Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These nitrogen-containing five-membered heterocycles are critical structural motifs found in numerous bioactive molecules, including well-known drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity of current therapeutics. The introduction of a trifluoromethyl group into these heterocyclic systems is particularly valuable, as it markedly enhances physicochemical properties such as metabolic stability, lipophilicity, and bioavailability. This patent presents a transformative approach that addresses long-standing challenges in synthesizing these high-value intermediates, offering a pathway that is both chemically elegant and commercially viable for global supply chains.
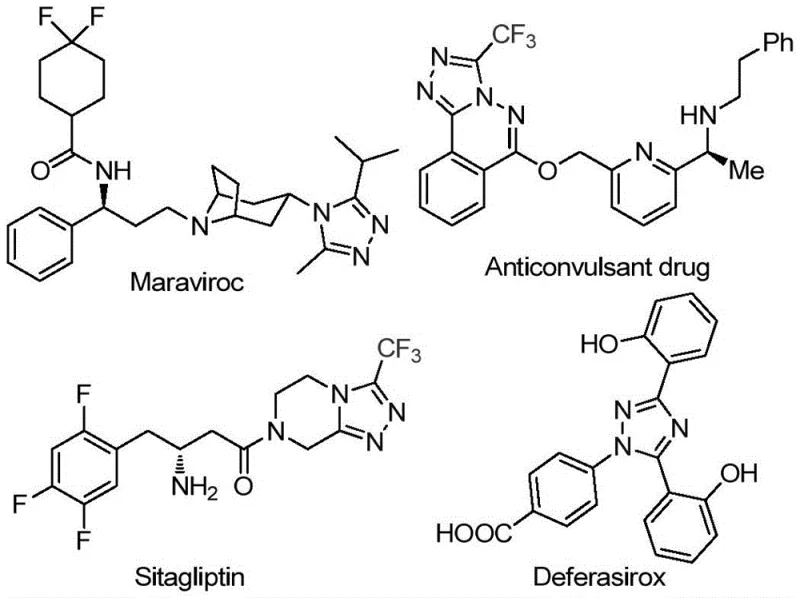
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing both trifluoromethyl and acyl groups, has been fraught with significant technical and economic hurdles. Traditional routes often rely on the use of expensive transition metal catalysts, which not only inflate the raw material costs but also introduce complex downstream processing requirements for removing trace metal impurities to meet stringent pharmaceutical standards. Furthermore, many existing protocols necessitate rigorous anhydrous and oxygen-free conditions, requiring specialized equipment and inert gas manifolds that increase capital expenditure and operational complexity. The sensitivity of reagents in conventional methods often leads to inconsistent yields and difficulties in scaling up from laboratory benchtop to multi-ton production, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to meet high-volume demand.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN113105402B leverages a non-metal promoted system utilizing elemental iodine and dimethyl sulfoxide (DMSO). This innovative strategy eliminates the need for toxic heavy metals and harsh reaction environments, thereby streamlining the entire manufacturing workflow. The process utilizes cheap and readily available starting materials, specifically aryl ethanones and trifluoroethylimine hydrazides, which are accessible from standard chemical markets. By avoiding the necessity for strict moisture exclusion, the method drastically simplifies operational procedures, allowing for easier handling and reduced risk of batch failure. This shift represents a paradigm change in cost reduction in API manufacturing, enabling producers to achieve high purity specifications with a significantly simplified purification protocol involving basic filtration and column chromatography.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic innovation lies in a tandem sequence initiated by the iodination and Kornblum oxidation of aryl ketones. In the presence of DMSO and iodine, the aryl ethanone undergoes oxidation to form an aryl diketone intermediate. This transformation is crucial as it activates the carbonyl functionality for subsequent nucleophilic attack. Following this initial oxidation, the generated diketone reacts with trifluoroethylimine hydrazide through a dehydration condensation process to yield a hydrazone intermediate. The reaction mixture, now containing the hydrazone, is subjected to further heating in the presence of base (pyridine) and additional iodine. This environment promotes an intramolecular cyclization event that closes the triazole ring,最终 resulting in the formation of the desired 3,4,5-trisubstituted 1,2,4-triazole scaffold. The mechanistic elegance ensures that the trifluoromethyl group is retained and positioned precisely within the heterocyclic core.
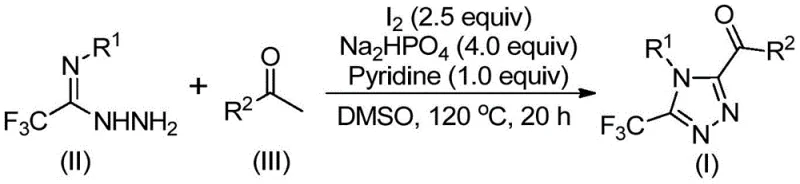
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on quality. The use of DMSO as both solvent and oxidant ensures high conversion rates of the starting ketone, minimizing the carryover of unreacted starting materials which can be difficult to separate. The specific molar ratios employed, such as the 4:1:2.5 ratio of sodium dihydrogen phosphate, pyridine, and iodine, are optimized to drive the reaction to completion while suppressing side reactions. The tolerance for various substituents on the aryl rings (R1 and R2), including alkyl, alkoxy, halogen, and trifluoromethyl groups, demonstrates the robustness of the catalytic cycle. This broad substrate scope means that a single standardized protocol can be adapted for a wide library of analogues, facilitating rapid process development and reducing the time required for route scouting in new drug discovery programs.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The procedure begins with the dissolution of aryl ethanone and iodine in DMSO, followed by a controlled heating phase to generate the reactive diketone species. Subsequent addition of the hydrazide component and buffering agents allows the cyclization to proceed smoothly under thermal conditions. While the general concept is straightforward, precise control over temperature gradients and reagent addition timing is essential to maximize yield and minimize byproduct formation. For detailed standard operating procedures and specific stoichiometric calculations tailored to your specific substrate, please refer to the standardized synthesis steps outlined below.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates directly into tangible strategic benefits regarding cost stability and supply continuity. The elimination of precious metal catalysts removes a major source of price volatility and supply risk, as iodine and DMSO are commodity chemicals with stable global availability. Furthermore, the ability to run reactions without specialized anhydrous infrastructure reduces the barrier to entry for contract manufacturing organizations, increasing the pool of potential qualified suppliers and fostering a more competitive pricing environment. This accessibility ensures that lead times for high-purity pharmaceutical intermediates can be significantly reduced, as manufacturers are not bottlenecked by complex setup requirements or scarce catalytic materials.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the substitution of expensive catalytic systems with inexpensive elemental iodine. By removing the requirement for costly transition metals and the associated ligand systems, the direct material cost per kilogram of product is substantially lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours. This efficiency drives down the overall cost of goods sold (COGS), allowing for more aggressive pricing strategies in the competitive API intermediate market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of ubiquitous starting materials like aryl ketones and hydrazides, which are produced by numerous chemical vendors worldwide. This diversification of the raw material base mitigates the risk of single-source dependency that often plagues processes relying on proprietary or niche reagents. The robustness of the reaction conditions means that production can be maintained across different geographic locations without significant re-validation, ensuring consistent supply even in the face of regional logistical disruptions or geopolitical instability affecting specific chemical sectors.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the absence of heavy metals simplifies waste stream management and reduces the regulatory burden associated with effluent treatment. The process generates less hazardous waste compared to traditional metal-catalyzed routes, aligning with increasingly strict global environmental regulations. This green chemistry profile facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates, accelerating the timeline from process development to full-scale commercial production. The method's proven scalability from gram to larger scales ensures that capacity can be ramped up quickly to meet surging market demand without compromising product quality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chain. The following questions address common concerns regarding the practical implementation and quality implications of this iodine-promoted method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers assessing the feasibility of this route for their specific projects.
Q: Does this synthesis method require expensive heavy metal catalysts?
A: No, the patented process utilizes elemental iodine as a promoter instead of toxic or expensive transition metal catalysts, significantly reducing raw material costs and simplifying waste treatment.
Q: What are the key advantages regarding reaction conditions?
A: The method operates under standard atmospheric conditions without the need for strict anhydrous or oxygen-free environments, making it highly suitable for large-scale industrial manufacturing.
Q: Is this process scalable for commercial production?
A: Yes, the patent explicitly states that the reaction can be easily expanded from gram-level laboratory synthesis to industrial scale production due to its operational simplicity and robust yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the technology described in CN113105402B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity intermediates that meet the most rigorous quality standards, utilizing our state-of-the-art facilities and rigorous QC labs to ensure every batch conforms to stringent purity specifications. Our capability to adapt this metal-free methodology allows us to offer a superior value proposition to our global partners, combining technical excellence with commercial reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
