Advanced Manufacturing of High-Purity 3-Acylpyrrole Intermediates: Scalable Synthesis for Pharmaceutical Innovation
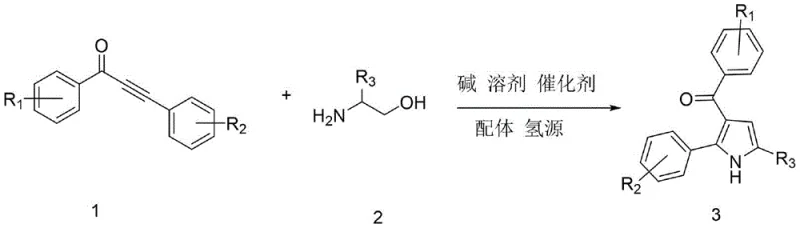
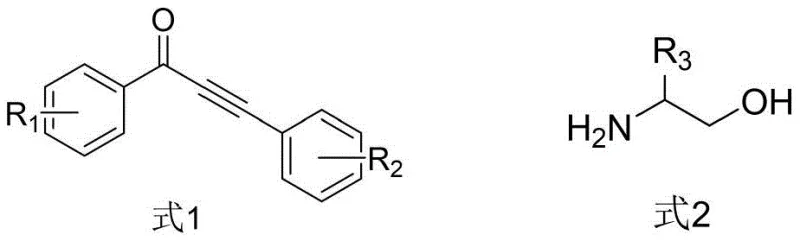
The recently granted Chinese patent CN113636968B introduces a transformative methodology for synthesizing pharmaceutical-grade 3-acylpyrrole intermediates through a ruthenium-catalyzed alcohol-hydrogen transfer coupling reaction. This innovation addresses critical challenges in heterocyclic compound production by establishing a streamlined pathway that achieves exceptional atom economy while maintaining compatibility with complex functional groups essential for advanced drug development. The patented process represents a significant advancement over conventional synthetic approaches by eliminating multi-step purification requirements and hazardous reagents that have historically constrained industrial-scale manufacturing of these pharmacologically vital building blocks. By leveraging commercially available starting materials and standard laboratory equipment, this methodology offers immediate scalability from laboratory validation to commercial production without requiring specialized infrastructure investments. The technical elegance of this approach lies in its ability to simultaneously address purity requirements for pharmaceutical applications while optimizing resource utilization across the entire manufacturing value chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing multi-substituted pyrrole compounds have been severely constrained by multiple fundamental limitations that impede industrial implementation. Existing methodologies often require toxic reagents such as IBX oxidants or expensive palladium catalysts that necessitate complex removal procedures and generate hazardous waste streams incompatible with modern environmental regulations. The multi-step nature of conventional approaches frequently involves unstable intermediates requiring cryogenic conditions or anhydrous environments that significantly increase operational complexity and failure risk during scale-up. Furthermore, the reliance on rare or difficult-to-source starting materials creates supply chain vulnerabilities that compromise production continuity, while the use of precious metal catalysts introduces substantial cost burdens that directly impact final product pricing. These combined factors have historically prevented the economical manufacturing of high-purity 3-acylpyrrole derivatives despite their well-documented therapeutic potential in critical drug classes including histone deacetylase inhibitors and HMG-CoA reductase modulators.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an elegant one-pot reaction system that utilizes triruthenium dodecacarbonyl catalyst with 4-methyl-1,10 phenanthroline ligand to facilitate direct coupling between α-amino alcohols and α,β-unsaturated alkynones under mild thermal conditions. This innovative process operates efficiently within standard laboratory equipment at temperatures ranging from 80°C to 150°C without requiring specialized pressure vessels or cryogenic systems, thereby dramatically simplifying operational requirements while maintaining exceptional selectivity. The strategic use of ethanol as both solvent and hydrogen source eliminates the need for external reducing agents while enabling straightforward product isolation through conventional chromatographic techniques. Crucially, the reaction demonstrates remarkable functional group tolerance across diverse substituent patterns, allowing pharmaceutical manufacturers to access structurally complex intermediates without extensive protection/deprotection sequences that previously compromised process efficiency and product yield.
Mechanistic Insights into Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Compounds
The catalytic cycle begins with oxidative addition of the ruthenium complex to the alkyne moiety of the α,β-unsaturated alkynone substrate, forming a key vinyl-ruthenium intermediate that facilitates nucleophilic attack by the α-amino alcohol component. This critical step establishes the carbon-nitrogen bond framework through a concerted hydrogen transfer mechanism where ethanol serves as both proton donor and hydride source, enabling simultaneous reduction and cyclization without external reductants. The subsequent intramolecular cyclization proceeds through a six-membered transition state that directs regioselective formation of the pyrrole ring system while minimizing competing side reactions. The catalyst's unique ligand environment provided by the methyl-substituted phenanthroline promotes efficient turnover by stabilizing reactive intermediates and preventing catalyst deactivation through dimerization pathways that commonly plague other transition metal systems.
Impurity profile management is achieved through precise control of reaction parameters that suppress common side products such as dimerized alkynes or over-reduced byproducts. The mild basic conditions maintained by potassium carbonate prevent acid-catalyzed decomposition pathways while facilitating proton transfer steps essential for ring closure. The solvent system's polarity profile selectively solubilizes desired intermediates while precipitating potential impurities during the reaction course, creating a self-purifying effect that significantly reduces downstream processing requirements. This inherent selectivity is further enhanced by the catalyst's ability to discriminate between competing reaction pathways based on steric and electronic factors within the substrate molecules, resulting in consistently high purity profiles across diverse structural variants without requiring additional purification steps beyond standard chromatography.
How to Synthesize High-Purity 3-Acylpyrrole Compounds Efficiently
This patented methodology represents a significant advancement in the manufacturing of complex heterocyclic intermediates through its innovative approach to catalytic coupling chemistry. The process demonstrates exceptional versatility across diverse substrate combinations while maintaining consistent product quality essential for pharmaceutical applications. Detailed operational parameters have been optimized to ensure reproducibility at commercial scale without requiring specialized equipment modifications. The following standardized synthesis protocol provides step-by-step guidance for implementing this technology within existing manufacturing infrastructure while achieving the stringent quality requirements demanded by regulatory authorities.
- Combine α-amino alcohol and α,β-unsaturated alkynone with triruthenium dodecacarbonyl catalyst, 4-methyl-1,10 phenanthroline ligand, ethanol hydrogen source, potassium carbonate base, and tert-amyl alcohol solvent in a Schlenk reactor under nitrogen atmosphere at 80°C for 1.5 hours
- Gradually increase reaction temperature to 150°C and maintain for 18 hours to complete the alcohol-hydrogen transfer coupling process while ensuring optimal catalyst efficiency
- Cool reaction mixture to room temperature, dilute with ethyl acetate, filter to remove solids, perform reduced-pressure distillation, and purify crude product using column chromatography with petroleum ether/ethyl acetate (5-20: 1) eluent
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing fundamental pain points in intermediate sourcing through scientifically validated process improvements. The elimination of multiple processing steps reduces vulnerability to supply chain disruptions while creating significant opportunities for cost optimization without compromising product quality or regulatory compliance requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized purification equipment creates substantial cost savings through reduced raw material expenditures and lower capital investment requirements. The one-pot methodology significantly decreases solvent consumption and waste generation compared to conventional multi-step processes, resulting in lower operational costs associated with solvent procurement, waste disposal, and environmental compliance management. The use of standard laboratory equipment enables seamless integration into existing manufacturing facilities without requiring costly infrastructure modifications.
- Enhanced Supply Chain Reliability: The reliance on globally available starting materials with established supply networks minimizes sourcing risks while enabling flexible production scheduling across multiple manufacturing sites. The process's robustness against minor variations in raw material quality ensures consistent output even when dealing with different supplier batches, reducing quality-related production delays. The elimination of air-sensitive reagents and cryogenic requirements creates greater operational flexibility during transportation and storage while maintaining product integrity throughout the supply chain.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory validation to commercial production volumes without requiring fundamental process modifications, enabling rapid technology transfer between development and manufacturing sites. The significant reduction in hazardous waste streams through atom-economical design simplifies environmental compliance management while aligning with global sustainability initiatives. The elimination of toxic reagents creates safer working conditions while reducing regulatory burden associated with handling hazardous materials during production operations.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics observed during extensive validation studies.
Q: How does this novel synthesis overcome the limitations of conventional methods for 3-acylpyrrole production?
A: The ruthenium-catalyzed one-pot methodology eliminates multiple purification steps required in traditional approaches while avoiding toxic reagents and precious metal catalysts. By utilizing readily available α-amino alcohols and alkynones with ethanol as hydrogen source, it achieves superior atom economy without hazardous intermediates that plagued previous synthetic routes.
Q: What are the key advantages of the ruthenium-catalyzed process in terms of supply chain reliability?
A: The process utilizes globally accessible raw materials with simplified logistics requirements. The elimination of air-sensitive reagents and cryogenic conditions enables consistent production across diverse manufacturing environments while maintaining stringent quality control standards throughout the supply chain.
Q: How does the one-pot methodology contribute to cost reduction in pharmaceutical intermediate manufacturing?
A: By consolidating multiple reaction steps into a single operation with high catalyst turnover, the process significantly reduces solvent consumption, energy requirements, and waste generation. The elimination of expensive transition metal catalysts and specialized equipment lowers both capital expenditure and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Compound Supplier
Our proprietary implementation of this patented technology demonstrates exceptional potential for producing high-purity pharmaceutical intermediates with unparalleled consistency across diverse structural variants. As a CDMO specialist with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we maintain stringent purity specifications through advanced analytical capabilities and rigorous QC labs that ensure compliance with global regulatory standards. Our manufacturing platform integrates this innovative methodology with comprehensive quality systems that provide complete traceability from raw material sourcing through final product release.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team directly. They will provide detailed COA data and route feasibility assessments demonstrating how this technology can optimize your supply chain while meeting your exact quality specifications for pharmaceutical intermediate manufacturing.
