Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
Introduction to Advanced Heterocyclic Synthesis
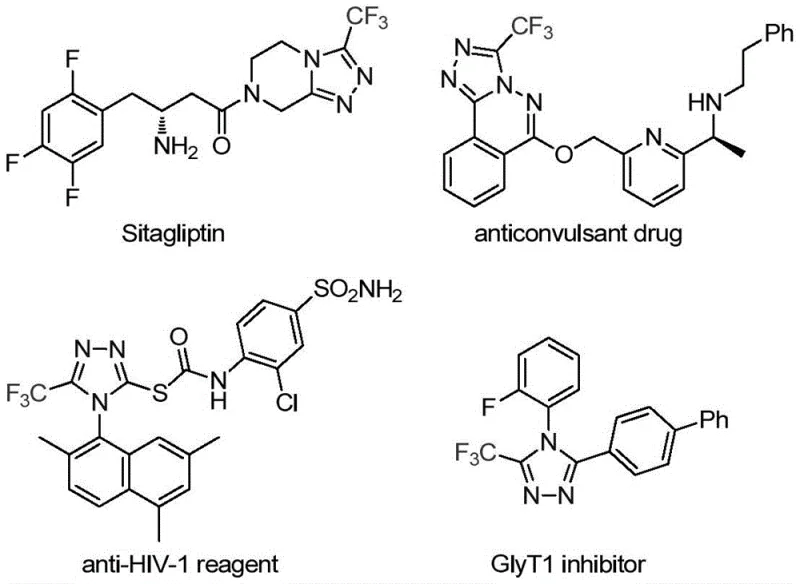
The pharmaceutical industry continuously demands robust and efficient synthetic routes for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural scaffolds are ubiquitous in modern medicinal chemistry, serving as core components in blockbuster drugs such as Sitagliptin and various antifungal agents. The strategic incorporation of the trifluoromethyl group significantly alters the physicochemical properties of the parent molecule, improving lipophilicity and electronegativity, thereby optimizing drug-receptor interactions.
This patented methodology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot cycloaddition strategy. By leveraging a unique molybdenum-copper co-catalytic system, the process achieves high reaction efficiency under remarkably mild conditions. For R&D directors and process chemists, this offers a compelling alternative to legacy routes that often suffer from harsh reaction parameters and complex purification protocols. The ability to access these valuable intermediates through a simplified workflow directly translates to accelerated lead optimization cycles and reduced time-to-market for new therapeutic candidates targeting diverse biological pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder scalability and cost-effectiveness. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical approaches often necessitate the use of unstable or hazardous precursors, such as diazonium salts and trifluorodiazoethane, which pose significant safety risks during handling and storage. Furthermore, copper-catalyzed multi-component reactions involving these volatile species frequently require stringent temperature controls and generate substantial amounts of toxic byproducts, complicating waste management and increasing the environmental footprint of the manufacturing process.
Another critical bottleneck in existing methodologies is the limited scope regarding substrate diversity. Most developed synthetic methods are optimized for fully substituted 1,2,4-triazoles, leaving a gap in the efficient synthesis of 3,4-disubstituted variants which are highly sought after in drug discovery. The reliance on expensive transition metal catalysts or specialized ligands further exacerbates the cost burden, making these routes less attractive for commercial-scale production. Additionally, the purification of the final products from complex reaction mixtures often requires extensive chromatographic separation, leading to significant material loss and reduced overall throughput, which is a major concern for supply chain managers aiming for consistent high-volume output.
The Novel Approach
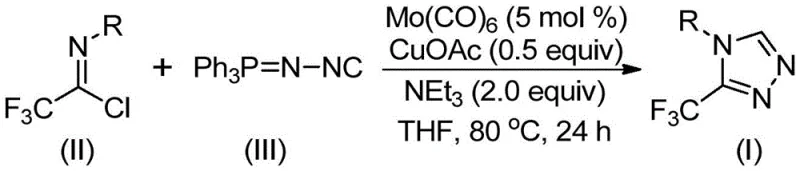
In stark contrast to these cumbersome legacy techniques, the invention described in CN113307778A introduces a highly efficient and operationally simple cycloaddition reaction. This novel approach utilizes readily available trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as starting materials, bypassing the need for dangerous diazonium intermediates. The reaction is driven by a synergistic catalytic system comprising molybdenum hexacarbonyl and cuprous acetate, which facilitates the formation of the triazole ring under温和 conditions ranging from 70°C to 90°C. This mild thermal profile not only enhances safety but also reduces energy consumption, aligning with green chemistry principles that are increasingly mandated by global regulatory bodies.
The versatility of this new method is evidenced by its broad substrate tolerance, allowing for the introduction of various functional groups including alkyl, alkoxy, halogen, and nitro substituents on the aromatic ring. This flexibility enables medicinal chemists to rapidly generate diverse libraries of analogues for structure-activity relationship (SAR) studies without redesigning the entire synthetic route. Moreover, the use of inexpensive and commercially abundant reagents significantly lowers the raw material costs, addressing a primary pain point for procurement teams. The streamlined post-treatment process, involving simple filtration and standard column chromatography, ensures high purity of the final product while minimizing solvent usage and processing time, thereby offering a distinct competitive advantage in terms of both economic and operational efficiency.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cyclization
The success of this transformation lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate a sophisticated cascade of bond-forming events. The proposed mechanism initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex. This activation step is crucial as it increases the nucleophilicity of the isonitrile carbon, priming it for subsequent attack. Simultaneously, the cuprous acetate acts as a Lewis acid promoter, coordinating with the trifluoroethylimidoyl chloride to facilitate the initial [3+2] cycloaddition. This dual-catalyst system effectively lowers the activation energy barrier for the ring-closure step, allowing the reaction to proceed smoothly at moderate temperatures where single-catalyst systems might fail or exhibit sluggish kinetics.
Following the cycloaddition, a five-membered ring intermediate is generated, which subsequently undergoes an elimination of triphenylphosphine oxide. This elimination step is driven by the presence of water within the reaction system or added molecular sieves, which help to sequester the byproduct and drive the equilibrium towards the desired 3-trifluoromethyl-substituted 1,2,4-triazole product. The precise control over stoichiometry, specifically the molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile (optimized at 1:1.5), ensures complete conversion of the limiting reagent while minimizing the formation of oligomeric side products. Understanding this mechanistic pathway allows process engineers to fine-tune reaction parameters such as catalyst loading and base concentration to maximize yield and selectivity, ensuring a robust and reproducible manufacturing process.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure optimal outcomes. The protocol involves charging a reaction vessel with the catalytic system and reagents in an aprotic solvent, followed by controlled heating. The choice of solvent is critical, with tetrahydrofuran (THF) demonstrating superior performance in dissolving reactants and promoting the catalytic cycle compared to alternatives like acetonitrile or DMF. Detailed standard operating procedures regarding reagent addition rates, temperature ramping, and quenching protocols are essential for maintaining batch-to-batch consistency.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike traditional methods that rely on custom-synthesized or hazardous intermediates, this process utilizes commodity chemicals such as trifluoroethylimidoyl chloride and functionalized isonitriles, which are readily available from multiple global suppliers. This diversification of the supplier base mitigates the risk of supply disruptions and provides leverage in price negotiations, ensuring a stable and cost-effective flow of materials essential for continuous manufacturing operations.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the elimination of expensive noble metal catalysts often used in similar transformations. By utilizing earth-abundant metals like copper and molybdenum, the direct material costs are significantly lowered. Furthermore, the mild reaction conditions reduce the energy load required for heating and cooling, contributing to lower utility bills. The high atom economy and reduced generation of hazardous waste also translate to decreased disposal costs, making the overall cost of goods sold (COGS) much more competitive compared to legacy processes.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures high reliability in production scheduling. The tolerance for various functional groups means that the same core process can be adapted for different derivatives without requiring entirely new process development campaigns. This flexibility allows manufacturers to respond quickly to changing market demands for specific API intermediates. Additionally, the scalability of the reaction from gram to kilogram levels, as demonstrated in the patent data, assures supply chain planners that the technology can seamlessly transition from R&D to commercial production without unforeseen bottlenecks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The avoidance of explosive diazonium salts and the use of less toxic solvents align with strict environmental regulations. The simplified workup procedure reduces the volume of organic solvents required for purification, minimizing the facility's volatile organic compound (VOC) emissions. This compliance ease not only avoids potential regulatory fines but also enhances the company's sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the practical application of this synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing pipelines.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, using Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, nitro, and naphthyl substituents with yields ranging from moderate to excellent.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states the method can be expanded to gram-level reactions and utilizes cheap, commercially available starting materials, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the Mo/Cu co-catalyzed synthesis described in CN113307778A and is prepared to offer this technology as part of our comprehensive CDMO services. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move from benchtop discovery to full-scale manufacturing without interruption. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development programs. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in fine chemical synthesis can accelerate your timeline and optimize your budget for bringing life-saving medicines to market.
