Advanced Nickel-Catalyzed Thioester Synthesis Enabling Scalable Pharmaceutical Intermediate Manufacturing with Enhanced Purity Control
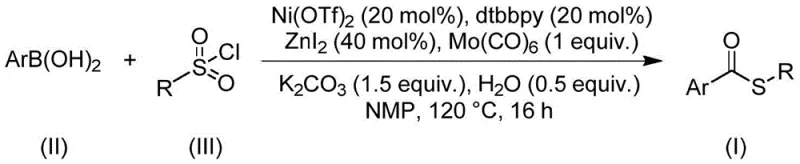
The present analysis examines Chinese Patent CN112239384B, which introduces a groundbreaking nickel-catalyzed carbonylation methodology for synthesizing structurally diverse thioester compounds. This innovation addresses critical limitations in conventional sulfur-containing intermediate production by employing sulfonyl chloride as a non-toxic sulfur source instead of malodorous thiols that typically cause catalyst deactivation. The process demonstrates exceptional substrate flexibility with tolerance for various functional groups including halogens and alkyl substituents across both arylboronic acid and sulfonyl chloride components. Crucially, the reaction operates under redox-neutral conditions without requiring additional oxidants or reductants, significantly simplifying process design while maintaining high yields across multiple compound classes. This patent represents a substantial advancement in sustainable synthesis routes for pharmaceutical intermediates where precise sulfur incorporation is essential for bioactive molecule construction.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional thioester synthesis predominantly relies on acylation reactions between thiols and carboxylic acid derivatives, presenting significant operational challenges due to the volatile nature and strong odor of thiol compounds which complicate handling and increase safety risks in manufacturing environments. These methods frequently require additional oxidants or reductants to achieve desired transformations, introducing extra processing steps that elevate production costs and generate complex waste streams requiring specialized treatment. Furthermore, the narrow functional group tolerance of conventional approaches often necessitates protective group strategies that reduce overall atom economy and extend synthetic pathways. Catalyst poisoning remains a persistent issue when using transition metal systems with thiol-based reagents, leading to inconsistent yields and requiring frequent catalyst replacement that impacts both process economics and reproducibility at scale. The inherent limitations in substrate scope also restrict the applicability of these methods for synthesizing complex pharmaceutical intermediates with specific structural requirements.
The Novel Approach
This patented methodology overcomes these constraints through a nickel-catalyzed carbonylation reaction utilizing arylboronic acids and sulfonyl chlorides as starting materials under optimized conditions. The system employs nickel triflate with dtbpy ligand and molybdenum carbonyl co-catalyst in NMP solvent at precisely controlled temperatures of 120°C for exactly 16 hours. By replacing problematic thiols with stable sulfonyl chloride reagents, the process eliminates catalyst poisoning issues while maintaining excellent functional group compatibility across diverse substrates including heteroaromatics and substituted phenyl systems. The reaction design operates without additional oxidants or reductants, significantly reducing process complexity and associated costs while generating cleaner reaction profiles that simplify downstream purification. This approach demonstrates superior scalability potential due to its robustness across varying substrate combinations and consistent performance metrics observed in multiple experimental trials documented within the patent.

Mechanistic Insights into Nickel-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of sulfonyl chloride to the nickel(0) species generated in situ from nickel triflate reduction by zinc iodide. This forms a nickel(II) sulfonyl complex that undergoes transmetalation with arylboronic acid through a base-assisted mechanism involving potassium carbonate. Subsequent decarbonylation of molybdenum carbonyl provides carbon monoxide that inserts into the nickel-carbon bond, forming an acyl nickel intermediate. Reductive elimination then releases the thioester product while regenerating the active nickel(0) catalyst. This mechanism operates under mild conditions without requiring external redox agents because the sulfonyl chloride serves dual roles as both sulfur source and internal oxidant. The dtbpy ligand plays a critical role in stabilizing the nickel center throughout the catalytic cycle while preventing unwanted side reactions through steric control of substrate approach geometry.
Impurity control is achieved through precise management of reaction parameters that minimize common side products such as homocoupling byproducts or hydrolysis products. The use of water in controlled stoichiometry (0.5 equivalents) facilitates proton transfer without promoting hydrolysis of sensitive intermediates. The broad functional group tolerance stems from the mild reaction conditions that avoid harsh oxidation states or strongly acidic/basic environments that could degrade sensitive substituents. Post-reaction purification via silica gel filtration followed by column chromatography effectively removes residual catalysts and minor impurities, yielding products with high purity suitable for pharmaceutical applications. This systematic approach ensures consistent quality profiles across different substrate combinations while maintaining excellent reproducibility in both laboratory and pilot-scale settings.

How to Synthesize Thioester Compound Efficiently
This section details the standardized procedure for implementing the patented nickel-catalyzed carbonylation method in industrial settings. The process leverages readily available starting materials and avoids specialized equipment requirements while maintaining high selectivity across diverse substrate combinations. Key operational parameters have been optimized through extensive experimentation to ensure consistent product quality and yield stability at various production scales. The following guidelines provide critical insights for successful implementation while highlighting essential quality control checkpoints that must be monitored throughout the synthesis process. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer from laboratory development to commercial manufacturing environments.
- Combine nickel triflate catalyst (20 mol%), dtbpy ligand (20 mol%), molybdenum carbonyl (1 equiv.), potassium carbonate (1.5 equiv.), zinc iodide (40 mol%), water (0.5 equiv.), arylboronic acid, and sulfonyl chloride in NMP solvent under inert atmosphere.
- Heat the reaction mixture to 120°C and maintain at this temperature for precisely 16 hours to ensure complete conversion without decomposition or side reactions.
- Perform post-treatment by filtration through silica gel followed by column chromatography purification to isolate high-purity thioester product with minimal residual catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route delivers substantial operational benefits that directly address critical pain points in pharmaceutical intermediate procurement and supply chain management. By eliminating reliance on problematic sulfur sources that create handling complexities and safety concerns, the process reduces operational risks while improving overall manufacturing reliability. The use of commercially available starting materials with established supply chains ensures consistent raw material availability without exposure to single-source dependencies that could disrupt production schedules. These advantages translate into more predictable lead times and enhanced supply chain resilience for customers requiring high-purity thioester intermediates for complex API manufacturing processes.
- Cost Reduction in Manufacturing: The elimination of expensive auxiliary reagents such as specialized oxidants or reductants significantly reduces raw material costs while simplifying waste treatment requirements. The use of stable sulfonyl chloride reagents instead of volatile thiols minimizes safety-related expenses including specialized containment systems and personnel protective equipment. Process intensification through fewer unit operations lowers energy consumption and labor costs while maintaining high product yields across diverse substrate combinations.
- Enhanced Supply Chain Reliability: Sourcing flexibility is achieved through multiple suppliers for all key starting materials including arylboronic acids and sulfonyl chlorides which are widely available from established chemical manufacturers globally. The robust nature of the reaction tolerates minor variations in raw material quality without compromising final product specifications, reducing qualification burdens for new suppliers. This reliability ensures consistent delivery schedules even during market fluctuations or regional supply disruptions affecting traditional thiol-based routes.
- Scalability and Environmental Compliance: The process demonstrates excellent linear scalability from laboratory to commercial production without requiring significant parameter adjustments or specialized equipment modifications. Mild reaction conditions eliminate the need for extreme temperature or pressure controls that complicate large-scale implementation while reducing energy consumption per unit output. The simplified waste profile containing primarily inorganic salts rather than complex organic byproducts facilitates straightforward treatment that meets stringent environmental regulations across major pharmaceutical manufacturing regions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology based on extensive experimental data and process validation studies. Each response draws directly from documented results within the patent specification to ensure accuracy and relevance for decision-makers evaluating this technology for commercial adoption. These insights reflect practical considerations encountered during scale-up studies and provide actionable information for procurement teams assessing supply chain implications.
Q: How does this method eliminate catalyst poisoning issues compared to conventional thiol-based approaches?
A: By utilizing sulfonyl chloride as the sulfur source instead of volatile thiols, this process avoids catalyst deactivation through sulfur-induced poisoning while maintaining high functional group tolerance across diverse substrates.
Q: What specific cost advantages does the elimination of additional oxidants/reductants provide in commercial production?
A: The redox-neutral reaction design removes expenses associated with purchasing, handling, and removing auxiliary reagents while simplifying waste stream management and reducing overall process complexity.
Q: How does the substrate design flexibility support scalable production of complex pharmaceutical intermediates?
A: The broad functional group compatibility allows direct synthesis of structurally diverse thioesters without protective group strategies, enabling rapid adaptation to specific API requirements while maintaining consistent purity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
Our company leverages this patented technology to deliver high-purity thioester intermediates with exceptional consistency through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities that ensure compliance with global pharmaceutical standards including ICH Q7 guidelines. Our dedicated technical team provides comprehensive support throughout the scale-up process, implementing robust quality management systems that guarantee batch-to-batch reproducibility while meeting demanding regulatory requirements across international markets.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific production needs by contacting our technical procurement team directly. They will provide detailed COA data demonstrating our product quality profiles along with route feasibility assessments that evaluate potential implementation timelines and resource requirements for your particular application scenarios.
