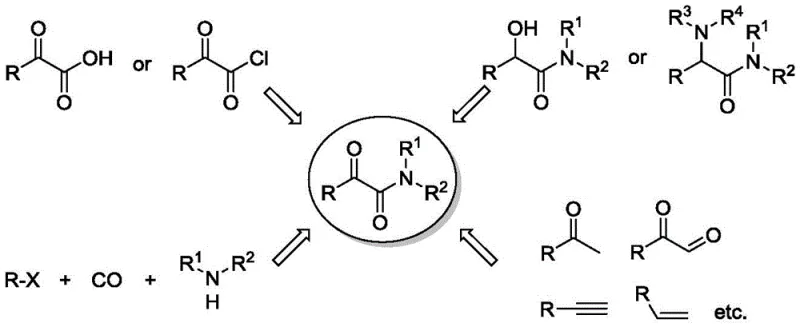
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-carbonyl amides has relied heavily on the condensation of alpha-keto acids or alpha-keto acid chlorides with amines. While chemically straightforward, these methods suffer from severe drawbacks, including the use of highly corrosive acid chlorides and the generation of substantial acidic waste. Alternative oxidative routes, such as the oxidation of alpha-hydroxy amides or alpha-amino amides, often require dangerous oxidizing agents and multi-step sequences that erode overall yield. Furthermore, transition metal-catalyzed amino-biscarbonylation of aryl halides, while useful, frequently struggles with selectivity issues, leading to the formation of unwanted monocarbonylated byproducts that are difficult to separate.
Recent advancements involving copper-catalyzed oxidative amidation have shown promise but often demand complex reaction systems. For instance, prior art methods described in the patent background utilize expensive additives like TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and require elevated temperatures in solvents like toluene. As illustrated in the comparison of existing technologies, these processes involve intricate catalytic cycles and harsh conditions that increase energy consumption and operational risks. The reliance on such complex systems hinders the ability to achieve cost reduction in pharmaceutical intermediate manufacturing, as the removal of metal residues and organic additives adds significant burden to the purification workflow.
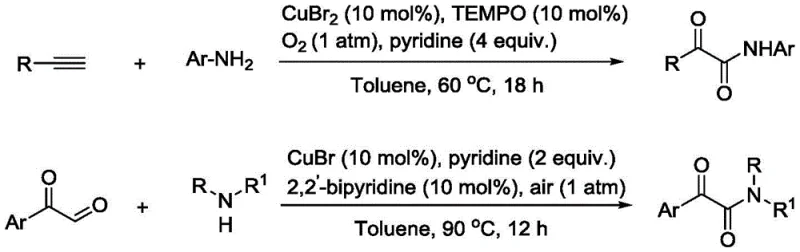
The Novel Approach
In stark contrast to these legacy methods, the technology protected by patent CN113248458B offers a streamlined, single-step oxidative amidation strategy. The core innovation lies in the reaction between an alpha-diazoketone compound and a cyclic secondary amine, catalyzed by cuprous iodide (CuI) with molecular oxygen serving as the terminal oxidant. This method operates remarkably at room temperature (25°C to 35°C), eliminating the need for energy-intensive heating or cooling systems. The reaction proceeds with high selectivity and efficiency, converting the starting materials directly into the desired alpha-carbonyl amide structure with nitrogen gas as the sole gaseous byproduct.
The versatility of this novel approach is demonstrated by its compatibility with a wide range of substrates. The aromatic component (Ar1) can be varied extensively to include unsubstituted phenyl groups, as well as those substituted with electron-donating groups like methyl and methoxy, or electron-withdrawing halogens such as fluorine, chlorine, bromine, and iodine. Additionally, the system accommodates fused ring systems like naphthalene and heterocycles like furan and thiophene. This broad substrate tolerance ensures that the method is not limited to a single niche application but serves as a robust platform for the commercial scale-up of complex pharmaceutical intermediates across diverse therapeutic areas.
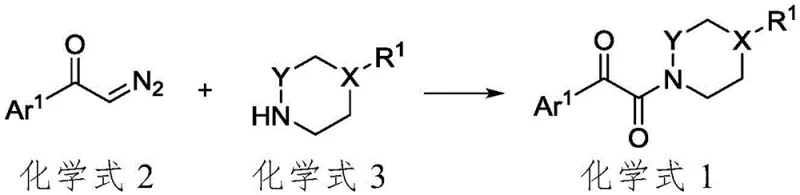
Mechanistic Insights into CuI-Catalyzed Oxidative Amidation
The mechanistic elegance of this transformation centers on the unique role of the cuprous iodide catalyst in activating the alpha-diazoketone species. Under an oxygen atmosphere, the Cu(I) species facilitates the formation of a reactive metal-carbene or radical intermediate from the diazo precursor. This activated species then undergoes nucleophilic attack by the cyclic secondary amine. The presence of molecular oxygen is crucial, as it acts as a hydrogen acceptor to drive the oxidative dehydrogenation necessary to form the final dicarbonyl amide bond. Unlike other copper salts tested in the patent's comparative examples, CuI provides the optimal balance of Lewis acidity and redox potential to sustain this catalytic cycle without decomposing the sensitive diazo functionality prematurely.
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on purity profiles. The reaction is highly atom-economical regarding the non-gaseous components, meaning fewer side reactions occur compared to methods using stoichiometric oxidants which often over-oxidize sensitive functional groups. The patent data indicates that competing catalysts like CuBr, CuCl, or Pd(OAc)2 result in significantly lower yields, suggesting that CuI minimizes the formation of polymeric byproducts or decomposition artifacts. This high selectivity translates to a cleaner crude reaction mixture, simplifying the isolation of high-purity alpha-carbonyl amides and reducing the load on analytical quality control laboratories during batch release.
How to Synthesize Alpha-Carbonyl Amide Efficiently
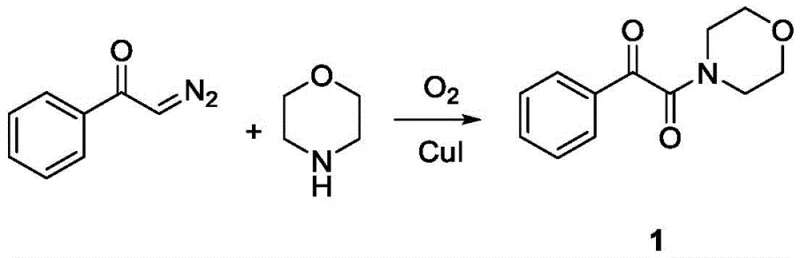
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and safety. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. A representative example involves the synthesis of 1-morpholino-2-phenylethane-1,2-dione, where the reactants are combined in acetonitrile under a balloon of oxygen. The simplicity of the setup allows for rapid screening of different amine and ketone combinations, accelerating the development timeline for new drug candidates.
- Prepare the reaction mixture by combining the cyclic secondary amine (e.g., morpholine), cuprous iodide (CuI) catalyst, and anhydrous acetonitrile solvent in a reaction vessel under an oxygen atmosphere.
- Add the alpha-diazoketone compound dissolved in anhydrous acetonitrile to the mixture and stir at room temperature (25°C to 35°C) for approximately 3 hours.
- Upon completion, purify the resulting alpha-carbonyl amide product using column chromatography with petroleum ether and ethyl acetate as the eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CuI-catalyzed methodology presents a compelling value proposition centered on risk mitigation and cost optimization. The elimination of hazardous reagents like acid chlorides and toxic oxidants drastically reduces the regulatory burden associated with chemical handling and waste disposal. Furthermore, the use of ambient air or pure oxygen as the oxidant removes the dependency on expensive, shelf-limited chemical oxidants, stabilizing the raw material supply chain against market volatility. This stability is crucial for maintaining continuous production schedules for critical API intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the reaction workflow. By operating at room temperature, the method eliminates the energy costs associated with heating reactors to high temperatures or maintaining cryogenic conditions. Additionally, the high catalytic efficiency of CuI means that lower catalyst loadings can potentially be used while maintaining high conversion rates. The absence of complex additives like TEMPO further reduces the bill of materials, leading to substantial cost savings in the overall manufacturing budget without compromising product quality.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically alpha-diazoketones and common cyclic amines like morpholine, are commercially available from multiple global vendors. This commoditization of raw materials prevents supply bottlenecks that often plague proprietary synthetic routes relying on exotic reagents. Moreover, the short reaction time of approximately 3 hours allows for faster throughput in multipurpose reactors, enabling manufacturers to respond more agilely to fluctuating market demands and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the benign nature of the byproducts. Since nitrogen gas is the only byproduct, there is no need for complex aqueous workups to remove inorganic salts or toxic organic waste. This 'green' profile aligns perfectly with modern ESG (Environmental, Social, and Governance) goals, making the supply chain more resilient to tightening environmental regulations. The simplified post-processing also reduces the volume of solvent required for purification, contributing to a lower overall environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within patent CN113248458B, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of using CuI over other copper catalysts for this synthesis?
A: According to patent CN113248458B, cuprous iodide (CuI) demonstrates significantly higher catalytic efficiency compared to alternatives like CuBr, CuCl, or Pd(OAc)2. Comparative examples in the patent show that CuI yields substantially higher product recovery (e.g., 78% vs 18% for CuBr), making it the optimal choice for cost-effective manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It operates at room temperature without the need for heating or refrigeration, uses oxygen (air) as a benign oxidant, and produces only nitrogen gas as a byproduct. These factors simplify safety protocols and reduce energy consumption, facilitating commercial scale-up.
Q: What types of substrates are compatible with this oxidative amidation method?
A: The method exhibits broad substrate scope. It tolerates various substituted phenyl groups (including electron-donating and electron-withdrawing substituents like halogens, methyl, and methoxy), as well as heterocyclic systems like naphthalene, furan, and thiophene. It also works with diverse cyclic secondary amines such as morpholine, piperidine, and pyrrolidine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the CuI-catalyzed oxidative amidation technology described in patent CN113248458B. As a leading CDMO partner, we possess the technical expertise to translate this innovative laboratory protocol into robust, GMP-compliant manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of alpha-carbonyl amide intermediate meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this green chemistry advantage for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your COGS (Cost of Goods Sold). Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you build a more sustainable and efficient supply chain together.
