Scalable Metal-Free Synthesis of 1,2,4-Triazole Derivatives Using Visible Light Photochemistry
Scalable Metal-Free Synthesis of 1,2,4-Triazole Derivatives Using Visible Light Photochemistry
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for sustainable and efficient manufacturing processes. Patent CN114456121A introduces a groundbreaking methodology for the construction of 1,2,4-triazole derivatives, a structural motif ubiquitous in bioactive molecules and functional materials. Unlike traditional approaches that rely heavily on expensive transition metal catalysts and harsh thermal conditions, this innovation leverages visible light photochemistry to drive the reaction forward. By utilizing blue LED irradiation, the process activates diazo compounds to generate active carbene species in situ, which are subsequently captured by nitrile solvents. This elegant strategy not only simplifies the reaction setup but also aligns perfectly with the principles of green chemistry by eliminating heavy metal contamination risks. For R&D directors and process chemists, this represents a pivotal shift towards cleaner, more controllable synthetic pathways that can be seamlessly integrated into modern pharmaceutical production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole derivatives has been dominated by methodologies requiring stoichiometric or catalytic amounts of transition metals such as copper or rhodium. These conventional routes often necessitate rigorous exclusion of air and moisture, specialized equipment, and extensive downstream purification to remove trace metal residues that are strictly regulated in pharmaceutical applications. Furthermore, the thermal activation required for many of these reactions can lead to the decomposition of sensitive functional groups, limiting the substrate scope and overall yield. The reliance on precious metal catalysts also introduces significant volatility in raw material costs and supply chain stability, creating bottlenecks for procurement managers aiming to optimize production budgets. Additionally, the generation of hazardous waste streams associated with metal removal processes poses substantial environmental compliance challenges for large-scale facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalyst-free, visible-light-driven multicomponent reaction that fundamentally alters the economic and operational dynamics of triazole synthesis. By employing simple blue LED lights as the sole energy source, the method activates diazo compounds under exceptionally mild conditions, typically at room temperature, thereby preserving the integrity of complex molecular architectures. The nitrile solvent plays a dual role, acting simultaneously as the reaction medium and a critical reactant that captures the transient carbene species to form a 1,3-dipole intermediate. This intermediate then undergoes a highly regioselective [3+2] cycloaddition with azo compounds to furnish the target 1,2,4-triazole scaffold. 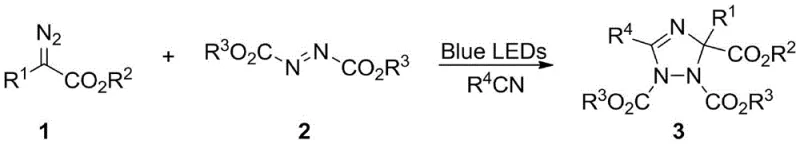 This streamlined process eliminates the need for expensive additives and simplifies the workup procedure to basic silica gel chromatography, offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing.
This streamlined process eliminates the need for expensive additives and simplifies the workup procedure to basic silica gel chromatography, offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Visible-Light Driven Carbene Capture and Cycloaddition
The mechanistic pathway of this transformation is a fascinating example of how photochemical energy can be harnessed to access reactive intermediates without thermal stress. Upon irradiation with blue LEDs, the diazo compound undergoes photolysis to extrude a molecule of nitrogen gas, generating a highly reactive carbene species. This step is critical as it avoids the use of metal carbenoids which are often difficult to control. The generated carbene is immediately intercepted by the lone pair of electrons on the nitrogen atom of the nitrile solvent, forming a nitrile ylide. This 1,3-dipolar species is the key linchpin of the reaction, possessing the necessary electronic configuration to engage in cycloaddition chemistry. The subsequent interaction between the nitrile ylide and the azo compound proceeds through a concerted [3+2] cycloaddition mechanism, constructing the five-membered triazole ring with high precision. ![Mechanism showing carbene generation from diazo compound, capture by nitrile to form ylide, and [3+2] cycloaddition with azo compound](/insights/img/1-2-4-triazole-synthesis-photochemical-pharma-supplier-20260305044445-02.webp) Understanding this mechanism allows chemists to fine-tune reaction parameters, such as light intensity and solvent choice, to maximize efficiency and minimize side reactions, ensuring a clean impurity profile essential for high-purity API intermediates.
Understanding this mechanism allows chemists to fine-tune reaction parameters, such as light intensity and solvent choice, to maximize efficiency and minimize side reactions, ensuring a clean impurity profile essential for high-purity API intermediates.
From an impurity control perspective, the absence of metal catalysts drastically reduces the complexity of the impurity spectrum. Traditional metal-catalyzed reactions often suffer from issues related to metal-ligand complexes or homocoupling byproducts that are structurally similar to the desired product and difficult to separate. In this photochemical protocol, the primary byproduct is nitrogen gas, which harmlessly vents from the reaction system, adhering to green chemistry ideals. The remaining impurities are typically unreacted starting materials or simple hydrolysis products that are easily removed during the standard silica gel column chromatography purification step described in the examples. This inherent cleanliness of the reaction profile significantly reduces the burden on quality control laboratories and shortens the overall production cycle time, providing a distinct advantage for supply chain heads focused on throughput and consistency.
How to Synthesize 1,2,4-Triazole Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory scale optimization and industrial scale-up. The protocol involves mixing the diazo and azo precursors in a nitrile solvent, followed by irradiation with commercially available blue LEDs. The reaction progress is conveniently monitored using thin-layer chromatography (TLC), allowing for precise endpoint determination to prevent over-reaction or decomposition. Once the reaction is complete, the solvent is removed under reduced pressure, and the crude product is purified using standard flash chromatography techniques with petroleum ether and ethyl acetate mixtures. The detailed standardized synthesis steps for implementing this protocol in your facility are outlined below.
- Combine the diazo compound (0.4 mmol) and azo compound (0.1 mmol) in a reaction vessel containing acetonitrile (1 mL) as both solvent and reactant.
- Irradiate the reaction mixture with blue LEDs at room temperature until TLC analysis indicates complete consumption of the starting materials.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this photochemical methodology offers compelling strategic advantages that extend beyond simple yield improvements. The elimination of transition metal catalysts removes a major cost driver and supply risk factor, as prices for metals like palladium, rhodium, and copper can be highly volatile and subject to geopolitical constraints. Furthermore, the simplified purification process reduces the consumption of specialized scavenging resins and solvents required for metal removal, leading to substantial cost savings in consumables. The mild reaction conditions also imply lower energy costs compared to high-temperature thermal processes, contributing to a reduced carbon footprint and enhanced sustainability metrics which are increasingly important for corporate social responsibility reporting.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete removal of expensive metal catalysts and ligands from the bill of materials. In traditional synthesis, these components can account for a significant portion of the raw material cost, especially when high purity is required. By replacing them with inexpensive blue LED lights and common nitrile solvents, the direct material cost is drastically lowered. Additionally, the simplified workup procedure reduces labor hours and solvent usage associated with complex metal scrubbing protocols, further enhancing the overall cost efficiency of the manufacturing process without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as acetonitrile and readily available diazo and azo precursors ensures a stable and resilient supply chain. Unlike specialized catalysts that may have long lead times or single-source suppliers, the reagents for this process are widely produced and stocked by multiple global vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the photochemical reaction allows for flexible manufacturing schedules, as the reaction can be paused or resumed by simply controlling the light source, offering greater operational agility in response to market demand fluctuations.
- Scalability and Environmental Compliance: The patent explicitly highlights the compatibility of this method with flow photochemistry, a technology renowned for its ability to scale light-mediated reactions safely and efficiently. Flow systems provide superior light penetration and heat transfer compared to batch reactors, enabling the safe handling of energetic diazo compounds on a multi-kilogram or ton scale. From an environmental standpoint, the generation of nitrogen gas as the sole byproduct minimizes waste disposal costs and regulatory burdens. The absence of heavy metals in the effluent stream simplifies wastewater treatment requirements, ensuring compliance with stringent environmental regulations and facilitating smoother permitting processes for new production lines.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is critical for integrating this novel synthetic route into existing manufacturing workflows effectively.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN114456121A operates without any transition metal catalysts or additives, relying solely on visible light energy to drive the reaction.
Q: What is the role of the nitrile solvent in this reaction?
A: The nitrile solvent acts as a carbene capture reagent, generating a reactive 1,3-dipole intermediate (nitrile ylide) which subsequently undergoes cycloaddition with the azo compound.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly mentions that the mild reaction conditions and use of flow photochemistry methods make this route highly amenable to large-scale synthesis and industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photochemical synthesis in modern drug development and are committed to helping our partners leverage these advancements. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114456121A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced flow photochemistry reactors and rigorous QC labs capable of meeting stringent purity specifications required for global pharmaceutical markets. We understand that every molecule tells a story, and we are dedicated to writing the next chapter of your success with efficiency and precision.
We invite you to explore how this metal-free technology can optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data for our existing triazole inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us partner with you to deliver high-quality chemical solutions that drive your business forward.
