Scalable Copper-Catalyzed Synthesis of Benzothiazolones for High-Purity Pharmaceutical Intermediates
Scalable Copper-Catalyzed Synthesis of Benzothiazolones for High-Purity Pharmaceutical Intermediates
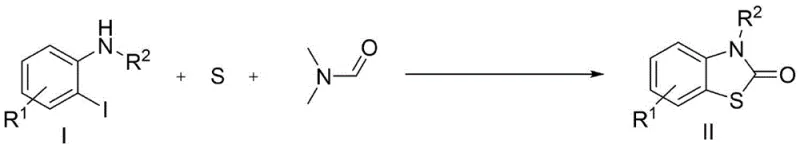
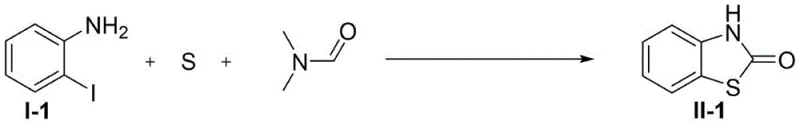
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and economically viable synthetic routes for constructing privileged heterocyclic scaffolds. Among these, the benzothiazolone core represents a critical structural motif found in numerous bioactive molecules, ranging from antipsychotic agents to potential anticancer therapeutics. A groundbreaking development in this domain is detailed in patent CN113582943A, which discloses a novel, one-pot synthetic methodology for preparing benzothiazolone compounds. This technology leverages an economical copper catalytic system to transform simple, commercially available starting materials—specifically o-iodoaniline derivatives, elemental sulfur, and DMF—into high-value benzothiazolone products. The process is distinguished by its remarkable efficiency, capable of delivering target yields of up to 90% within a drastically reduced reaction timeframe of merely 3 hours. For R&D directors and process chemists, this represents a significant leap forward in synthetic strategy, offering a pathway that bypasses the harsh conditions and toxic reagents associated with legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzothiazolone skeleton has been fraught with significant synthetic challenges and safety hazards that impede large-scale manufacturing. Traditional protocols often rely on the cyclization of ortho-aminothiophenols with phosgene or its equivalents, which involves the handling of highly toxic carbon monoxide sources. Alternatively, other established routes require the prior synthesis of substituted ortho-aminothiophenes, which are frequently difficult to access, unstable, and malodorous, posing severe occupational health risks and complicating supply chain logistics. Furthermore, earlier reported one-pot methods, such as those utilizing potassium sulfide and copper sulfate, have suffered from prolonged reaction times extending up to 18 hours and the necessity for stoichiometric amounts of strong bases and ligands. These inefficiencies not only drive up energy consumption and production costs but also limit the throughput capacity of manufacturing facilities, creating bottlenecks for the reliable supply of pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in patent CN113582943A introduces a streamlined, atom-economical solution that fundamentally reshapes the production landscape for benzothiazolones. By employing elemental sulfur as the sulfur source and DMF as both the solvent and the carbonyl source, this new route eliminates the need for hazardous gases and unstable thiol precursors. The reaction operates under remarkably mild conditions, utilizing a catalytic amount of inexpensive cuprous chloride (CuCl) and an organic base like DBU. Crucially, the process does not require a strictly inert atmosphere; optimization data confirms that the reaction proceeds with high efficiency even under ambient air, removing the need for complex nitrogen purging systems. This shift from multi-step, hazard-prone sequences to a direct, 3-hour one-pot transformation under aerobic conditions exemplifies a major advancement in green chemistry and process intensification.
Mechanistic Insights into CuCl-Catalyzed Cyclization
The success of this synthetic transformation relies on a sophisticated yet operationally simple copper-catalyzed cascade that facilitates the simultaneous formation of C-S and C-N bonds alongside carbonyl insertion. The mechanism likely initiates with the oxidative addition of the low-valent copper species into the carbon-iodine bond of the o-iodoaniline substrate, generating a reactive aryl-copper intermediate. This species subsequently undergoes insertion of elemental sulfur, a step that is traditionally challenging due to the polymeric nature of sulfur, but is effectively managed here by the specific coordination environment provided by the solvent and base. The presence of DBU plays a dual role: it acts as a proton scavenger to neutralize the HI byproduct generated during the initial activation, and it likely assists in activating the DMF molecule for the subsequent carbonyl transfer. This intricate interplay allows for the seamless construction of the benzothiazolone ring system without the accumulation of toxic intermediates, ensuring a clean reaction profile that is essential for maintaining high purity standards in API manufacturing.
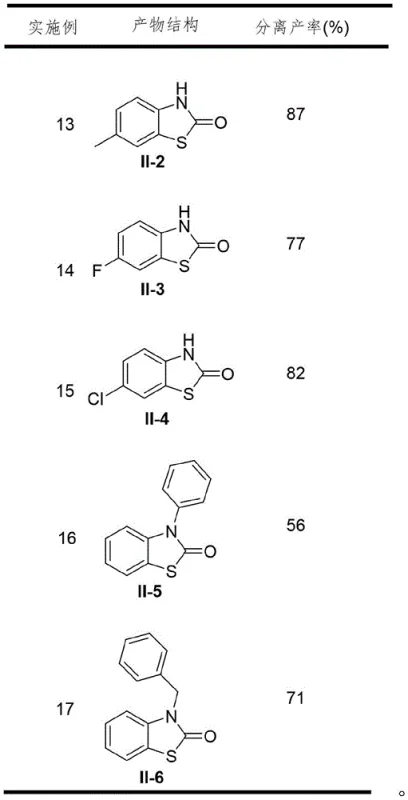
Furthermore, the robustness of this catalytic system is evidenced by its exceptional tolerance to a broad spectrum of functional groups, a critical factor for medicinal chemists designing diverse compound libraries. The electronic nature of substituents on the aromatic ring, whether electron-donating groups like methyl and methoxy or electron-withdrawing groups like halogens and nitro groups, does not significantly impede the catalytic cycle. This universality suggests that the rate-determining step is likely the initial oxidative addition or the sulfur insertion, both of which are facilitated by the specific choice of the CuCl/DBU catalyst system. The ability to accommodate N-substituted anilines, yielding N-aryl and N-benzyl benzothiazolones, further expands the utility of this method for generating structurally complex analogs. Such mechanistic resilience ensures that the process remains stable and predictable even when scaling up to multi-kilogram batches, minimizing the risk of batch-to-batch variability.
How to Synthesize Benzothiazolone Efficiently
Implementing this cutting-edge synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The protocol is designed for simplicity, avoiding the need for specialized high-pressure equipment or glovebox techniques. The following guide outlines the standardized procedure derived from the optimal conditions identified in the patent examples, specifically referencing the high-yielding model reaction. Operators should note that while the reaction is tolerant to air, maintaining consistent stirring and temperature control is vital for reproducibility. For detailed safety data sheets regarding elemental sulfur and DMF handling, please consult your site's EHS guidelines before proceeding.
- Charge a reactor with o-iodoaniline derivative, elemental sulfur, cuprous chloride catalyst, and DBU base in DMF solvent.
- Heat the reaction mixture to 120°C and stir under an air atmosphere for 3 hours to facilitate cyclization.
- Upon completion, dilute with ether, filter through silica gel, concentrate, and purify via flash chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The transition to this methodology directly addresses several pain points associated with the sourcing and manufacturing of heterocyclic intermediates. By replacing expensive and hazardous reagents with commodity chemicals like elemental sulfur and DMF, the raw material cost profile is significantly optimized. Moreover, the drastic reduction in reaction time from nearly a day to just 3 hours translates into a substantial increase in asset utilization, allowing existing reactor infrastructure to produce significantly more volume over the same period. This efficiency gain is compounded by the elimination of rigorous inert gas requirements, simplifying the engineering controls needed for safe operation and reducing utility costs associated with nitrogen generation and purification.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of costly catalysts and reagents with earth-abundant copper and bulk chemicals. Traditional methods often rely on palladium catalysts or specialized ligands that contribute heavily to the bill of materials; in contrast, the use of cuprous chloride at low loading (0.05 equiv) minimizes heavy metal residue concerns and lowers catalyst procurement costs. Additionally, the simplified workup procedure, which involves basic filtration and chromatography, reduces the consumption of solvents and silica gel compared to multi-step extractions. These factors collectively contribute to a leaner cost structure, enabling more competitive pricing for the final pharmaceutical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche starting materials that have limited global suppliers. This new method utilizes o-iodoanilines and elemental sulfur, both of which are produced at massive scales for various industrial applications, ensuring a stable and diversified supply base. The robustness of the reaction under air atmosphere further de-risks the manufacturing process by removing the dependency on high-purity nitrogen supplies, which can be a logistical bottleneck in certain geographic regions. Consequently, manufacturers can maintain consistent production schedules and meet tight delivery windows for downstream API clients, fostering stronger long-term partnerships and reducing the risk of stockouts.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles, facilitating easier permitting and waste management. The avoidance of toxic carbon monoxide and malodorous thiols significantly improves the workplace safety profile and reduces the burden on scrubber systems and waste treatment facilities. The high atom economy and reduced reaction time also mean lower energy consumption per kilogram of product, contributing to a smaller carbon footprint. As regulatory bodies increasingly scrutinize the environmental impact of chemical manufacturing, adopting such cleaner technologies positions companies favorably for audits and sustainability certifications, ensuring long-term operational viability in a tightening regulatory landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzothiazolone synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production workflows or for assessing the quality of intermediates sourced from external partners.
Q: What are the advantages of this benzothiazolone synthesis method over traditional routes?
A: Unlike traditional methods that often require toxic carbon monoxide or difficult-to-source ortho-aminothiophenes, this novel approach utilizes readily available o-iodoaniline and elemental sulfur. It operates under mild conditions with a significantly reduced reaction time of only 3 hours compared to previous methods requiring up to 18 hours, while achieving yields up to 90%.
Q: Is an inert atmosphere required for this copper-catalyzed reaction?
A: No, one of the key operational advantages of this protocol is that it proceeds efficiently under an air atmosphere. Optimization studies demonstrated that using air instead of nitrogen did not compromise the yield, with Example 8 achieving 90% yield under aerobic conditions, thereby simplifying the equipment requirements and reducing operational costs.
Q: What is the substrate scope for this benzothiazolone synthesis?
A: The method exhibits excellent functional group tolerance. It is compatible with a wide range of substituents on the phenyl ring, including halogens (fluorine, chlorine), alkyl groups (methyl, tert-butyl), and various N-substituents (phenyl, benzyl). This versatility makes it highly suitable for generating diverse libraries of bioactive benzothiazolone scaffolds for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced on an industrial scale. We are equipped with state-of-the-art reactors capable of handling the specific thermal and atmospheric conditions required for this copper-catalyzed process, alongside stringent purity specifications enforced by our rigorous QC labs. Whether you require custom synthesis of novel benzothiazolone derivatives or reliable supply of established intermediates, our infrastructure is designed to deliver consistency and quality that meets the exacting standards of the global pharmaceutical industry.
We invite you to leverage our technical proficiency to optimize your supply chain and reduce your overall cost of goods. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team to request specific COA data for our benzothiazolone portfolio and to discuss route feasibility assessments for your proprietary molecules. Let us help you navigate the complexities of fine chemical manufacturing with a solution that balances efficiency, cost, and reliability.
