Revolutionizing Azo Compound Production: Catalyst-Free Aerobic Oxidation for Commercial Scale-up
The landscape of organic synthesis is constantly evolving towards greener, more efficient methodologies that reduce environmental impact while maximizing yield. A significant breakthrough in this domain is documented in patent CN115368268A, which introduces a highly efficient method for preparing azo compounds and their applications. This technology represents a paradigm shift from traditional oxidative coupling methods that rely heavily on stoichiometric oxidants or toxic transition metal catalysts. By leveraging a catalyst-free aerobic oxidation strategy, this invention enables the direct conversion of hydroazoaromatic compounds into valuable azo derivatives under mild conditions. The process utilizes simple heating in a reaction solvent under an oxygen or air atmosphere, eliminating the need for complex additive systems. This approach not only streamlines the synthetic route but also enhances the overall sustainability of producing high-purity pharmaceutical intermediates and dye precursors. For industry leaders seeking reliable suppliers of complex organic building blocks, understanding such technological advancements is crucial for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aromatic azo compounds has been fraught with significant technical and economic challenges that hinder large-scale manufacturing efficiency. Traditional pathways often involve the Mills condensation reaction, oxidative coupling of anilines, or the reduction of nitroaromatic compounds, each carrying inherent drawbacks regarding atom economy and waste generation. Specifically, the direct dehydrogenation of hydroazobenzene compounds, while theoretically effective, has rarely been reported due to the lack of robust methodologies. Existing methods frequently employ strong oxidants like potassium chlorate in sulfuric acid or rely on transition metal catalysts such as vanadium, palladium, or titanium complexes. These conventional approaches suffer from high catalyst loading requirements, which necessitate expensive raw materials and complicate the post-reaction purification process. Furthermore, the presence of heavy metal residues often requires rigorous and costly removal steps to meet the stringent purity specifications demanded by the pharmaceutical and agrochemical industries. The low yields and complicated workup procedures associated with these legacy methods create bottlenecks in the supply chain, leading to increased lead times and higher production costs for high-purity azo intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach outlined in the patent data offers a streamlined, additive-free solution that fundamentally alters the economics of azo compound synthesis. This method achieves the efficient conversion of hydrogenated azobenzene and hydrogenated heterocyclic azo derivatives into their corresponding azo forms simply by heating in a solvent under an oxygen or air atmosphere. The elimination of external catalysts means there are no metal residues to remove, drastically simplifying the downstream processing to a mere water wash and extraction. This simplicity translates directly into operational efficiency, as the reaction conditions are mild, typically ranging from 40°C to 150°C, and the reaction times are reasonable, spanning from 2 to 15 hours depending on the substrate. The versatility of this system is further highlighted by its compatibility with a wide array of solvents including DMF, DMSO, and ethyl acetate, providing flexibility for process optimization. By adopting this catalyst-free aerobic oxidation strategy, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through reduced material costs and simplified waste management protocols.
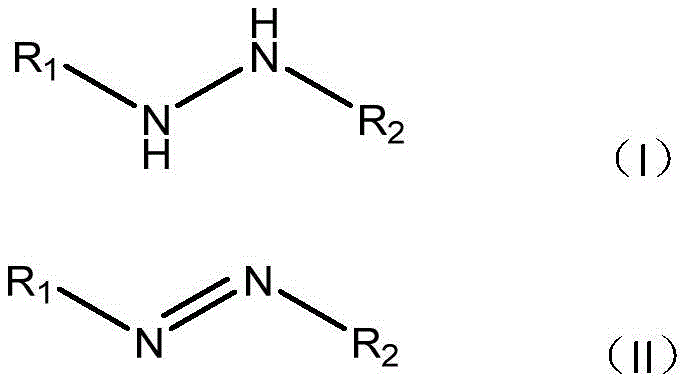
Mechanistic Insights into Catalyst-Free Aerobic Oxidation
The core mechanistic advantage of this technology lies in its ability to utilize molecular oxygen as a green oxidant without the need for activation by transition metals. In traditional catalytic cycles, the metal center facilitates electron transfer, but this new method suggests a direct thermal activation pathway where the hydrazo bond is susceptible to oxidation by ambient oxygen when energized by heat in a polar aprotic solvent like DMF. This mechanism avoids the formation of metal-ligand complexes that can sequester the product or generate difficult-to-remove impurities. The reaction proceeds through a dehydrogenation pathway where the N-N single bond in the hydrazo precursor is oxidized to an N=N double bond, releasing water as the primary byproduct. This clean transformation is critical for maintaining high purity profiles, as it minimizes the generation of side products that typically arise from over-oxidation or catalyst decomposition. The robustness of this mechanism is evidenced by its tolerance to diverse functional groups, allowing for the synthesis of complex molecules without protecting group strategies. For R&D directors focused on impurity control, this mechanism offers a predictable and clean reaction profile that simplifies analytical validation and regulatory filing.
Furthermore, the patent elucidates a fascinating tandem application where the oxidative power of the system is harnessed for multi-component reactions. Beyond simple dehydrogenation, the method can be coupled with enone compounds to facilitate epoxidation reactions, effectively synthesizing epoxychalcones alongside the azo products. This dual functionality implies that the reactive oxygen species generated in situ or the specific reaction environment created by the heating of hydrazo compounds in DMF can activate other substrates. In the context of epoxychalcone synthesis, the addition of a carbonate base, such as potassium carbonate, enables the concurrent formation of the epoxide ring while the hydrazo compound is oxidized to the azo form. This synergistic effect maximizes atom economy and reactor utilization, as two valuable chemical transformations occur in a single pot. Such mechanistic ingenuity reduces the number of unit operations required, thereby lowering energy consumption and capital expenditure for equipment. Understanding these mechanistic nuances allows process chemists to design more efficient synthetic routes for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Azo Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal yield and safety. The general procedure involves dissolving the hydrazo precursor in a suitable solvent such as DMF and subjecting the mixture to an oxygen-rich environment at elevated temperatures. The simplicity of the workup, involving water addition and organic extraction, makes this protocol highly accessible for rapid scale-up. Detailed standard operating procedures regarding stoichiometry, specific temperature ramps, and safety precautions for handling oxygen atmospheres are critical for successful execution. The following guide outlines the fundamental steps derived from the patent examples to assist technical teams in replicating this high-efficiency process.
- Dissolve the hydrazo compound precursor in DMF solvent within a reaction vessel equipped for heating.
- Introduce an oxygen or air atmosphere and heat the mixture to a temperature between 40°C and 150°C for 2 to 15 hours.
- Upon completion, add water to the reaction mixture, extract with ethyl acetate, wash with brine, dry, and concentrate to obtain the pure azo product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology presents a compelling value proposition centered around cost stability and supply reliability. The primary driver for cost optimization is the complete elimination of expensive transition metal catalysts and stoichiometric oxidants, which are subject to volatile market pricing and supply constraints. By removing these inputs, the variable cost of goods sold is significantly reduced, and the dependency on specialized chemical suppliers is minimized. Additionally, the simplified purification process, which avoids column chromatography and extensive metal scavenging steps, leads to substantial savings in labor, consumables, and waste disposal fees. This streamlined workflow enhances the overall throughput of the manufacturing facility, allowing for faster turnaround times on customer orders. The ability to produce high-purity azo intermediates with minimal environmental footprint also aligns with increasingly strict global regulations on industrial emissions and hazardous waste, mitigating compliance risks.
- Cost Reduction in Manufacturing: The absence of catalysts eliminates the need for costly metal removal resins and reduces the consumption of high-purity reagents, leading to a leaner cost structure. The use of air or oxygen as the oxidant is economically superior to purchasing specialized chemical oxidants, providing a sustainable long-term advantage in raw material sourcing. Furthermore, the high yields reported across various substrates mean less starting material is wasted, improving the overall material balance and reducing the cost per kilogram of the final product. These factors collectively contribute to a more competitive pricing model for downstream customers seeking cost-effective solutions for their synthesis pipelines.
- Enhanced Supply Chain Reliability: Relying on abundant commodities like air and common solvents like DMF reduces the risk of supply disruptions caused by shortages of niche catalysts. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with pharmaceutical clients. The simplified process flow also reduces the likelihood of operational delays caused by complex purification bottlenecks, ensuring that delivery schedules are met reliably. This stability is crucial for just-in-time manufacturing environments where inventory buffers are kept to a minimum to optimize working capital.
- Scalability and Environmental Compliance: The straightforward nature of the reaction, involving simple heating and stirring, translates easily from gram-scale laboratory experiments to ton-scale commercial production without significant re-engineering. The reduction in hazardous waste generation, particularly heavy metal sludge, simplifies wastewater treatment requirements and lowers the environmental compliance burden. This green chemistry approach not only improves the corporate sustainability profile but also future-proofs the manufacturing process against tightening environmental regulations. The ability to scale up complex azo derivatives efficiently positions the manufacturer as a strategic partner capable of supporting growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aerobic oxidation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios.
Q: Does this method require expensive transition metal catalysts?
A: No, the patented process operates without any additives or catalysts, utilizing molecular oxygen or air as the sole oxidant, which significantly simplifies downstream purification.
Q: What is the functional group tolerance of this oxidation strategy?
A: The method demonstrates excellent tolerance for various substituents including halogens, alkoxy groups, nitro groups, and heterocycles, making it suitable for complex pharmaceutical intermediates.
Q: Can this process be coupled with other reactions?
A: Yes, the generated intermediate can participate in tandem reactions, such as the epoxidation of enones, allowing for the simultaneous synthesis of epoxychalcones and azo compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azo Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free synthesis technologies in modernizing the production of fine chemical intermediates. Our team of expert process chemists is dedicated to translating innovative academic and patent discoveries into robust, commercially viable manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the laboratory are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of azo compounds or epoxychalcones delivered meets the highest international standards. We are committed to being a reliable partner who not only supplies chemicals but also provides technical solutions that enhance our clients' operational efficiency.
We invite you to engage with our technical procurement team to discuss how this advanced oxidation method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener synthesis route. We encourage you to contact us to obtain specific COA data for relevant intermediates and to receive detailed route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the global supply chain for pharmaceutical and agrochemical intermediates.
