Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
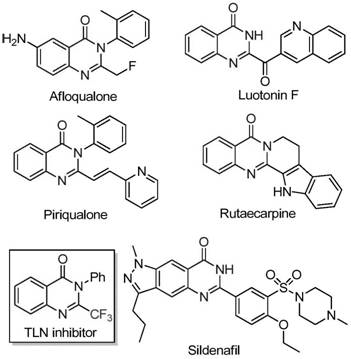
The landscape of modern pharmaceutical development is increasingly defined by the incorporation of fluorine atoms into heterocyclic scaffolds to enhance metabolic stability and bioavailability. Within this domain, quinazolinone derivatives stand out as privileged structures found in numerous bioactive natural products and synthetic drugs, exhibiting potent anti-cancer, anticonvulsant, and anti-inflammatory properties. As illustrated in the structural diversity of known bioactive molecules, the strategic introduction of a trifluoromethyl group can drastically alter the physicochemical profile of these core structures. A groundbreaking preparation method disclosed in patent CN111675662B offers a transformative approach to accessing these valuable 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages a cost-effective iron-catalyzed cyclization strategy that bypasses the limitations of traditional noble metal catalysis, presenting a compelling opportunity for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionality has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. While chemically feasible, these conventional pathways are frequently plagued by severe operational drawbacks that hinder their utility in large-scale commercial settings. The reaction conditions often require harsh temperatures or aggressive reagents that limit functional group tolerance, leading to significant decomposition of sensitive substrates. Furthermore, the reliance on expensive fluorinating agents and the generation of stoichiometric amounts of acidic waste streams create substantial environmental and economic burdens. These factors collectively result in narrow substrate scopes and inconsistent yields, making the supply chain for high-purity OLED material or API precursors vulnerable to disruption and cost volatility.
The Novel Approach
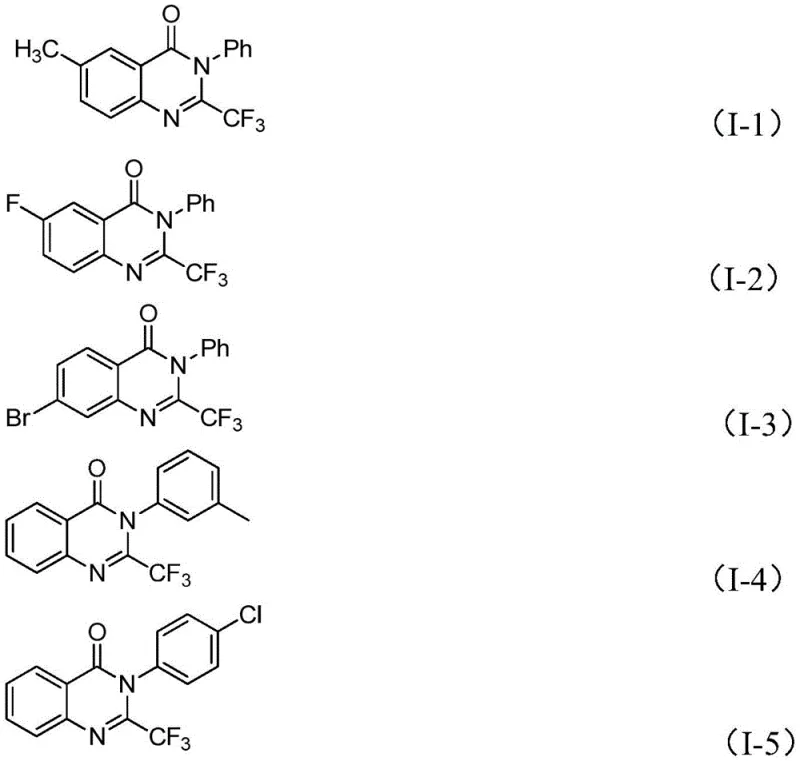
In stark contrast, the novel methodology utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as the foundational building blocks for constructing the quinazolinone core. This innovative route, catalyzed by inexpensive ferric chloride, operates under relatively mild conditions while delivering exceptional yields across a broad range of substrates. As demonstrated by the successful synthesis of derivatives I-1 through I-5, the process accommodates diverse electronic environments, including electron-donating methyl groups and electron-withdrawing halogens, without compromising efficiency. This robustness allows for the rapid generation of compound libraries for drug discovery programs, ensuring that cost reduction in electronic chemical manufacturing or pharma intermediate production is achieved through streamlined chemistry rather than compromised quality.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
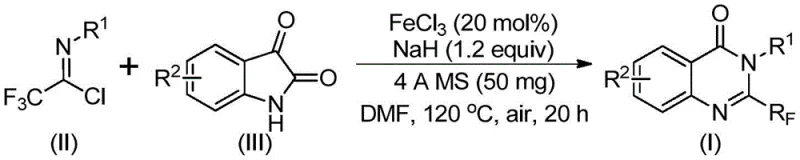
The mechanistic elegance of this transformation lies in the dual role of the reaction components in facilitating bond formation and subsequent rearrangement. Initially, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate, generating a transient trifluoroacetamidine intermediate. This step is critical for setting up the requisite geometry for the subsequent cyclization event. Following this initial coupling, the iron catalyst mediates a decarbonylation and cyclization sequence that effectively closes the heterocyclic ring. The use of 4Å molecular sieves plays a pivotal role in sequestering moisture, thereby protecting the sensitive imidoyl chloride species from hydrolysis and driving the equilibrium toward product formation. This careful orchestration of reagents ensures that the reaction proceeds with high atom economy and minimal side-product formation.
From an impurity control perspective, the choice of ferric chloride as a Lewis acid catalyst offers distinct advantages over stronger or more oxophilic metals. The moderate Lewis acidity prevents over-reaction or polymerization of the sensitive isatin starting material, which is a common failure mode in alternative synthetic routes. Furthermore, the reaction conditions allow for the tolerance of halogen substituents, such as bromine and chlorine, which are often susceptible to dehalogenation under more vigorous catalytic regimes. This high fidelity in preserving functional groups means that downstream purification is significantly simplified, reducing the burden on process chemistry teams to remove complex byproduct mixtures. The result is a cleaner crude reaction profile that translates directly into higher isolated yields and superior purity specifications for the final active pharmaceutical ingredient.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory-scale optimization and pilot plant campaigns. The procedure involves a straightforward one-pot sequence where all reagents are combined in a polar aprotic solvent, typically DMF, which effectively solubilizes both the organic substrates and the inorganic base. The reaction initiates at a lower temperature to facilitate the initial coupling before ramping up to drive the cyclization to completion. For detailed standard operating procedures and safety guidelines regarding the handling of sodium hydride and trifluoroethylimidoyl chloride, please refer to the technical documentation below.
- Combine ferric chloride, sodium hydride, 4Å molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- Stir the mixture at 40°C for approximately 10 hours to initiate the reaction, then increase the temperature to 120°C.
- Maintain the reaction at 120°C for an additional 20 hours under air, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-catalyzed methodology represents a strategic shift towards more resilient and cost-effective sourcing models. By eliminating the dependency on scarce and price-volatile precious metals, manufacturers can stabilize their raw material costs and mitigate the risks associated with geopolitical supply disruptions. The use of commodity chemicals like isatin and simple aromatic amines ensures a steady supply of starting materials, while the robust nature of the reaction reduces the likelihood of batch failures that can delay project timelines. This reliability is essential for maintaining the continuity of supply for critical therapeutic areas where quinazolinone scaffolds are prevalent.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with ferric chloride results in a drastic reduction in catalyst costs, which is particularly impactful when scaling to multi-kilogram or ton-level production. Additionally, the elimination of complex ligand systems and the use of simple workup procedures, such as filtration and standard column chromatography, significantly lower the operational expenditure associated with purification. This streamlined process flow reduces solvent consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing footprint that aligns with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: The reliance on widely available and commercially stable starting materials ensures that production schedules are not held hostage by the lead times of specialized reagents. Isatin and its derivatives are produced globally in large quantities, providing a buffer against regional shortages. Furthermore, the reaction's tolerance to air and moisture, facilitated by the use of molecular sieves rather than strictly anhydrous conditions, simplifies the engineering requirements for the reactor setup. This flexibility allows for faster turnaround times between batches and enhances the overall agility of the supply chain in responding to market demands.
- Scalability and Environmental Compliance: The protocol is explicitly designed with scalability in mind, having been validated from milligram to gram scales with consistent performance. The absence of toxic heavy metals simplifies the regulatory pathway for drug approval, as residual metal limits are easier to meet with iron than with palladium or platinum. Moreover, the reduced generation of hazardous waste and the potential for solvent recycling make this process environmentally compliant with stringent international regulations. This alignment with sustainability goals not only reduces liability but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these nuances is crucial for R&D teams evaluating route selection and for procurement specialists assessing vendor qualifications.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts?
A: Using ferric chloride significantly reduces raw material costs compared to palladium or rhodium catalysts, eliminates the need for expensive heavy metal scavenging steps, and simplifies the regulatory compliance profile for pharmaceutical intermediates.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level and provides strong potential for industrial scale application due to the use of cheap, commercially available starting materials and robust reaction conditions.
Q: What is the substrate scope for this quinazolinone synthesis?
A: The method exhibits excellent functional group tolerance, successfully accommodating various substituents on the aryl ring including methyl, fluoro, bromo, chloro, methoxy, and nitro groups, allowing for the design of diverse molecular scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch of 2-trifluoromethyl quinazolinone delivered adheres to the highest industry standards.
We invite you to collaborate with us to leverage this advanced iron-catalyzed technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that will demonstrate how our optimized processes can accelerate your drug development timeline while maximizing value.
