Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Scale Production
Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Scale Production
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex nitrogen-containing heterocyclic scaffolds, which serve as critical cores for bioactive molecules. Patent CN109776546B introduces a transformative approach for the preparation of indolopyrrolone compounds, a class of structures widely recognized for their presence in natural products and their potential as active pharmaceutical ingredients. This technology leverages a sophisticated catalytic system involving a rhenium complex, a Lewis acid, and a zinc reagent to facilitate a direct C-H bond activation and C-N bond cleavage reaction. By enabling the coupling of indole derivatives with various alkynes, this method addresses long-standing challenges in heterocyclic synthesis, offering a pathway to high-purity indolopyrrolone compounds that is both chemically elegant and industrially viable for reliable indolopyrrolone supplier networks seeking to expand their portfolio.
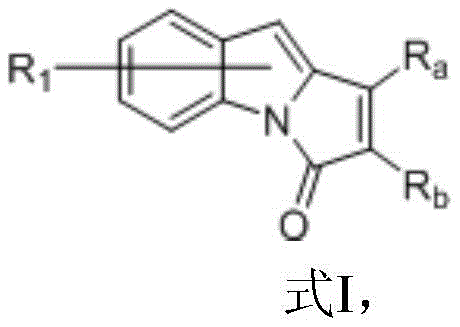
Indolopyrrolone compounds represent a privileged structural motif in medicinal chemistry, yet their efficient construction has historically been hindered by restrictive reaction conditions and poor substrate tolerance. The traditional reliance on precious metal catalysts often resulted in inconsistent outcomes, particularly when attempting to introduce diverse functional groups onto the pyrrolone ring. The methodology disclosed in CN109776546B overcomes these barriers by utilizing a decacarbonyldirhenium catalyst in conjunction with zinc chloride and dimethylzinc. This specific combination allows for the transformation of N,N-diphenyl-1H-indole-1-carboxamides and internal alkynes into the desired fused ring system with remarkable efficiency. The ability to operate with such broad substituent compatibility means that manufacturers can access a wider chemical space without needing to develop entirely new synthetic routes for each derivative, significantly enhancing the strategic value of this technology for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of indolopyrrolone frameworks was largely dependent on transition metal catalysts that exhibited significant limitations in scope and efficiency. For instance, literature precedents such as those published in J. Org. Chem. (2017) utilized ruthenium catalysts which, while effective for simple methyl-substituted alkynes, struggled considerably when aryl groups were introduced, resulting in yields often below 79 percent. Similarly, cobalt-catalyzed systems described in J. Am. Chem. Soc. (2014) faced analogous challenges, with yields dropping below 72 percent for aryl-substituted substrates, rendering them economically unviable for large-scale production of diverse analogues. Furthermore, alternative approaches involving sulfamides, as noted in Chem. Lett. (2015), demonstrated even poorer performance with yields as low as 39 percent for indole-containing reactions. These inefficiencies create substantial bottlenecks in the supply chain, necessitating excessive starting materials and generating significant chemical waste, which complicates the commercial scale-up of complex heterocyclic intermediates.
The Novel Approach
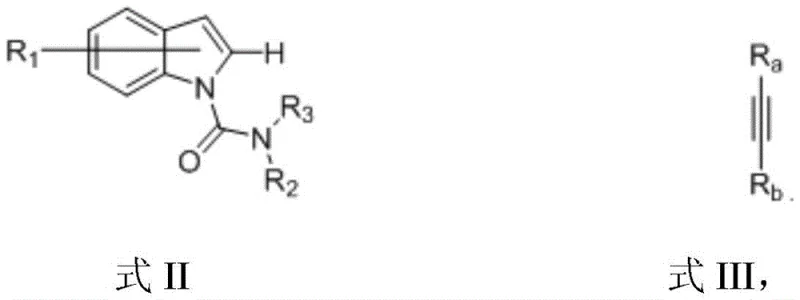
In stark contrast to these legacy methods, the novel approach detailed in the patent employs a rhenium-based catalytic system that demonstrates exceptional versatility and high conversion rates across a broad spectrum of substrates. By reacting Formula II indole compounds with Formula III alkynes in the presence of Re2(CO)10, ZnCl2, and ZnMe2 in toluene at 150°C, the process achieves yields ranging from 76 percent to an impressive 98 percent, even with sterically demanding or electronically diverse substituents. This method successfully accommodates alkyl, aryl, alkoxy, and halogen groups on both the indole and alkyne components, as evidenced by the successful synthesis of 7-methyl, 7-methoxy, and 7-chloro derivatives with high fidelity. The robustness of this reaction profile ensures that reducing lead time for high-purity indolopyrrolone compounds becomes a tangible reality, as fewer optimization cycles are required to adapt the process to new target molecules within the same chemical class.
Mechanistic Insights into Re-Catalyzed C-H Activation and C-N Cleavage
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the rhenium catalyst, which orchestrates a tandem sequence of C-H bond activation and C-N bond cleavage. The decacarbonyldirhenium complex acts as the primary driver for activating the inert C-H bonds on the indole ring, a step that is traditionally difficult to achieve without harsh conditions or directing groups that are hard to remove. Simultaneously, the presence of the zinc chloride Lewis acid and the organozinc reagent creates a cooperative environment that promotes the cleavage of the amide C-N bond, allowing for the cyclization that forms the pyrrolone ring. This dual-activation mechanism is critical because it bypasses the need for pre-functionalized substrates, thereby simplifying the overall synthetic tree. The reaction proceeds through a coordinated cycle where the metal center stabilizes the transition states, ensuring that the energy barrier for ring closure is sufficiently lowered to allow the reaction to proceed efficiently at 150°C over a period of 10 to 100 hours.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that typically plague radical-based or less selective catalytic systems. The specific coordination geometry of the rhenium complex, combined with the stoichiometric control provided by the zinc reagents (typically used at 30 mol percent relative to the indole), directs the reaction pathway strongly towards the desired fused ring product. Experimental data from the patent examples shows that even with sensitive functional groups like bromine or methoxy present on the aromatic rings, the reaction maintains high selectivity, avoiding dehalogenation or ether cleavage. This high level of chemoselectivity is paramount for R&D directors focused on purity profiles, as it reduces the burden on downstream purification processes such as column chromatography or recrystallization, ultimately leading to a cleaner crude product and higher overall recovery of the active pharmaceutical ingredient intermediate.
How to Synthesize Indolopyrrolone Compounds Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric balance of the catalytic system and the maintenance of an inert atmosphere to prevent oxidation of the sensitive organozinc species. The general protocol involves charging a reaction vessel with the indole substrate and the alkyne coupling partner, followed by the addition of the rhenium catalyst, zinc chloride, and dimethylzinc in anhydrous toluene. The mixture is then heated to reflux conditions, typically around 150°C, and monitored until the starting materials are fully consumed, which usually occurs within 24 to 48 hours for most aryl-substituted alkynes. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the indole compound (Formula II), alkyne (Formula III), decacarbonyldirhenium catalyst, zinc chloride Lewis acid, and dimethylzinc reagent in anhydrous toluene under nitrogen protection.
- Heat the reaction mixture to a temperature range of 50°C to 150°C, specifically maintaining 150°C for optimal conversion, and stir for a duration of 10 to 100 hours depending on substrate reactivity.
- Upon completion, dilute the reaction with dichloromethane, filter through silica gel, wash with ethyl acetate, concentrate the filtrate, and purify the crude product via column chromatography to obtain the target indolopyrrolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhenium-catalyzed methodology presents a compelling value proposition centered on efficiency, scalability, and risk mitigation. The primary advantage stems from the drastic improvement in reaction yields compared to prior art; achieving yields near quantitative levels (e.g., 98 percent for methyl-substituted derivatives) means that less raw material is required to produce the same amount of product, directly translating to substantial cost savings in raw material procurement. Furthermore, the broad substrate scope implies that a single manufacturing platform can be used to produce a wide library of analogues, reducing the need for specialized equipment or distinct process validation for each new derivative. This flexibility enhances supply chain reliability by allowing manufacturers to pivot quickly between different product SKUs based on market demand without incurring significant changeover costs or delays.
- Cost Reduction in Manufacturing: The elimination of inefficient catalytic systems that require excessive loading or produce significant byproducts leads to a streamlined production process with lower waste disposal costs. By utilizing a catalyst system that operates effectively at moderate loadings (1-30 mol percent) and achieves high conversion, the overall cost of goods sold is optimized through better atom economy and reduced solvent usage per kilogram of product. Additionally, the use of toluene as a solvent, which is a commodity chemical with established recovery protocols, further supports cost-effective operations compared to more exotic or difficult-to-recycle solvent systems often found in academic precedents.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. Since the method tolerates a wide variety of commercially available starting materials, including substituted indoles and diarylalkynes, the risk of supply disruption due to the unavailability of a specific niche precursor is minimized. This diversity in sourcing options allows procurement teams to negotiate better terms with multiple vendors for raw materials, thereby securing the continuity of supply for high-purity indolopyrrolone compounds even in volatile market conditions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard heating and stirring equipment capable of handling temperatures up to 150°C without requiring high-pressure reactors or cryogenic conditions. This simplicity facilitates easier technology transfer from laboratory to pilot and commercial scales, reducing the time to market for new intermediates. Moreover, the high selectivity of the reaction reduces the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations and supporting the company's sustainability goals by lowering the E-factor of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and scope definitions provided in the patent documentation, ensuring accuracy for technical decision-makers evaluating this technology for integration into their existing manufacturing workflows.
Q: What are the limitations of conventional ruthenium or cobalt catalysts for indolopyrrolone synthesis?
A: Conventional methods utilizing ruthenium or cobalt catalysts often suffer from limited substrate scope, particularly showing low yields (less than 79% for Ru and 72% for Co) when aryl substituents are present on the alkyne, whereas the novel rhenium method accommodates diverse groups effectively.
Q: What represents the key innovation in patent CN109776546B regarding reaction conditions?
A: The key innovation lies in the synergistic use of a rhenium complex containing CO ligands, a zinc-based Lewis acid, and an organozinc reagent, which enables efficient C-H activation and C-N bond cleavage under relatively standard thermal conditions in toluene.
Q: How does this method improve supply chain reliability for pharmaceutical intermediates?
A: By achieving significantly higher yields (up to 98%) and tolerating a wide range of functional groups including halogens and alkoxy groups, this method reduces the need for multiple synthetic routes, thereby streamlining procurement and ensuring consistent supply of high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolopyrrolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the Re-catalyzed C-H activation route and is fully prepared to leverage this methodology for the custom synthesis of complex indolopyrrolone derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require gram-scale samples for drug discovery or metric ton quantities for clinical trials, our facilities can deliver with precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch produced.
We invite you to collaborate with us to unlock the full potential of this efficient synthetic route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates how switching to this high-yield methodology can impact your overall budget and timeline. We encourage you to contact us today to discuss your requirements,索取 specific COA data for related intermediates, and receive comprehensive route feasibility assessments tailored to your target molecules. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and delivering the high-quality intermediates your business demands.
