Advanced Palladium-Catalyzed Synthesis of Fluorine-Containing Alkylthio Olefin Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce fluorine atoms into organic scaffolds, given the profound impact fluorination has on metabolic stability and bioavailability. Patent CN114369045B addresses this critical need by disclosing a novel synthesis method for fluorine-containing alkylthio substituted olefin derivatives. This technology leverages a palladium-catalyzed nucleophilic substitution strategy, utilizing readily available alkenyl sulfonium salts and fluoride salts to construct complex fluorinated architectures. The general structure of the target molecules, as depicted in the patent, showcases a versatile framework where R groups can be varied to include hydrogen, methyl, aryl, or acetyl functionalities, providing a broad chemical space for drug discovery.
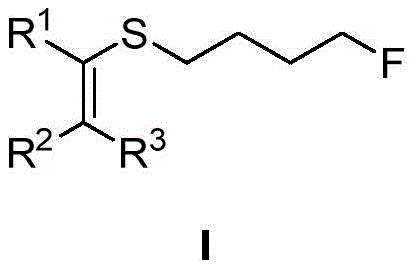
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such transformations is vital for scaling production. The disclosed method not only offers high reaction efficiency but also ensures excellent stereoselectivity, which is paramount when synthesizing chiral drug candidates. By employing mild reaction conditions and avoiding hazardous reagents often associated with traditional fluorination techniques, this process represents a significant leap forward in green chemistry practices for the manufacture of high-purity OLED material precursors and active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of fluorine-containing alkylthio groups onto olefinic backbones has been fraught with synthetic challenges. Conventional routes often rely on harsh reaction conditions, such as extremely low temperatures or the use of highly toxic and corrosive fluorinating agents that pose significant safety risks in a commercial manufacturing environment. Furthermore, traditional methods frequently suffer from poor regioselectivity and limited substrate scope, failing to accommodate complex functional groups commonly found in advanced drug molecules. These limitations often result in low yields, difficult purification processes, and the generation of substantial chemical waste, thereby inflating the cost reduction in electronic chemical manufacturing and pharma sectors. The inability to precisely control the stereochemistry of the double bond in these conventional processes further complicates downstream processing, leading to mixtures of isomers that are difficult to separate.
The Novel Approach
In stark contrast, the methodology described in CN114369045B utilizes a sophisticated palladium-catalyzed system that operates under significantly milder conditions. The core innovation lies in the use of alkenyl sulfonium salts as key intermediates, which act as superior electrophiles for nucleophilic attack by fluoride ions. As illustrated in the reaction scheme below, the transformation proceeds smoothly in solvents like 1,4-dioxane at moderate temperatures ranging from 60-120°C.

This novel approach eliminates the need for cryogenic conditions and dangerous reagents, streamlining the operational workflow. The use of commercially available fluoride salts, such as Cesium Fluoride (CsF), coupled with inexpensive palladium promoters like Pd(OAc)2, drastically simplifies the supply chain logistics. Moreover, the reaction exhibits remarkable functional group tolerance, allowing for the synthesis of derivatives with diverse substituents including halogens, nitro groups, and heterocycles, thus enabling the commercial scale-up of complex polymer additives and pharmaceutical intermediates without extensive protecting group strategies.
Mechanistic Insights into Palladium-Catalyzed Nucleophilic Substitution
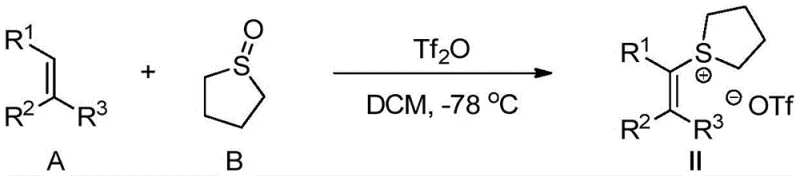
The success of this synthesis hinges on the unique reactivity of the alkenyl sulfonium salt intermediate. Mechanistically, the palladium catalyst likely coordinates with the sulfur moiety or the olefinic system to activate the carbon-sulfur bond towards nucleophilic displacement. The sulfonium group serves as an excellent leaving group, facilitating the attack of the fluoride ion to form the stable carbon-fluorine bond. This pathway avoids the high energy barriers typically associated with direct C-H fluorination. The preparation of the crucial alkenyl sulfonium salt precursor itself is achieved through the reaction of an olefin with sulfolane and trifluoromethanesulfonic anhydride at low temperatures, ensuring the formation of the reactive species with high fidelity.
From an impurity control perspective, the mild nature of the reaction conditions plays a pivotal role. By avoiding extreme thermal stress or highly acidic/basic environments, the formation of side products such as polymerization byproducts or elimination products is minimized. The specific choice of solvent, particularly aprotic non-polar solvents like 1,4-dioxane, further enhances the solubility of the ionic fluoride species while stabilizing the transition state of the catalytic cycle. This precise control over the reaction environment ensures that the final product maintains high purity levels, reducing the burden on downstream purification units and aligning with the stringent quality requirements of a reliable agrochemical intermediate supplier.
How to Synthesize Fluorine-Containing Alkylthio Olefin Derivatives Efficiently
The practical implementation of this technology involves a straightforward two-stage process that is amenable to standard laboratory and pilot plant equipment. First, the alkenyl sulfonium salt is prepared and isolated or used in situ, followed by the palladium-catalyzed fluorination step. The detailed protocol emphasizes the importance of stoichiometry, specifically recommending a molar ratio of alkenyl sulfonium salt to fluoride salt of approximately 1:2.0 to drive the reaction to completion. For R&D teams looking to replicate this chemistry, the following guide outlines the standardized operational parameters derived from the patent examples.
- Preparation of Alkenyl Sulfonium Salt: React olefin A with sulfolane B and trifluoromethanesulfonic anhydride in DCM at -78°C to generate the key intermediate alkenyl sulfonium salt II.
- Palladium-Catalyzed Fluorination: Combine the alkenyl sulfonium salt II with a fluoride salt (preferably CsF) and a palladium catalyst (such as Pd(OAc)2) in 1,4-dioxane solvent.
- Reaction and Isolation: Heat the mixture to 80-100°C for 5-12 hours under inert atmosphere, then purify the resulting fluorine-containing olefin derivative I via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift towards this palladium-catalyzed method addresses several critical pain points in the sourcing of fluorinated building blocks. By utilizing commodity chemicals as starting materials, manufacturers can mitigate the risks associated with volatile raw material markets and ensure a steady flow of production inputs. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or safety incidents related to hazardous reagent handling.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of relatively inexpensive catalysts and fluoride sources. Unlike proprietary fluorinating reagents that command premium prices, salts like CsF and KF are widely available in bulk quantities. Furthermore, the elimination of complex protection-deprotection sequences and the high atom economy of the substitution reaction contribute to substantial cost savings. The ability to run the reaction at atmospheric pressure and moderate temperatures also reduces energy consumption, leading to a lower overall cost of goods sold (COGS) for the final fluorinated intermediates.
- Enhanced Supply Chain Reliability: Dependence on exotic or tightly controlled reagents often creates bottlenecks in the supply chain. This method relies on a palette of chemicals that are standard inventory items for most fine chemical distributors. The simplicity of the workup procedure, which typically involves standard extraction and chromatography, allows for faster turnaround times from synthesis to shipment. This agility enables suppliers to respond more rapidly to fluctuating demand from downstream pharmaceutical clients, ensuring continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often reveals hidden inefficiencies, but this protocol is designed with scalability in mind. The use of 1,4-dioxane and other common solvents facilitates easy solvent recovery and recycling, aligning with modern environmental, health, and safety (EHS) standards. The reduced generation of hazardous waste streams simplifies disposal compliance and lowers the environmental footprint of the manufacturing site. This makes the process attractive for large-scale production facilities aiming to meet rigorous sustainability goals while maintaining high output volumes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope and operational details. These insights are drawn directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process chemists.
Q: What are the optimal reaction conditions for this fluorination process?
A: The patent specifies that the reaction proceeds most efficiently using Cesium Fluoride (CsF) as the fluoride source and Palladium Acetate (Pd(OAc)2) as the catalyst in 1,4-dioxane solvent at temperatures between 80-100°C.
Q: Can this method tolerate diverse functional groups on the olefin substrate?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substrates with aryl, heteroaryl, acetyl, and benzoyl groups, making it suitable for complex pharmaceutical intermediate synthesis.
Q: What is the significance of the alkenyl sulfonium salt intermediate?
A: The alkenyl sulfonium salt serves as a highly reactive electrophile that facilitates the nucleophilic attack by the fluoride ion under mild palladium catalysis, overcoming the kinetic barriers often associated with direct fluorination of unactivated olefins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Alkylthio Substituted Olefin Derivative Supplier
The technological advancements detailed in CN114369045B represent a powerful tool for the next generation of fluorinated therapeutics and functional materials. At NINGBO INNO PHARMCHEM, we recognize the potential of this chemistry to revolutionize the production of high-value intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of fluorine-containing olefin derivatives meets the exacting standards required by global regulatory bodies.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall development costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate our commitment to being your trusted partner in advanced chemical manufacturing.
