Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access privileged heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinolin-2(1H) ketone derivatives. These compounds are not merely academic curiosities; they serve as critical pharmacophores in a wide array of bioactive molecules, including MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, as illustrated in the structural diversity shown below. The innovation lies in a palladium-catalyzed aminocarbonylation strategy that leverages benzisoxazole as a unique dual-purpose reagent, acting simultaneously as the nitrogen source and the formyl source. This approach represents a paradigm shift from traditional methodologies, offering a reliable pharmaceutical intermediate supplier with a distinct competitive edge in terms of operational simplicity and raw material accessibility.
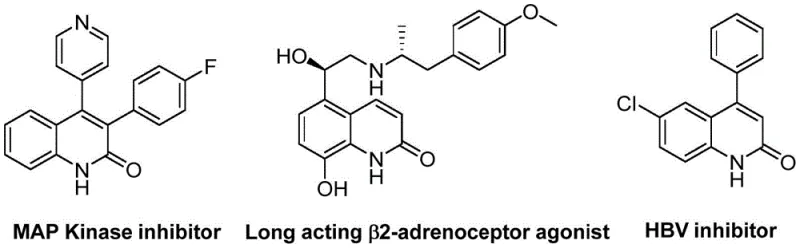
For R&D directors and process chemists, the ability to construct complex heterocycles with high atom economy and minimal waste is paramount. The disclosed technology addresses the longstanding challenges associated with quinolinone synthesis by utilizing readily available benzisoxazoles and benzyl chlorides. By circumventing the need for hazardous gaseous carbon monoxide and employing a solid carbonyl source, the process significantly enhances laboratory safety and simplifies reactor engineering requirements. Furthermore, the reaction demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and cyano groups without compromising yield. This versatility ensures that the method can be adapted for the synthesis of a broad library of analogs, facilitating rapid structure-activity relationship (SAR) studies during drug discovery phases while maintaining a clear path toward cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)-one core has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-documented, they often suffer from significant drawbacks that hinder their applicability in modern, green chemistry-focused manufacturing environments. Traditional routes frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles that are difficult to purge. Moreover, many conventional carbonylation strategies depend on the use of toxic carbon monoxide gas, necessitating specialized high-pressure equipment and rigorous safety protocols that increase capital expenditure and operational complexity. The reliance on multi-step sequences to install the necessary nitrogen and carbonyl functionalities further reduces overall process efficiency, resulting in lower cumulative yields and higher production costs that are unsustainable for large-scale commercial operations.
The Novel Approach
In stark contrast, the novel methodology described in the patent data introduces a streamlined, one-pot synthetic route that elegantly solves these legacy issues. By employing a palladium-catalyzed system with molybdenum hexacarbonyl as a safe, solid surrogate for carbon monoxide, the reaction proceeds under relatively mild conditions at 100°C. The genius of this approach lies in the utilization of benzisoxazole, which serves a dual role: it provides the nitrogen atom for the heterocyclic ring and the carbonyl carbon through an innovative ring-opening and insertion mechanism. This eliminates the need for pre-functionalized precursors and reduces the step count, thereby enhancing the overall mass balance of the process. The use of simple benzyl chlorides as coupling partners further underscores the practicality of this method, as these reagents are commodity chemicals available in vast quantities. This strategic redesign of the synthetic pathway not only improves safety and environmental compliance but also drastically simplifies the supply chain logistics for key starting materials.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The success of this transformation hinges on a sophisticated catalytic cycle involving a palladium(0) species generated in situ from palladium acetate and the chiral ligand (S)-BINAP. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium catalyst, forming a benzyl-palladium(II) intermediate. Subsequently, the benzisoxazole undergoes coordination and insertion, facilitated by the presence of molybdenum hexacarbonyl which releases carbon monoxide under the reaction conditions. This step is critical as it effectively transfers the carbonyl moiety into the growing molecular framework. The presence of triethylamine as a base and a stoichiometric amount of water plays a vital role in promoting the ring-opening of the benzisoxazole and the subsequent cyclization to form the quinolinone core. The careful optimization of the ligand-to-metal ratio and the choice of solvent, specifically ethylene glycol dimethyl ether (DME), ensures high turnover numbers and minimizes catalyst deactivation. This mechanistic understanding allows for precise control over the reaction parameters, ensuring consistent quality and reproducibility essential for GMP manufacturing environments.
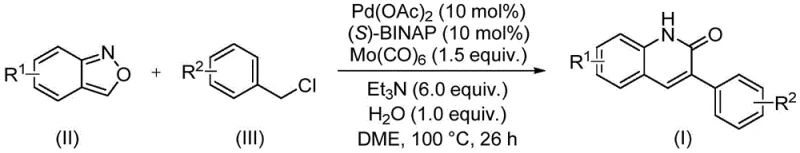
From an impurity control perspective, the robustness of this catalytic system is evident in its high tolerance for diverse electronic and steric environments on the aromatic rings. The patent data indicates that substituents such as electron-withdrawing cyano groups, electron-donating methoxy groups, and bulky tert-butyl groups are all well-tolerated, yielding the desired products with excellent purity profiles. The reaction conditions are sufficiently mild to prevent side reactions such as homocoupling of the benzyl chloride or decomposition of the sensitive benzisoxazole ring prior to the desired transformation. Furthermore, the post-processing steps involve simple filtration followed by silica gel chromatography, which effectively removes palladium residues and other metal contaminants. This is a crucial consideration for pharmaceutical applications where residual metal levels are strictly regulated. The ability to achieve high yields, often exceeding 90% for optimized substrates, combined with a clean impurity profile, makes this method superior to older technologies that often require extensive recrystallization or preparative HPLC to meet purity specifications.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The implementation of this synthesis protocol is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves charging a sealed tube or reactor with the palladium catalyst, ligand, carbonyl source, base, water, and the two primary organic substrates in a suitable solvent. The mixture is then heated to a specific temperature range, typically around 100°C, and maintained for a defined period to drive the reaction to completion. The simplicity of the reagent list and the absence of sensitive air-free techniques (beyond standard inert atmosphere handling for the catalyst) make this process highly accessible. For detailed operational parameters, stoichiometry, and specific workup instructions tailored to your specific substrate, please refer to the standardized synthesis guide provided below.
- Combine palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the use of commodity-grade starting materials. Benzisoxazoles and benzyl chlorides are produced on a massive industrial scale for various applications, ensuring a stable and competitive supply market. Unlike specialized reagents that may have single-source suppliers and long lead times, these inputs can be sourced from multiple vendors globally, mitigating the risk of supply disruptions. Additionally, the replacement of gaseous carbon monoxide with solid molybdenum hexacarbonyl eliminates the need for complex gas handling infrastructure and safety certifications, significantly reducing the barrier to entry for manufacturing this intermediate. This simplification of the process hardware translates directly into lower capital investment and reduced maintenance costs for the production facility.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom efficiency and the elimination of expensive protecting group strategies often required in traditional syntheses. By combining the nitrogen and carbonyl sources into a single, inexpensive reagent (benzisoxazole), the material cost per kilogram of the final product is significantly lowered. Furthermore, the high yields reported in the patent data mean that less raw material is wasted, and the throughput of the reactor is maximized. The simplified workup procedure, which avoids complex extractions or distillations in favor of filtration and chromatography, reduces solvent consumption and energy usage. These factors collectively contribute to a substantial reduction in the cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the stability of the reagents. The reaction tolerates a wide range of functional groups, meaning that variations in the quality of starting materials (within specification) are less likely to cause batch failures. This robustness reduces the frequency of failed batches and the associated costs of rework or disposal. Moreover, since the key reagents are stable solids or liquids with long shelf lives, inventory management becomes more predictable and less prone to spoilage. The ability to source materials locally in major chemical hubs further shortens the logistics chain, reducing lead times and transportation costs. This reliability is critical for maintaining just-in-time manufacturing schedules and meeting the demanding delivery timelines of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the homogeneous nature of the reaction and the absence of hazardous gases. The use of DME as a solvent, while requiring appropriate recovery systems, is manageable within standard solvent recovery units. The process generates minimal hazardous waste compared to traditional methods that might produce stoichiometric amounts of salt waste or require chlorinated solvents. The high efficiency of the catalyst system means that palladium loading can be kept low, reducing the burden on wastewater treatment facilities for heavy metal removal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of adopting this technology for your specific production needs.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method utilizes benzisoxazole as a dual nitrogen and formyl source, eliminating the need for toxic carbon monoxide gas and simplifying the reaction setup compared to traditional Vilsmeier-Haack or Friedlander reactions.
Q: What catalyst system is employed for this transformation?
A: The process employs a robust catalytic system consisting of palladium acetate (Pd(OAc)2), (S)-BINAP as the chiral ligand, and molybdenum hexacarbonyl (Mo(CO)6) as the solid carbonyl source.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the protocol uses commercially available and inexpensive starting materials, operates at moderate temperatures (100°C), and involves a straightforward workup procedure, making it highly amenable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
The technological advancements presented in patent CN113045489B highlight the immense potential of 3-arylquinolin-2(1H)-one derivatives as versatile building blocks for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methods into robust, commercial-scale manufacturing processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our existing catalog or to discuss route feasibility assessments for your custom synthesis projects. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by a commitment to innovation, safety, and operational excellence.
