Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Pharmaceutical Manufacturing
Advanced Rhenium-Catalyzed Synthesis of Indolopyrrolones for Commercial Pharmaceutical Manufacturing
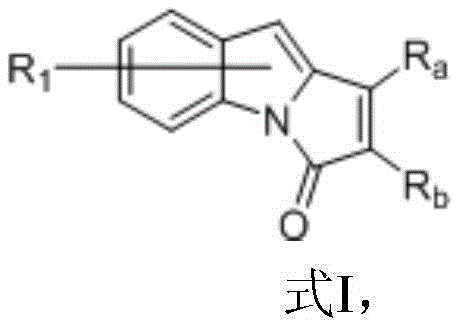
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and versatile synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN109776546B, which discloses a novel method for preparing indolopyrrolone compounds. These nitrogen-containing heterocycles are pivotal structural units found in numerous natural products and pharmaceutical agents, possessing diverse biological activities that make them highly valuable in drug discovery. The patent outlines a robust protocol involving C-H bond activation and C-N bond cleavage, utilizing a specific catalytic system that overcomes many limitations associated with prior art methodologies. For R&D directors and process chemists, understanding the nuances of this transformation is critical for integrating these scaffolds into modern medicinal chemistry pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolopyrrolone core has relied heavily on transition metal catalysis, yet existing methods often suffer from significant drawbacks regarding substrate scope and efficiency. Prior literature, such as studies published in the Journal of Organic Chemistry and the Journal of the American Chemical Society, describes the use of Ruthenium and Cobalt catalysts to mediate the reaction between indoles and alkynes. However, these conventional approaches exhibit a marked preference for methyl-substituted alkynes, struggling significantly when faced with bulkier aryl substituents. When aryl groups are introduced, the reported yields frequently drop below acceptable thresholds, often remaining under 79% for Ruthenium systems and even lower for Cobalt variants. Furthermore, alternative strategies involving sulfamides have shown extremely limited applicability to indole substrates, with isolated examples yielding as little as 39%. These inefficiencies create substantial bottlenecks in the supply chain for high-purity pharmaceutical intermediates, necessitating costly purification steps and resulting in poor atom economy.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN109776546B introduces a highly effective catalytic system centered around Rhenium complexes. This innovative approach leverages decacarbonyldirhenium in conjunction with a Lewis acid and an organozinc reagent to drive the cyclization forward with remarkable efficiency. The reaction proceeds through a concerted C-H activation and C-N bond cleavage mechanism, allowing for the direct assembly of the pyrrolidone ring onto the indole framework. Crucially, this method demonstrates exceptional tolerance for a wide array of substituents on both the indole and alkyne components, including halogens, alkyl groups, and various aryl moieties. By shifting the catalytic paradigm from Ruthenium or Cobalt to a Rhenium-based system, the process achieves consistently high yields, often exceeding 85% and reaching up to 98% for specific substrates. This represents a transformative improvement in synthetic reliability, ensuring that complex heterocyclic targets can be accessed with greater predictability and reduced waste generation.
Mechanistic Insights into Re-Catalyzed C-H Activation and Cyclization
The core of this synthetic advancement lies in the unique interplay between the Rhenium catalyst, the Lewis acid promoter, and the organozinc additive. The proposed mechanism initiates with the coordination of the decacarbonyldirhenium species to the indole substrate, facilitating the activation of the proximal C-H bond. This step is critical as it lowers the energy barrier for the subsequent insertion of the alkyne moiety. The presence of a Lewis acid, specifically zinc chloride in the preferred embodiment, plays a dual role by activating the carbonyl group of the indole-1-carboxamide precursor and stabilizing the transition state during the cyclization event. Simultaneously, the organozinc reagent, such as dimethylzinc, acts as a mild base or transmetallating agent that assists in the cleavage of the C-N bond, ultimately releasing the final indolopyrrolone product and regenerating the active catalytic species. This synergistic effect allows the reaction to proceed at elevated temperatures, typically around 150°C in toluene, without the degradation of sensitive functional groups.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or harsh acidic conditions often seen in older protocols. The specificity of the C-H activation minimizes the formation of regioisomers, which are common byproducts in non-directed functionalization reactions. Furthermore, the use of well-defined Rhenium carbonyl complexes reduces the likelihood of metal aggregation or the formation of insoluble metal sludge, which can complicate downstream processing. The reaction conditions are sufficiently mild to preserve stereochemical integrity in chiral substrates, although the current examples focus primarily on achiral building blocks. For quality control teams, this translates to a cleaner crude reaction profile, simplifying the purification workflow and reducing the burden on analytical resources. The ability to tolerate electron-withdrawing groups like chlorines and bromines without dehalogenation further underscores the chemoselectivity of this catalytic system, making it ideal for synthesizing late-stage intermediates where functional group fidelity is paramount.
How to Synthesize Indolopyrrolone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent data to ensure maximum yield and reproducibility. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen, to prevent oxidation of the sensitive organometallic reagents. The stoichiometry is precise, with a molar ratio of indole to alkyne maintained at approximately 1:2.5 to drive the equilibrium towards product formation. The catalyst loading is kept relatively low at 5 mol%, while the Lewis acid and zinc reagent are used in slight excess to ensure complete conversion. The following section outlines the standardized operational procedure derived from the exemplary embodiments provided in the intellectual property documentation.
- Combine N,N-diphenyl-1H-indole-1-carboxamide, diphenylacetylene, Re2(CO)10 catalyst, zinc chloride, and dimethylzinc in anhydrous toluene under nitrogen protection.
- Heat the reaction mixture to 150°C and maintain stirring for approximately 24 hours to facilitate C-H activation and cyclization.
- Dilute the reaction with DCM, filter through silica gel, wash with ethyl acetate, and purify the concentrated filtrate via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Rhenium-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply reliability. Traditional methods relying on Ruthenium or Cobalt often require specialized ligands or excessive catalyst loadings to achieve moderate yields, driving up the raw material costs significantly. In contrast, the new protocol utilizes commercially available decacarbonyldirhenium and simple zinc salts, which are generally more accessible and cost-effective on a metric ton scale. The elimination of complex ligand synthesis steps further streamlines the supply chain, reducing the number of vendors required and mitigating the risk of raw material shortages. Additionally, the high yields reported across a broad substrate scope mean that less starting material is wasted, directly improving the overall mass balance and reducing the cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the reduced need for extensive purification. Because the reaction generates fewer byproducts and regioisomers, the downstream processing involves simpler chromatographic separations or crystallizations, which significantly lowers solvent consumption and energy usage. The ability to use toluene, a commodity solvent, instead of exotic or highly regulated solvents further contributes to operational expenditure savings. Moreover, the robustness of the catalyst system allows for potential recycling or recovery strategies in large-scale continuous flow setups, although batch processing remains highly efficient. These factors combine to create a manufacturing route that is not only chemically superior but also financially optimized for high-volume production environments.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the versatility of the substrate scope, which allows manufacturers to source a wide variety of substituted indoles and alkynes from global chemical suppliers without fear of reaction failure. The tolerance for halogenated substrates means that downstream cross-coupling reactions can be performed on the final product, offering flexibility in synthetic route design for medicinal chemists. This adaptability ensures that production schedules are not disrupted by the unavailability of a single specific building block, as alternative analogs can be synthesized using the same core protocol. Furthermore, the reaction conditions do not require cryogenic cooling or ultra-high pressure equipment, meaning that production can be executed in standard multipurpose reactors available at most CDMO facilities worldwide.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with green chemistry principles by minimizing waste generation and avoiding the use of toxic heavy metals in excessive quantities. The reaction operates at atmospheric pressure in a closed system, reducing the risk of volatile organic compound emissions compared to processes requiring reflux of low-boiling solvents. The use of zinc chloride and dimethylzinc, while requiring careful handling, is well-established in industrial organic synthesis, and standard quenching protocols effectively neutralize these reagents before waste disposal. The high conversion rates reduce the volume of mother liquor containing unreacted starting materials, simplifying wastewater treatment processes. This compliance with environmental standards facilitates faster regulatory approvals and supports the sustainability goals of modern pharmaceutical manufacturing partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and claims within the patent specification, providing a realistic overview of what can be achieved in a production setting. Understanding these details is essential for project planning and risk assessment when integrating new synthetic methodologies into existing workflows.
Q: What are the advantages of using Rhenium catalysts over Ruthenium for this synthesis?
A: The patented method utilizes decacarbonyldirhenium which demonstrates superior compatibility with diverse aryl substituents compared to traditional Ruthenium systems, often achieving higher yields for bulky substrates.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction uses standard solvents like toluene and operates at moderate temperatures (150°C), making it highly suitable for scale-up in standard stainless steel reactors without requiring cryogenic conditions.
Q: What is the substrate scope regarding the alkyne component?
A: The method exhibits excellent tolerance for diaryl alkynes with electron-donating or withdrawing groups, although dialkyl alkynes may require optimized conditions due to lower reactivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolopyrrolone Supplier
The development of advanced synthetic routes like the Rhenium-catalyzed cyclization described in CN109776546B highlights the continuous innovation required to meet the demands of the global pharmaceutical industry. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of these technological advancements, translating cutting-edge academic and patent research into robust, scalable manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need gram-scale samples for screening or metric tons for clinical supply, we can deliver with consistency. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of characterizing complex heterocyclic intermediates to the highest international standards, guaranteeing the quality required for GMP environments.
We invite potential partners to leverage our technical expertise to optimize their supply chains for indolopyrrolone derivatives and related heterocycles. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates how implementing this specific catalytic technology can reduce your overall cost of goods sold. We encourage you to contact our technical procurement team to request specific COA data for similar compounds and discuss route feasibility assessments tailored to your target molecules. Let us help you navigate the complexities of fine chemical manufacturing with solutions that are both scientifically sound and commercially viable.
