Scalable Synthesis of 3-Arylquinolin-2(1H)-one Derivatives via Palladium-Catalyzed Aminocarbonylation
Scalable Synthesis of 3-Arylquinolin-2(1H)-one Derivatives via Palladium-Catalyzed Aminocarbonylation
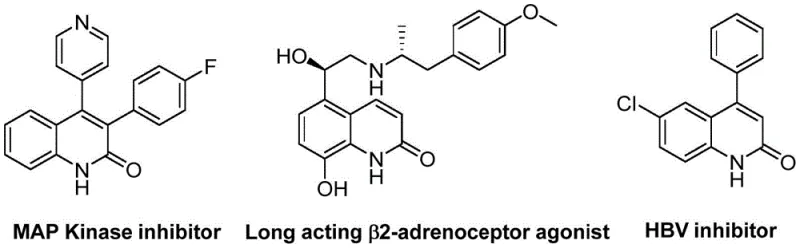
The quinolin-2(1H)-one scaffold represents a privileged structure in medicinal chemistry, serving as the core backbone for a diverse array of biologically active compounds including antibiotics, antiplatelet agents, and antitumor drugs. As depicted in the structural examples below, these derivatives are critical intermediates for developing MAP Kinase inhibitors and HBV inhibitors, highlighting their immense value in the pharmaceutical pipeline. A recent technological breakthrough detailed in patent CN113045489B introduces a highly efficient preparation method for these valuable heterocycles. This novel approach leverages a palladium-catalyzed aminocarbonylation strategy that utilizes benzisoxazole as a unique dual-purpose reagent, acting simultaneously as the nitrogen source and the formyl donor. This innovation addresses long-standing challenges in heterocyclic synthesis by offering a pathway that is not only operationally simple but also exhibits exceptional functional group tolerance, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking robust synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolinone core has relied on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these traditional protocols are well-established in academic literature, they often suffer from significant drawbacks when translated to industrial manufacturing environments. Many of these legacy processes require harsh reaction conditions, including the use of strong acids or bases, elevated temperatures, and toxic reagents that pose severe safety and environmental hazards. Furthermore, transition metal-catalyzed improvements to these methods frequently necessitate the use of high-pressure carbon monoxide gas, which demands specialized autoclave equipment and rigorous safety protocols to prevent leakage and exposure. These factors collectively contribute to increased capital expenditure and operational complexity, creating bottlenecks in cost reduction in pharmaceutical intermediate manufacturing where efficiency and safety are paramount concerns for procurement teams.
The Novel Approach
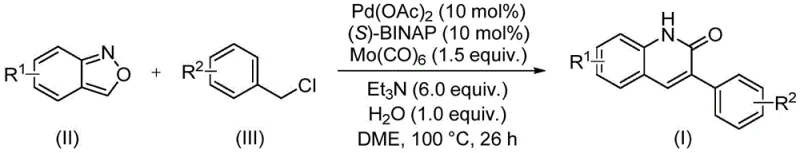
In stark contrast to these conventional limitations, the methodology disclosed in the present invention offers a transformative solution by employing a solid carbonyl source, molybdenum hexacarbonyl (Mo(CO)6), in conjunction with a palladium catalyst system. This strategic substitution eliminates the need for handling gaseous carbon monoxide, thereby drastically simplifying the reactor requirements and enhancing workplace safety. The reaction proceeds under relatively mild thermal conditions at 100°C in dimethoxyethane (DME) solvent, utilizing triethylamine as a base. By integrating benzisoxazole as the key building block, the process achieves a remarkable atom economy and step efficiency. This new route not only streamlines the synthetic workflow but also expands the scope of accessible derivatives, allowing for the incorporation of diverse substituents such as halogens, alkoxy groups, and cyano groups without compromising yield, thus providing a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this synthetic advancement lies in the sophisticated palladium-catalyzed cycle that orchestrates the coupling of benzisoxazole and benzyl chloride. The reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species generated in situ from palladium acetate and the chiral ligand (S)-BINAP. Following this activation, the molybdenum hexacarbonyl serves as a controlled reservoir for carbon monoxide, releasing CO into the coordination sphere of the palladium center to facilitate migratory insertion. This step is critical for forming the acyl-palladium intermediate, which subsequently undergoes nucleophilic attack by the nitrogen atom derived from the ring-opening of the benzisoxazole moiety. The final reductive elimination step releases the 3-arylquinolin-2(1H)-one product and regenerates the catalytic species, completing the cycle. This mechanistic pathway ensures high regioselectivity and minimizes the formation of side products, which is essential for maintaining a clean impurity profile in high-purity pharmaceutical intermediate production.
From an impurity control perspective, the use of benzisoxazole provides a distinct advantage by locking the nitrogen and carbonyl sources into a single molecular entity, reducing the statistical probability of mismatched coupling events that often plague multi-component reactions. The presence of water in the reaction mixture, as specified in the protocol, plays a subtle yet vital role in facilitating the hydrolysis or proton transfer steps necessary for the aromatization of the quinolinone ring. The robustness of the (S)-BINAP ligand system further ensures that the catalyst remains stable throughout the extended reaction time of 26 hours, preventing premature deactivation that could lead to incomplete conversion and difficult-to-remove starting material residues. This level of mechanistic control translates directly into downstream processing benefits, as the crude reaction mixtures are cleaner and require less aggressive purification measures, aligning perfectly with the stringent quality standards expected by R&D directors overseeing process development.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The procedure involves charging a sealed reaction vessel with the requisite amounts of palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative. The mixture is then heated to 100°C for 26 hours to drive the reaction to completion. Post-reaction processing is straightforward, involving filtration to remove insoluble salts followed by silica gel treatment and column chromatography. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup parameters optimized for different substrates, please refer to the standardized guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical yield. The primary driver for cost optimization is the utilization of benzisoxazole and benzyl chlorides, which are commodity chemicals available in bulk quantities from multiple global suppliers, ensuring a stable and competitive pricing structure. By avoiding the use of high-pressure carbon monoxide gas, manufacturers can utilize standard glass-lined or stainless steel reactors without the need for expensive pressure-rated autoclaves, resulting in significant capital expenditure savings and reduced maintenance costs. Furthermore, the elimination of hazardous gas handling simplifies regulatory compliance and lowers insurance premiums associated with chemical manufacturing operations.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive or hazardous reagents with inexpensive, shelf-stable solids like Mo(CO)6 and benzisoxazole. The high reaction efficiency, with yields reaching up to 97% for certain substrates, minimizes raw material waste and maximizes output per batch. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, further lowering the variable costs associated with production. This economic efficiency makes the method highly attractive for cost reduction in API manufacturing where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions that often occur with specialized or custom-synthesized reagents. Benzyl chlorides and substituted benzisoxazoles are produced on a massive scale for various industries, ensuring consistent availability and short lead times. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in raw material quality, providing a buffer against supply chain variability. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to meet tight delivery schedules without compromising on quality.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure (in a sealed tube context, but effectively low pressure compared to CO gas methods) and moderate temperature, making it inherently safer and easier to scale from kilogram to tonne levels. The absence of toxic gas emissions aligns with increasingly strict environmental regulations, reducing the burden on waste treatment facilities. The use of DME as a solvent, while requiring recovery, is manageable within standard solvent recycling loops. This environmental compatibility facilitates smoother regulatory approvals and supports sustainable manufacturing practices, a key priority for modern supply chain strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility, catalyst loading, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for external carbon monoxide gas and simplifying the reaction setup significantly.
Q: What is the typical yield range for this palladium-catalyzed method?
A: According to the patent data, the method demonstrates high reaction efficiency with isolated yields ranging from 68% to 97% across various substrates with different electronic properties.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and operates at moderate temperatures (100°C), making it highly amenable to commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
As the demand for complex heterocyclic intermediates continues to grow, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)-one derivatives meets the highest industry standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering consistent results.
We invite you to engage with our technical procurement team to discuss your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how this novel synthetic route can optimize your budget. We encourage potential partners to contact us directly to obtain specific COA data for related compounds and comprehensive route feasibility assessments tailored to your project's unique needs, ensuring a collaborative path forward in bringing your therapeutic candidates to market.
