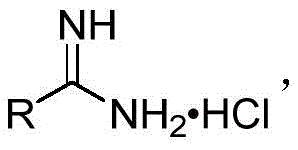
Scalable Catalyst-Free Synthesis of Polysubstituted 1,3,5-Triazines for Advanced Pharmaceutical Applications
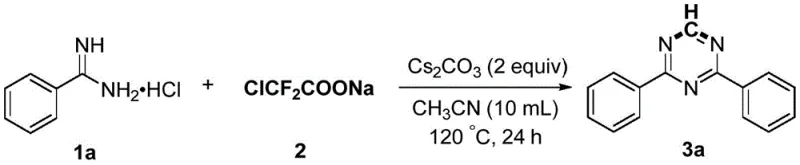
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways to access nitrogen-containing heterocycles, particularly the versatile 1,3,5-triazine scaffold. Patent CN109810069B discloses a groundbreaking preparation method for polysubstituted 1,3,5-triazines that fundamentally shifts the paradigm from traditional metal-catalyzed processes to a more economical, base-mediated approach. This innovation utilizes substituted formamidine hydrochloride as the primary reaction substrate and sodium difluorochloroacetate as a unique carbon synthon. By leveraging the cleavage of carbon-chlorine, carbon-carbon, and carbon-fluorine bonds under the action of an equivalent inorganic base, this technology enables the efficient construction of symmetrical or asymmetrical polysubstituted 1,3,5-triazines. The significance of this patent lies not only in its chemical elegance but also in its potential to serve as a reliable pharmaceutical intermediate supplier strategy, offering a route that is devoid of heavy metal contamination risks and compatible with stringent regulatory requirements for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3,5-triazines has relied heavily on the high-temperature condensation of benzamidine hydrochloride with various formylation reagents, a process documented in literature dating back to the 1960s. While foundational, these traditional methods suffer from significant drawbacks, including a narrow substrate scope, generally low yields, and harsh reaction conditions that limit their utility in modern process chemistry. More recent advancements have attempted to address these issues by employing substituted formamidine hydrochlorides with reagents like N,N-dimethylformamide or dimethyl sulfoxide acting as carbon synthons under copper or iodine catalysis. However, these catalytic systems introduce new complexities; they necessitate the use of stoichiometric oxidants and generate substantial quantities of environmentally unfriendly byproducts. Furthermore, the presence of transition metals requires rigorous and costly purification steps to meet the strict residual metal limits imposed by pharmacopeial standards, thereby increasing the overall cost reduction challenges in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to the legacy technologies, the novel approach detailed in the patent utilizes sodium difluorochloroacetate as a highly reactive carbon source in conjunction with simple inorganic bases. This method operates effectively without any transition metal catalysts or external oxidants, representing a significant leap forward in green chemistry principles. The reaction proceeds smoothly in polar aprotic solvents like acetonitrile at temperatures ranging from 80°C to 120°C. One of the most compelling features of this methodology is its exceptional versatility regarding substrate tolerance. It accommodates a wide array of substituents on the aromatic ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, bromo, trifluoromethyl, and nitro. Additionally, the system is robust enough to handle heteroaromatic substrates like pyridyl derivatives and even aliphatic cyclic groups like cyclopropyl, ensuring broad applicability for diverse drug discovery programs.
Mechanistic Insights into Base-Mediated Cyclization
The core of this transformation involves a complex cascade of bond-breaking and bond-forming events initiated by the inorganic base. The mechanism likely begins with the deprotonation of the formamidine hydrochloride to generate the free base, which then nucleophilically attacks the difluorochloroacetate species. The unique reactivity of the difluorochloroacetate allows for the sequential cleavage of the carbon-chlorine and carbon-fluorine bonds, facilitating the release of fluoride and chloride ions while constructing the central triazine ring. The use of strong bases like cesium carbonate appears to be critical in driving this equilibrium forward, potentially by sequestering the acidic byproducts and activating the carbon synthon for cyclization. This base-mediated pathway avoids the radical mechanisms often associated with copper catalysis, leading to a cleaner reaction profile with fewer side products derived from oxidative coupling or homocoupling of the starting materials.
From an impurity control perspective, this mechanism offers distinct advantages for process development. Since the reaction does not involve transition metals, the risk of metal-catalyzed decomposition or metal-complex formation is entirely eliminated. The primary byproducts identified are inorganic salts, carbon dioxide, and water, all of which are easily removed during the aqueous workup procedure. This simplicity translates directly into higher purity profiles for the crude product, reducing the burden on downstream purification units. For R&D teams focused on scaling complex pharmaceutical intermediates, understanding that the reaction tolerates both electron-rich and electron-poor substrates without significant modification of conditions suggests a robust platform technology. The ability to synthesize asymmetrical triazines by simply adjusting the molar ratio of two different formamidine substrates further highlights the mechanistic flexibility, allowing for the rapid generation of diverse chemical libraries for SAR studies.
How to Synthesize Polysubstituted 1,3,5-Triazines Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory-scale optimization and industrial production. The standard protocol involves charging a reaction vessel with the substituted formamidine hydrochloride, sodium difluorochloroacetate, and the chosen inorganic base, followed by the addition of the solvent. The mixture is then heated to the requisite temperature, typically around 120°C, and maintained for a period of 24 hours to ensure complete conversion. Upon completion, the reaction is quenched with water, and the product is extracted into an organic phase such as ethyl acetate. Following drying and concentration, the crude material can be purified via standard column chromatography using petroleum ether and ethyl acetate mixtures. For detailed standardized synthesis steps, please refer to the guide below.
- Charge the reactor with substituted formamidine hydrochloride, sodium difluorochloroacetate, and an inorganic base such as cesium carbonate.
- Add acetonitrile as the reaction solvent and heat the mixture to a temperature range of 80-120°C for approximately 24 hours.
- Quench the reaction with water, extract with ethyl acetate, dry the organic phase, and purify via column chromatography to isolate the target triazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst-free technology presents a compelling value proposition centered on cost efficiency and supply security. By removing the dependency on precious metal catalysts like copper or iodine, the raw material costs are significantly reduced, and the volatility associated with the pricing of these metals is eliminated from the supply chain equation. Moreover, the absence of toxic oxidants and heavy metals simplifies the environmental compliance landscape, reducing the costs associated with waste disposal and effluent treatment. The use of readily available commodity chemicals like sodium difluorochloroacetate and common inorganic bases ensures a stable and continuous supply of raw materials, mitigating the risk of production delays caused by specialized reagent shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and oxidants directly lowers the bill of materials for every batch produced. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor time and solvent consumption. The high yields reported, often exceeding 90% for symmetrical substrates, mean that less starting material is wasted, maximizing the atom economy and overall process efficiency. This cumulative effect results in substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous inorganic bases and commercially abundant fluorinated acetates enhances the resilience of the supply chain. Unlike specialized catalytic systems that may have long lead times or single-source suppliers, the reagents for this process are widely available from multiple global vendors. This redundancy ensures that production schedules can be maintained even during market fluctuations. Additionally, the robustness of the reaction conditions allows for flexible manufacturing across different facilities without the need for highly specialized equipment, further securing the supply continuity for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The generation of benign byproducts such as water, carbon dioxide, and inorganic salts aligns perfectly with modern green chemistry mandates and environmental regulations. This reduces the regulatory burden on manufacturing sites and minimizes the environmental footprint of the production process. The straightforward scale-up potential, evidenced by the simple thermal conditions and lack of sensitive catalytic species, means that moving from kilogram to ton-scale production can be achieved with minimal process re-engineering. This scalability is crucial for meeting the growing demand for triazine-based APIs in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, optimization, and practical application. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this triazine synthesis method over traditional catalytic routes?
A: This method eliminates the need for expensive transition metal catalysts like copper or iodine and avoids toxic oxidants. It produces environmentally benign byproducts such as inorganic salts, carbon dioxide, and water, significantly simplifying downstream purification and waste treatment.
Q: Can this process be used to create asymmetrical triazine derivatives?
A: Yes, the protocol supports the use of two different substituted formamidine hydrochlorides in a specific molar ratio (e.g., 8:2 or 2:8) alongside the carbon synthon to successfully generate asymmetrical polysubstituted 1,3,5-triazines with good selectivity.
Q: What represents the optimal reaction conditions for maximum yield?
A: Experimental data indicates that using cesium carbonate as the base in acetonitrile solvent at 120°C provides the highest yields, often exceeding 90% for symmetrical substrates like 2,4-diphenyl-1,3,5-triazine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 1,3,5-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted 1,3,5-triazine delivered meets the highest international standards. We are committed to leveraging this green chemistry innovation to provide our clients with superior quality products at competitive prices.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this metal-free route can impact your bottom line. Please contact us today to request specific COA data for our triazine portfolio and to discuss route feasibility assessments for your next-generation drug candidates. Let us be your partner in driving innovation and efficiency in your supply chain.
