Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Pharmaceutical Manufacturing
Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Pharmaceutical Manufacturing
The landscape of nitrogen-containing heterocyclic synthesis is undergoing a significant transformation driven by the need for more efficient and versatile methodologies in drug discovery. Patent CN109776546B introduces a groundbreaking approach for the preparation of indolopyrrolone compounds, a class of molecules renowned for their diverse biological activities and presence in numerous natural products. This technology leverages a sophisticated C-H bond activation and C-N bond cleavage strategy, utilizing a rhenium-based catalytic system to achieve superior yields compared to traditional methods. For research and development teams seeking reliable indolopyrrolone supplier partnerships, this patent represents a pivotal advancement in accessing complex scaffolds with high purity and structural diversity. The ability to synthesize these cores efficiently opens new avenues for developing active pharmaceutical ingredients and exploring novel chemical space.
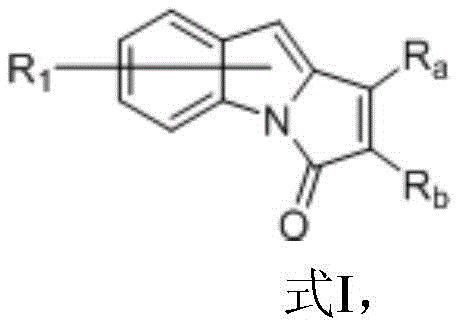
Indolopyrrolone compounds serve as critical structural units in medicinal chemistry, yet their synthesis has historically been plagued by limitations in substrate scope and reaction efficiency. The conventional reliance on precious metal catalysts often resulted in moderate yields, particularly when dealing with sterically demanding or electronically diverse substituents. This new methodology addresses these pain points by introducing a robust catalytic cycle that tolerates a wide array of functional groups, including alkyl, aryl, alkoxy, and halogen substituents. By enabling the direct transformation of readily available indole derivatives and alkynes into fused pyrrolidone systems, this process significantly streamlines the synthetic route. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, the high atom economy and reduced waste generation associated with this protocol offer compelling economic advantages over legacy technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for constructing the indolopyrrolone scaffold have predominantly relied on ruthenium or cobalt catalysts, which exhibit significant drawbacks in terms of versatility and yield. For instance, literature precedents such as those published in J. Org. Chem. (2017) and J. Am. Chem. Soc. (2014) demonstrate that while ruthenium and cobalt systems can facilitate the coupling of indoles with alkynes, they suffer from poor performance when the alkyne substrate bears aryl substituents other than methyl. Yields in these traditional processes often struggle to exceed 79%, and in some cases involving sulfamides, drop as low as 39%. Furthermore, these older methods frequently require harsh conditions or specialized ligands that complicate the purification process and increase the overall cost of goods. The limited functional group tolerance restricts the chemical diversity accessible to medicinal chemists, forcing them to rely on longer, more convoluted synthetic sequences to introduce necessary modifications.
The Novel Approach
In stark contrast, the method disclosed in CN109776546B employs a decacarbonyldirhenium catalyst combined with a Lewis acid and an organozinc reagent to drive the reaction with exceptional efficiency. This novel system allows for the successful coupling of indoles with a broad spectrum of alkynes, including those with bulky aryl, halogenated aryl, and alkyl groups, achieving isolated yields as high as 98% in optimized examples. The reaction proceeds through a unique C-H activation and C-N bond cleavage mechanism that is far more forgiving of steric hindrance and electronic variations than its predecessors. By utilizing common solvents like toluene and operating at manageable temperatures between 50 to 150°C, this approach simplifies the operational complexity typically associated with heterocycle synthesis. This technological leap ensures that high-purity indolopyrrolone intermediates can be produced consistently, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Re2(CO)10-Catalyzed C-H Activation
The core of this synthetic breakthrough lies in the intricate interplay between the rhenium catalyst, the Lewis acid, and the zinc reagent. The decacarbonyldirhenium complex acts as the primary driver for C-H bond activation on the indole ring, initiating the cyclization process. Simultaneously, the presence of a Lewis acid, preferably zinc chloride, coordinates with the carbonyl oxygen of the indole amide precursor, increasing the electrophilicity of the carbonyl carbon and facilitating the subsequent nucleophilic attack. The organozinc reagent, such as dimethylzinc, plays a crucial role in regenerating the active catalytic species and assisting in the cleavage of the C-N bond, which is the rate-determining step in many similar transformations. This synergistic effect creates a highly active catalytic cycle that minimizes side reactions and maximizes the formation of the desired fused ring system. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters for specific substrate classes.
Furthermore, the impurity profile of the resulting indolopyrrolones is significantly improved due to the high selectivity of this catalytic system. Traditional methods often generate substantial amounts of regioisomers or polymerization byproducts from the alkyne starting material, necessitating rigorous and yield-lowering purification steps. In this rhenium-catalyzed protocol, the specific coordination environment around the metal center directs the reaction pathway exclusively towards the desired 5-exo-dig or 6-endo-dig cyclization modes, depending on the substrate geometry. The use of zinc chloride also helps to suppress the formation of homocoupling byproducts of the alkyne. Consequently, the crude reaction mixtures are cleaner, allowing for simpler workup procedures such as filtration through silica gel followed by standard column chromatography. This level of control over the reaction trajectory ensures that the final API intermediates meet stringent purity specifications required for downstream drug development.
How to Synthesize 1,2-Diphenyl-3H-pyrrolo[1,2-a]indol-3-one Efficiently
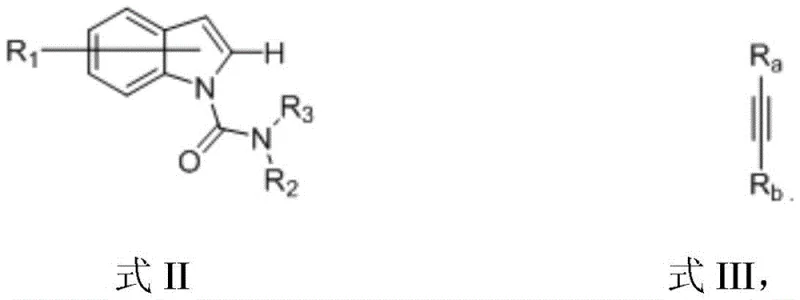
To implement this synthesis effectively, one must adhere to precise stoichiometric ratios and environmental controls to maximize yield and safety. The process begins with the preparation of the indole carbamate precursor (Formula II) and the selection of the appropriate alkyne (Formula III), ensuring both are dry and free from oxygen. The reaction is typically conducted in a Schlenk flask under an inert nitrogen atmosphere to prevent the oxidation of the sensitive organozinc reagent and the rhenium catalyst. A molar ratio of indole to alkyne of approximately 1:2.5 is recommended to drive the equilibrium towards product formation, while the catalyst loading is kept low at around 5 mol% to maintain cost efficiency without sacrificing activity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the indole compound (Formula II), alkyne (Formula III), Re2(CO)10 catalyst, zinc chloride Lewis acid, and dimethylzinc in anhydrous toluene under nitrogen protection.
- Heat the reaction mixture to a temperature range of 50 to 150°C, specifically maintaining 150°C for optimal conversion, and stir for a duration of 10 to 100 hours.
- Upon completion, dilute the reaction liquid with DCM, filter through silica gel, wash with ethyl acetate, concentrate the filtrate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this rhenium-catalyzed methodology offers tangible benefits that extend beyond mere chemical yield. The elimination of exotic or difficult-to-handle catalysts found in older protocols simplifies the sourcing of raw materials and reduces the risk of supply disruptions. Since the reaction utilizes commercially available reagents like decacarbonyldirhenium and zinc chloride, the supply chain becomes more resilient and less dependent on custom-synthesized ligands or rare earth metals that are subject to geopolitical volatility. Moreover, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or mixing rates. This reliability translates directly into enhanced supply chain reliability for critical pharmaceutical intermediates, ensuring that production timelines are met consistently.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic improvement in reaction yields and the simplification of the purification workflow. By achieving yields approaching quantitative levels in many cases, the amount of starting material wasted is significantly reduced, directly lowering the cost per kilogram of the final product. Additionally, the cleaner reaction profile means that less solvent and stationary phase are required for chromatography, further driving down operational expenses. The removal of expensive transition metal scavengers, often needed for ruthenium or palladium residues, also contributes to substantial cost savings. These efficiencies compound at scale, making the commercial production of these complex heterocycles far more economically viable than previously possible.
- Enhanced Supply Chain Reliability: The use of stable and widely available reagents ensures that production is not bottlenecked by the availability of niche chemicals. Zinc chloride and dimethylzinc are commodity chemicals with established global supply chains, reducing the lead time for high-purity pharmaceutical intermediates. The reaction's tolerance to various substituents means that a single manufacturing platform can be adapted to produce a wide library of analogues without requiring extensive re-validation of the process for each new derivative. This flexibility allows manufacturers to respond rapidly to changing demand from drug discovery teams, providing a strategic advantage in a fast-paced market environment where speed to clinic is paramount.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles by minimizing waste generation and avoiding the use of highly toxic heavy metals where possible. The solvent system, primarily based on toluene, is well-understood and easily recyclable in industrial settings, facilitating compliance with increasingly stringent environmental regulations. The ability to run the reaction at concentrations up to 0.2 mol/L without significant loss of efficiency supports efficient reactor utilization, allowing for larger batch sizes and better throughput. This scalability ensures that the transition from gram-scale laboratory synthesis to multi-ton commercial production is smooth and predictable, mitigating the risks often associated with process scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering practical guidance for technical teams evaluating this technology for their own pipelines. Understanding these nuances is essential for successfully transferring this chemistry from the lab bench to pilot plant operations.
Q: What catalyst system is utilized in this novel indolopyrrolone synthesis?
A: The process utilizes a decacarbonyldirhenium (Re2(CO)10) catalyst in conjunction with a Lewis acid such as zinc chloride and an organic zinc reagent like dimethylzinc.
Q: How does this method improve upon previous ruthenium or cobalt catalyzed routes?
A: Unlike prior art using Ru or Co catalysts which often yielded less than 79% with aryl substituents, this method achieves yields up to 98% and accommodates a broader range of substituents including halogens and alkoxy groups.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: Optimal conditions involve using toluene as the solvent at a concentration of 0.2 mol/L, heating to 150°C for approximately 24 hours, with a catalyst loading of roughly 5 mol%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolopyrrolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN109776546B for accelerating drug discovery programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from early-stage development to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions under strictly controlled conditions, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering high-quality indolopyrrolone intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic ally dedicated to optimizing your supply chain and reducing your time to market.
