Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
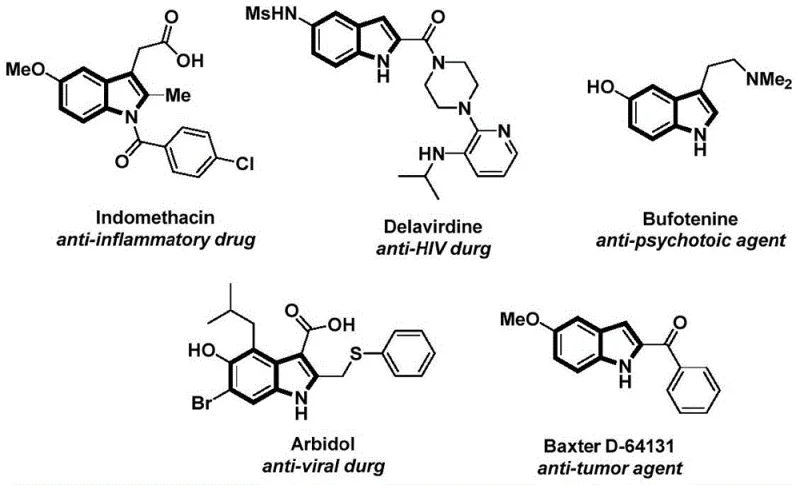
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly the indole nucleus, which serves as a privileged structure in numerous bioactive molecules. As illustrated in the structural diversity of known drugs such as Indomethacin and Delavirdine, the N-acyl indole motif is ubiquitous in anti-inflammatory, anti-HIV, and anti-tumor agents. Addressing the critical need for efficient synthetic routes, Chinese Patent CN112898192B discloses a groundbreaking preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. This innovation represents a significant leap forward in process chemistry, offering a streamlined pathway that bypasses the hazards associated with traditional gaseous carbon monoxide usage while maintaining high reaction efficiency and broad substrate tolerance.
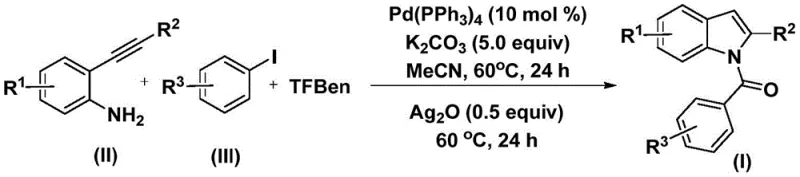
The disclosed technology utilizes readily available 2-alkynyl anilines and aryl iodides as starting materials, reacting them in the presence of a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen). This approach not only mitigates safety risks but also simplifies the engineering controls required for manufacturing. For R&D directors and process chemists, the ability to generate these high-value intermediates under relatively mild conditions (60°C) using a homogeneous palladium system offers a compelling alternative to multi-step sequences or harsh acidic cyclizations. The method ensures that the resulting N-acyl indoles are obtained with high purity, directly addressing the stringent quality specifications demanded by global regulatory bodies for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has relied heavily on direct carbonylation using carbon monoxide gas, a reagent that poses severe safety challenges due to its high toxicity and flammability. Traditional protocols often require high-pressure autoclaves and specialized gas handling infrastructure, which drastically increases capital expenditure and operational complexity for chemical manufacturers. Furthermore, conventional methods frequently suffer from poor atom economy and limited functional group tolerance, necessitating extensive protection and deprotection strategies that elongate the synthetic timeline. The reliance on gaseous reagents also introduces variability in mass transfer rates, leading to inconsistent batch-to-batch reproducibility and potential impurity profiles that are difficult to control. These factors collectively hinder the cost-effective commercial scale-up of complex pharmaceutical intermediates, creating bottlenecks in the supply chain for critical drug substances.
The Novel Approach
In stark contrast, the novel methodology described in the patent employs a solid carbon monoxide substitute, TFBen, which decomposes in situ to release CO in a controlled manner. This shift from gas to solid reagents fundamentally alters the risk profile of the reaction, allowing it to be conducted in standard glass-lined or stainless steel reactors without the need for high-pressure containment. The process operates at a moderate temperature of 60°C in acetonitrile, utilizing potassium carbonate as a base and tetrakis(triphenylphosphine)palladium as the catalyst. This mild condition profile preserves sensitive functional groups on the aromatic rings, enabling the direct synthesis of diversified N-acyl indoles from simple precursors. By integrating the carbonylation and cyclization steps into a cohesive one-pot sequence followed by a silver oxide-mediated oxidation, the new route significantly reduces waste generation and solvent consumption, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction mechanism proceeds through a sophisticated catalytic cycle initiated by the oxidative addition of the aryl iodide to the zero-valent palladium species. This forms an aryl-palladium(II) intermediate, which subsequently undergoes migratory insertion of the carbon monoxide generated from the thermal decomposition of TFBen. The resulting acyl-palladium species then reacts with the amine functionality of the 2-alkynyl aniline, forming an amide bond through reductive elimination. This step is critical as it establishes the N-acyl linkage prior to ring closure. The use of TFBen ensures a steady, low-concentration supply of CO, preventing catalyst poisoning that often occurs with excess gaseous CO, thereby maintaining high turnover numbers throughout the 24-hour initial reaction phase.
Following the formation of the amide intermediate, the addition of silver oxide triggers the final cyclization event. Silver oxide acts as both an oxidant and a promoter for the intramolecular hydroamination or carbopalladation of the alkyne moiety, closing the five-membered pyrrole ring to yield the final N-acyl indole structure. This two-stage thermal protocol, with each stage lasting 24 hours at 60°C, ensures complete conversion of the starting materials. From an impurity control perspective, the specificity of the palladium insertion and the mild nature of the silver-mediated cyclization minimize the formation of side products such as homocoupling dimers or over-carbonylated species. This mechanistic clarity provides process engineers with the confidence to optimize parameters for large-scale production, ensuring that the critical quality attributes of the API intermediate remain within tight specifications.
How to Synthesize N-Acyl Indole Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for generating N-acyl indoles with high fidelity. The procedure begins with the precise weighing of tetrakis(triphenylphosphine)palladium, potassium carbonate, TFBen, 2-alkynylaniline, and the specific aryl iodide derivative. These components are suspended in acetonitrile, a solvent chosen for its ability to dissolve both organic substrates and inorganic bases effectively. The mixture is subjected to thermal agitation at 60°C for a full day to allow the carbonylation cascade to reach completion. Subsequently, silver oxide is introduced to the reaction vessel to drive the cyclization, followed by another 24-hour heating period. Detailed standardized synthesis steps are provided in the guide below to ensure operational consistency.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide to the mixture and continue heating at 60°C for an additional 24 hours to promote cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers transformative benefits regarding cost structure and logistical stability. The elimination of high-pressure carbon monoxide gas removes a major safety hazard and the associated regulatory burden, simplifying facility compliance and insurance costs. Moreover, the reliance on commercially available, shelf-stable solid reagents like TFBen and aryl iodides ensures a resilient supply chain that is less susceptible to the disruptions often seen with specialty gases. The mild reaction conditions also translate to lower energy consumption compared to high-temperature or high-pressure alternatives, contributing to a reduced carbon footprint and lower utility expenses per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The substitution of gaseous CO with a solid surrogate eliminates the need for expensive high-pressure reactor vessels and specialized gas delivery infrastructure, leading to substantial capital expenditure savings. Additionally, the high atom economy of the carbonylation step minimizes raw material waste, while the use of a common solvent like acetonitrile facilitates efficient recovery and recycling, further driving down the overall cost of goods sold for these valuable intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted anilines and aryl iodides, manufacturers can diversify their supplier base and mitigate the risk of single-source dependency. The robustness of the reaction against varying electronic properties of substrates means that grade variations in raw materials are less likely to cause batch failures, ensuring consistent on-time delivery performance for downstream pharmaceutical clients who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The protocol's operation at atmospheric pressure and moderate temperatures makes it inherently safer and easier to scale from pilot plant to multi-ton commercial production without significant re-engineering. The simplified workup procedure, involving filtration and standard chromatography, reduces the volume of hazardous waste streams, aiding facilities in meeting increasingly stringent environmental discharge regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented technology in an industrial setting. Understanding the nuances of reagent selection and reaction kinetics is essential for successful technology transfer. The answers provided are derived directly from the experimental data and mechanistic insights contained within the patent documentation, offering a reliable foundation for process development decisions.
Q: What is the advantage of using TFBen over carbon monoxide gas?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas cylinders, significantly enhancing operational safety and simplifying reactor requirements for industrial scale-up.
Q: What is the role of Silver Oxide in this synthesis?
A: Silver oxide acts as a crucial oxidant and cyclization promoter in the second stage of the reaction, facilitating the intramolecular closure to form the indole core structure under mild thermal conditions.
Q: Does this method support diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, and alkoxy groups on both the aniline and aryl iodide components without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the drug development lifecycle. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of N-acyl indole meets the exacting standards required by global regulatory agencies. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that accelerate our partners' clinical timelines.
We invite you to collaborate with us to leverage this advanced carbonylation technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall development costs.
