Advanced Nickel-Catalyzed Asymmetric Nitration for High-Purity Chiral 3-Nitroindole Manufacturing
The landscape of medicinal chemistry is constantly evolving, driven by the demand for complex chiral scaffolds that can serve as potent bioactive agents. A pivotal advancement in this domain is detailed in Chinese patent CN113666862A, which discloses a highly efficient method for preparing chiral 3-nitroindole compounds via a nickel-catalyzed asymmetric nitration reaction. This technology addresses a critical gap in organic synthesis by enabling the direct construction of quaternary carbon centers bearing a nitro group with exceptional stereocontrol. For R&D directors and process chemists, this represents a transformative approach to accessing high-value pharmaceutical intermediates that were previously difficult to synthesize with high optical purity. The method leverages inexpensive nickel catalysts and readily available tert-butyl nitrite, offering a sustainable alternative to traditional noble metal catalysis.
Nitro-functionalized indoles are ubiquitous in drug discovery, serving as versatile precursors for amines, hydroxylamines, and other nitrogen-containing heterocycles. The ability to introduce a nitro group asymmetrically at the C3 position of an oxindole core opens new avenues for the synthesis of bioactive molecules targeting various therapeutic areas. By utilizing the specific catalytic system outlined in the patent, manufacturers can achieve high enantiomeric excess values, often exceeding 90% ee, which is crucial for meeting the stringent regulatory requirements of modern drug development. This technological breakthrough not only enhances the synthetic toolbox but also aligns with the industry's shift towards greener and more cost-effective manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-nitro-2-oxindoles has been plagued by significant challenges regarding selectivity and environmental impact. Prior art, such as the method disclosed in CN107200705A, relies on reacting indole-2-one derivatives with tert-butyl nitrite in 1,4-dioxane. While this approach operates under mild conditions, it fundamentally fails to induce chirality, yielding only racemic mixtures that require costly and inefficient resolution steps to separate the desired enantiomer. Furthermore, another existing method described in CN110590639A utilizes copper nitrate as both the oxidant and nitrogen source. This process is economically and environmentally disadvantageous because it requires a stoichiometric amount (1.5 equivalents) of copper salt, generating substantial heavy metal waste that complicates downstream purification and disposal. These conventional routes lack the atom economy and stereocontrol necessary for modern large-scale pharmaceutical production.
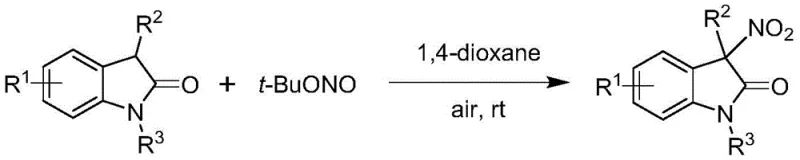
In addition to the issues of racemization and metal waste, traditional nitration methods often suffer from harsh reaction conditions that can degrade sensitive functional groups on the substrate. The lack of regioselectivity in electrophilic aromatic substitution can lead to mixtures of products, further reducing overall yield and increasing purification burdens. For procurement managers, these inefficiencies translate directly into higher raw material costs and longer lead times. The inability to directly access single-configuration products means that additional synthetic steps or chiral resolution techniques must be employed, inflating the cost of goods sold (COGS) and extending the timeline from bench to clinic. Consequently, there has been a persistent industry need for a catalytic system that combines mild conditions, high stereoselectivity, and economic viability.
The Novel Approach
The methodology presented in patent CN113666862A revolutionizes this synthetic challenge by employing a chiral nickel catalytic system. This novel approach utilizes a divalent nickel catalyst, specifically nickel chloride dimethoxyethane (NiCl₂·DME), in conjunction with a chiral Ph-PyBOX ligand, such as (S)-4-Cl-Ph-PyBox. This combination creates a highly organized chiral environment around the metal center, allowing for the precise differentiation of enantiotopic faces during the nitration event. Unlike the stoichiometric copper methods, this catalytic cycle operates with low catalyst loading (0.05-0.1 equivalents), drastically reducing the amount of metal required and minimizing waste generation. The use of tert-butyl nitrite as the nitro source remains consistent with mild protocols but is now harnessed within a stereocontrolled framework.

Furthermore, the inclusion of specific additives, such as tert-butyl alcohol, and desiccants like 4Å molecular sieves, plays a crucial role in optimizing reaction performance. The additive helps modulate the reactivity of the nitro source, while the molecular sieves ensure an anhydrous environment that is critical for maintaining catalyst integrity and high enantioselectivity. This refined protocol allows the reaction to proceed smoothly at low temperatures (0 °C to -20 °C) under air or oxygen, eliminating the need for expensive inert gas setups. For supply chain heads, this robustness translates to easier scale-up potential and reduced operational complexity. The result is a streamlined process that delivers chiral 3-nitroindoles with high yields and excellent optical purity, effectively solving the limitations of previous racemic or wasteful methods.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Nitration
The success of this transformation hinges on the intricate interplay between the nickel center and the chiral ligand. The proposed mechanism involves the coordination of the indole-2-one substrate to the nickel-ligand complex, activating the C3 position for nucleophilic attack or radical coupling, depending on the specific oxidative pathway initiated by tert-butyl nitrite and oxygen. The chiral Ph-PyBOX ligand, with its rigid bis(oxazoline) structure, imposes a specific spatial arrangement that shields one face of the reactive intermediate. This steric hindrance forces the incoming nitro group to approach from the less hindered face, thereby establishing the quaternary stereocenter with high fidelity. The electronic properties of the ligand, particularly the chloro-substituent in the preferred (S)-4-Cl-Ph-PyBox variant, further tune the Lewis acidity of the nickel center, enhancing both reaction rate and selectivity.
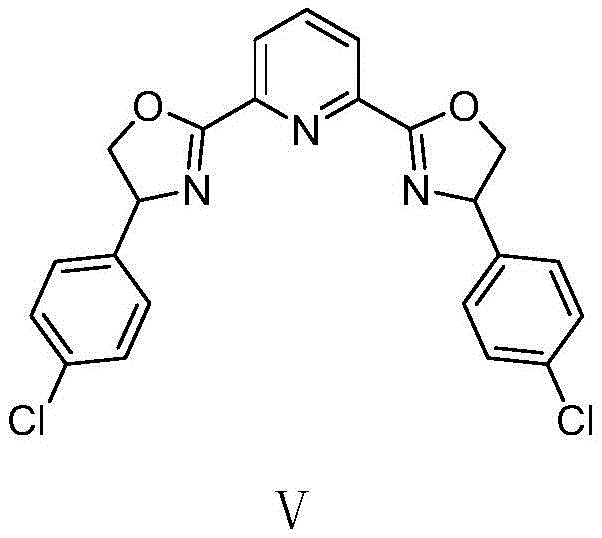
Impurity control is another critical aspect managed by this mechanistic design. In non-catalytic or poorly controlled reactions, over-nitration or oxidation of the indole ring can occur, leading to complex impurity profiles. However, the specific coordination sphere created by the nickel-ligand complex directs the reaction exclusively towards the desired C3-nitration. The mild conditions (0 °C) further suppress side reactions such as polymerization or decomposition of the sensitive nitro product. Additionally, the use of molecular sieves prevents hydrolysis of the tert-butyl nitrite or the product, ensuring a clean reaction profile. For quality control teams, this means a simpler purification process, often achievable through standard silica gel chromatography, resulting in a final product that meets stringent purity specifications required for GMP manufacturing.
How to Synthesize Chiral 3-Nitroindole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters. The general procedure involves charging a reaction vessel with the indole-2-one substrate, tert-butyl nitrite, the nickel catalyst, the chiral ligand, tert-butanol as an additive, and activated 4Å molecular sieves in a suitable solvent like dichloromethane. The mixture is then stirred under an oxygen atmosphere at controlled low temperatures. Monitoring the reaction progress via TLC is essential to determine the endpoint, typically around 12 hours. Upon completion, the workup involves simple filtration to remove the molecular sieves and catalyst residues, followed by solvent removal and column chromatography. The detailed standardized synthesis steps for specific derivatives are provided in the structured guide below.
- Charge a reaction vessel with indole-2-one substrate, tert-butyl nitrite, nickel catalyst (NiCl2·DME), chiral ligand ((S)-4-Cl-Ph-PyBox), additive (tert-butanol), and molecular sieves in dichloromethane.
- Stir the reaction mixture under an oxygen atmosphere at 0 °C for approximately 12 hours until TLC indicates complete consumption of the starting material.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography using a gradient of ethyl acetate and petroleum ether to isolate the pure chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this nickel-catalyzed technology offers substantial strategic benefits beyond mere technical feasibility. The shift from stoichiometric copper reagents to a catalytic nickel system represents a fundamental improvement in cost structure and environmental compliance. By eliminating the need for large quantities of expensive and toxic copper salts, the process significantly reduces raw material costs and waste disposal fees. This aligns perfectly with global initiatives to reduce the environmental footprint of chemical manufacturing. Moreover, the use of earth-abundant nickel instead of precious metals like palladium insulates the supply chain from the volatility of precious metal markets, ensuring more stable pricing and supply continuity for long-term projects.
- Cost Reduction in Manufacturing: The transition to a catalytic process inherently lowers the cost of goods by reducing the molar equivalent of metal reagents required. Since the nickel catalyst is used in sub-stoichiometric amounts (5-10 mol%), the material cost per kilogram of product is drastically simplified compared to methods requiring 1.5 equivalents of copper nitrate. Additionally, the avoidance of chiral resolution steps, which typically discard 50% of the material, effectively doubles the theoretical yield from the starting material, providing a massive boost in overall process efficiency and cost-effectiveness.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including tert-butyl nitrite and nickel chloride, are commodity chemicals with robust global supply chains. This contrasts with specialized chiral auxiliaries or rare metal catalysts that may face sourcing bottlenecks. The operational simplicity of running the reaction under air rather than strict inert gas conditions further reduces the dependency on specialized infrastructure, making it easier for contract manufacturing organizations (CMOs) to adopt the process quickly without extensive retrofitting, thereby securing reliable supply lines.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this method is superior. The reduction in heavy metal waste simplifies the effluent treatment process, lowering the burden on environmental health and safety (EHS) departments. The high selectivity of the reaction minimizes the formation of byproducts, leading to cleaner crude materials that require less solvent for purification. This reduction in solvent usage not only cuts costs but also aligns with green chemistry principles, making the final API or intermediate more attractive to environmentally conscious pharmaceutical partners and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed asymmetric nitration technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear understanding of the process capabilities and limitations. Understanding these nuances is vital for project managers evaluating the feasibility of integrating this route into their existing development pipelines.
Q: What is the primary advantage of using nickel catalysis over copper or palladium for this nitration?
A: Nickel catalysis offers a significantly lower cost profile compared to palladium while avoiding the heavy metal waste issues associated with stoichiometric copper nitrate methods. Furthermore, the specific nickel-chiral ligand system described in patent CN113666862A achieves high enantioselectivity (up to 94% ee), which traditional copper or uncatalyzed methods fail to provide.
Q: Can this method tolerate diverse functional groups on the indole substrate?
A: Yes, the protocol demonstrates excellent substrate scope. It successfully nitrates indole-2-ones bearing various substituents such as halogens (fluorine, chlorine), alkyl groups, alkoxy groups, and even complex heteroaryl moieties like thiophene, maintaining high yields and stereoselectivity across the board.
Q: Is the reaction sensitive to moisture or air?
A: The process is robust and operates effectively under an air or oxygen atmosphere, which simplifies operational requirements compared to strictly inert conditions. However, the inclusion of 4Å molecular sieves is critical to sequester moisture and maintain high catalytic activity and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Nitroindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the nickel-catalyzed asymmetric nitration pathway described in CN113666862A and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale optimization to multi-ton manufacturing. Our state-of-the-art facilities are designed to handle sensitive catalytic reactions with precision, maintaining stringent purity specifications and rigorous QC labs to guarantee batch-to-batch consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug discovery programs. Whether you require custom synthesis of novel 3-nitroindole derivatives or scale-up of known intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget.
