Advanced Axial Chiral Phosphine Catalysts: Scalable Synthesis and Commercial Applications for Fine Chemical Manufacturing
Introduction to Novel Axial Chiral Catalysis Technology
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for more efficient and selective chiral catalysts. Patent CN111777637B introduces a groundbreaking class of axial chiral oxindole-derived styrene phosphine catalysts, represented by Formula 6. This technology addresses a significant gap in the prior art, where axial chiral phosphine catalysts were predominantly restricted to binaphthyl skeletons. By leveraging a unique oxindole-styrene framework, this invention provides a robust platform for catalyzing asymmetric [4+2] and [3+2] cyclization reactions. The strategic design of these molecules allows for precise control over stereochemistry, offering a valuable tool for the construction of complex chiral architectures found in high-value pharmaceutical intermediates and fine chemicals.
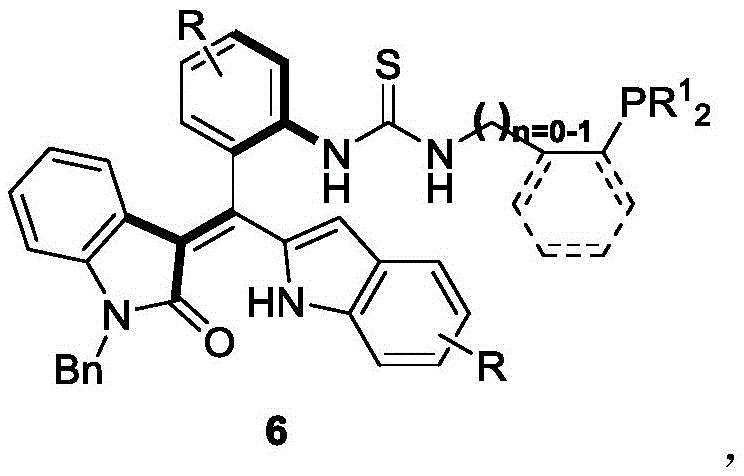
For procurement specialists and supply chain managers, the emergence of such specialized catalysts represents a shift towards more reliable fine chemical intermediates supplier networks. The ability to access novel catalytic scaffolds that are not dependent on scarce natural products or complex multi-step resolutions enhances supply chain resilience. Furthermore, the synthetic methodology described in the patent utilizes standard organic synthesis techniques, suggesting a pathway for cost reduction in fine chemical manufacturing through streamlined production processes. As the industry seeks to diversify its catalytic portfolio, understanding the technical nuances and commercial implications of this patent is crucial for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of chiral phosphine catalysis has been heavily dominated by catalysts possessing central chirality or those based on the ubiquitous binaphthyl axial chiral skeleton. While effective for many transformations, these conventional systems often face limitations in substrate scope and stereoselectivity when applied to sterically demanding or electronically distinct substrates. The reliance on binaphthyl backbones means that the steric environment is somewhat fixed, limiting the tunability required for optimizing difficult asymmetric transformations. Additionally, the synthesis of some traditional chiral phosphines can involve harsh conditions or expensive resolving agents, which complicates the supply chain and increases the overall cost of goods. These factors collectively hinder the widespread adoption of asymmetric catalysis in large-scale industrial settings where cost and consistency are paramount.
The Novel Approach
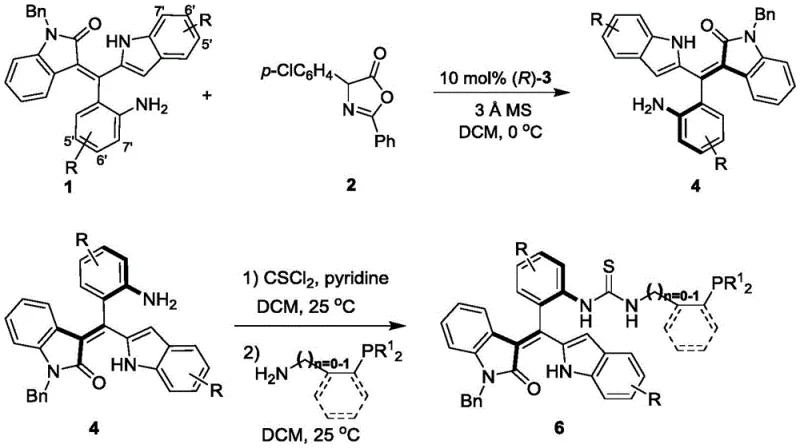
The approach detailed in patent CN111777637B diverges significantly from tradition by introducing an oxindole-derived styrene backbone. This structural innovation creates a distinct chiral environment that complements existing catalyst families. The synthesis begins with a highly enantioselective construction of the axially chiral styrene intermediate (Formula 4) using a chiral phosphoric acid catalyst at a mild temperature of 0°C. This step achieves excellent enantiomeric excess values, often exceeding 95% ee, ensuring high optical purity from the outset. Subsequent functionalization to install the phosphine moiety proceeds under ambient conditions (25°C), demonstrating the robustness of the molecular framework. This novel route not only expands the chemical space available for catalysis but also offers a more accessible entry point for producing high-purity OLED material precursors and pharmaceutical building blocks.
Mechanistic Insights into Asymmetric Cyclization Catalysis
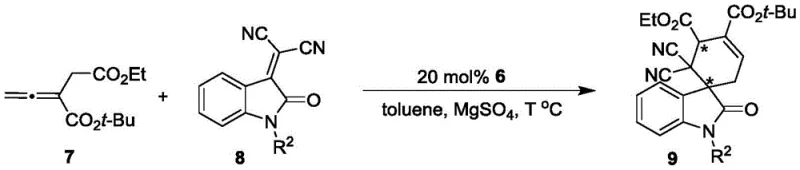
The utility of the Formula 6 catalyst lies in its ability to facilitate complex bond-forming reactions with high levels of stereocontrol. In the asymmetric [4+2] cyclization of allenoates with isatin-derived olefins, the catalyst likely operates through a nucleophilic activation mechanism. The phosphine center attacks the allenoate to generate a zwitterionic intermediate, which is then held in a specific chiral pocket defined by the rigid oxindole-styrene backbone. This spatial arrangement directs the approach of the electrophilic olefin, ensuring that the new stereocenters are formed with the desired configuration. The moderate to good yields observed in the patent examples, coupled with the formation of complex spirocyclic frameworks, highlight the catalyst's potential for rapid complexity generation in drug discovery campaigns.
Furthermore, the catalyst demonstrates versatility by also promoting asymmetric [3+2] cyclizations between MBH esters and ninhydrin-derived olefins. In these reactions, the control of diastereoselectivity is particularly noteworthy, with ratios often exceeding 95:5. The mechanism likely involves the formation of a 1,3-dipole equivalent which undergoes cycloaddition. The rigid axial chirality of the catalyst prevents unfavorable rotational conformers, thereby minimizing the formation of unwanted byproducts and simplifying downstream purification. This level of impurity control is critical for meeting the stringent purity specifications required in the production of active pharmaceutical ingredients, reducing the burden on quality control laboratories and ensuring batch-to-batch consistency.
How to Synthesize Axial Chiral Phosphine Catalyst Efficiently
The preparation of these advanced catalysts follows a logical and scalable three-step sequence that avoids the need for cryogenic conditions or exotic reagents. The process begins with the assembly of the chiral backbone, followed by activation and coupling. This streamlined workflow is designed to maximize atom economy and minimize waste generation, aligning with modern green chemistry principles. The detailed standardized synthesis steps for producing Formula 6 with high optical purity are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale operations.
- React Formula 1 and Formula 2 compounds in dichloromethane with 3Å molecular sieves and 10 mol% chiral phosphoric acid catalyst at 0°C to obtain axially chiral styrene intermediate Formula 4.
- Treat Formula 4 with thiophosgene (CSCl2) and pyridine in dichloromethane at 25°C to form the isothiocyanate intermediate.
- React the intermediate with aminophosphine compounds (Formula 5) in dichloromethane at 25°C to yield the final Formula 6 phosphine catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers several strategic benefits beyond mere technical performance. The synthesis protocol relies on readily available starting materials such as substituted anilines and isatin derivatives, which are commodities in the fine chemical sector. This availability mitigates the risk of supply disruptions often associated with specialized chiral pool reagents. Moreover, the reaction conditions are exceptionally mild, typically ranging from 0°C to 25°C, which reduces energy consumption and lowers the operational expenditure associated with heating or cooling large reactors. These factors combine to create a more resilient and cost-effective supply chain for high-value chiral intermediates.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts in the final application phase, as the organophosphine catalyst itself is metal-free. This removal of heavy metals significantly simplifies the purification process, as there is no need for costly metal scavenging resins or extensive washing protocols to meet residual metal limits. Consequently, the overall processing time is reduced, and the yield of the final API intermediate is preserved, leading to substantial cost savings in the manufacturing budget without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the catalyst synthesis, which tolerates various substituents on the aromatic rings, allows for flexibility in raw material sourcing. If a specific substituted aniline becomes unavailable, alternative analogs can often be substituted without redesigning the entire catalytic system. This modularity ensures continuity of supply even in volatile market conditions. Additionally, the stability of the final phosphine catalyst under ambient storage conditions reduces the logistical complexities and costs associated with cold chain transportation, making it a reliable choice for global distribution networks.
- Scalability and Environmental Compliance: The use of common solvents like dichloromethane and toluene, combined with ambient pressure reactions, facilitates easy scale-up from gram to kilogram scales. The process generates minimal hazardous waste compared to traditional methods involving stoichiometric chiral auxiliaries. The high atom economy of the cyclization reactions further contributes to a reduced environmental footprint. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral catalyst technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for integration into their R&D pipelines or production facilities.
Q: What distinguishes this axial chiral catalyst from traditional binaphthyl phosphine catalysts?
A: Unlike traditional biaryl axial chiral catalysts limited to binaphthyl skeletons, this invention utilizes a novel oxindole-derived styrene backbone. This unique structural motif offers different steric and electronic environments, potentially expanding the substrate scope for asymmetric [4+2] and [3+2] cyclization reactions beyond what conventional catalysts can achieve.
Q: What are the typical reaction conditions for synthesizing the Formula 6 catalyst?
A: The synthesis operates under mild conditions. The initial asymmetric construction of the backbone occurs at 0°C in dichloromethane using a chiral phosphoric acid. The subsequent functionalization steps proceed at room temperature (25°C), avoiding extreme thermal stress and facilitating easier scale-up compared to high-temperature protocols.
Q: Can this catalyst be used for large-scale pharmaceutical intermediate production?
A: Yes, the process utilizes commercially available starting materials and standard solvents like dichloromethane and toluene. The absence of transition metals in the catalyst structure itself simplifies purification and regulatory compliance for pharmaceutical applications, making it highly suitable for commercial scale-up of complex fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of novel catalytic technologies like the axial chiral oxindole-derived styrene phosphine catalysts described in CN111777637B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the synthesis of complex chiral intermediates with stringent purity specifications, supported by rigorous QC labs that ensure every batch meets the highest international standards. We are committed to bridging the gap between academic innovation and industrial application, providing our clients with access to cutting-edge chemical solutions.
We invite you to collaborate with us to explore the full potential of this catalyst system for your specific synthetic challenges. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain not just a supplier, but a strategic ally dedicated to accelerating your time-to-market and optimizing your manufacturing efficiency for high-purity pharmaceutical intermediates.
