Advanced Pd-Catalyzed Synthesis of Fluorine-Containing Olefin Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The rapid evolution of medicinal chemistry has placed fluorine-containing organic compounds at the forefront of new drug discovery, driven by their unique ability to enhance metabolic stability and bioavailability. Patent CN114369045B, published in October 2022, introduces a groundbreaking methodology for the synthesis of fluorine-containing alkylthio substituted olefin derivatives, addressing a critical gap in the efficient construction of these valuable scaffolds. This technology leverages the structural diversity of alkenyl sulfonium salts to achieve selective fluorination under mild conditions, offering a robust pathway for generating high-purity pharmaceutical intermediates. The innovation lies in the strategic use of a palladium catalyst to promote nucleophilic substitution, transforming readily available starting materials into complex fluorinated architectures with exceptional efficiency. For global R&D teams seeking reliable pharmaceutical intermediates suppliers, this patent represents a significant leap forward in accessing diverse fluorinated building blocks that were previously difficult or costly to synthesize.
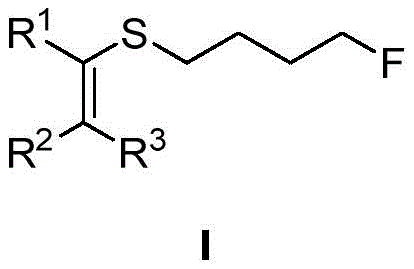
The significance of this invention extends beyond mere academic interest; it provides a practical solution for the industrial manufacturing of fluorine-rich molecules used in agrochemicals and electronic materials. By enabling the introduction of fluorine atoms into olefinic systems with high stereoselectivity, the method supports the development of next-generation active pharmaceutical ingredients (APIs). The versatility of the substituents R1, R2, and R3 allows chemists to tailor the electronic and steric properties of the final molecule, facilitating the optimization of drug candidates. As the demand for fluorinated compounds continues to surge in the global market, mastering such efficient synthetic routes becomes a strategic imperative for any organization aiming to maintain a competitive edge in fine chemical intermediates production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fluorine-containing olefin derivatives has been plagued by significant challenges related to reagent toxicity, cost, and operational complexity. Conventional fluorination strategies often rely on hazardous reagents such as diethylaminosulfur trifluoride (DAST) or Deoxo-Fluor, which are not only expensive but also pose severe safety risks due to their thermal instability and potential for explosive decomposition. Furthermore, these reagents frequently require stringent anhydrous conditions and cryogenic temperatures, complicating the scale-up process and increasing the overall cost of goods sold (COGS). Another major drawback of older methodologies is the limited functional group tolerance, which often necessitates extensive protecting group strategies, thereby elongating the synthetic sequence and reducing overall atom economy. For procurement managers focused on cost reduction in fluorine-containing olefin derivatives manufacturing, these inefficiencies translate into higher raw material expenditures and increased waste disposal costs, creating a substantial burden on the supply chain.
The Novel Approach
In stark contrast, the novel approach disclosed in CN114369045B utilizes alkenyl sulfonium salts as versatile synthons, enabling a streamlined and safer fluorination process. By employing a palladium catalyst to facilitate the nucleophilic substitution of the sulfonium group with a fluoride ion, this method operates under significantly milder conditions, typically between 60-120°C, eliminating the need for cryogenic setups. The use of inexpensive and commercially available fluoride salts, such as cesium fluoride (CsF), drastically reduces the reliance on proprietary fluorinating agents, thereby enhancing the economic viability of the process. This strategy not only simplifies the operational workflow but also improves the safety profile of the manufacturing environment, making it highly attractive for large-scale industrial applications. The ability to tolerate a wide range of functional groups without degradation ensures that complex molecular architectures can be constructed efficiently, supporting the rapid iteration required in modern drug discovery pipelines.

Mechanistic Insights into Palladium-Catalyzed Nucleophilic Substitution
The core of this innovative synthesis lies in the palladium-catalyzed activation of the alkenyl sulfonium salt, which serves as an excellent leaving group for the subsequent nucleophilic attack by the fluoride ion. Mechanistically, the palladium species, preferably palladium acetate (Pd(OAc)2), coordinates with the electron-rich olefinic system or facilitates the dissociation of the sulfonium moiety, thereby lowering the activation energy for the substitution reaction. This catalytic cycle allows for the efficient displacement of the tetrahydrothiophene group by the fluoride anion, resulting in the formation of the carbon-fluorine bond with high regioselectivity. The choice of solvent plays a crucial role in this mechanism; aprotic polar solvents like 1,4-dioxane are preferred as they stabilize the ionic intermediates and enhance the solubility of the inorganic fluoride salt without interfering with the catalytic cycle. Understanding these mechanistic nuances is vital for R&D directors aiming to optimize reaction parameters for specific substrates, ensuring maximum yield and minimal impurity formation during the synthesis of high-purity OLED material or pharmaceutical precursors.
Furthermore, the reaction demonstrates remarkable tolerance to various substituents on the olefinic backbone, including aryl, alkyl, and heteroaryl groups, which is attributed to the robustness of the palladium catalytic system. The stereoselectivity observed in the products suggests that the reaction proceeds through a well-defined transition state that preserves the geometric integrity of the double bond, a critical feature for maintaining the biological activity of the final drug candidate. Impurity control is inherently managed by the specificity of the catalyst and the mild reaction conditions, which minimize side reactions such as polymerization or elimination that are common in harsher fluorination protocols. This level of control over the reaction outcome ensures that the resulting fluorine-containing alkylthio substituted olefin derivatives meet the stringent purity specifications required for clinical applications, reducing the need for extensive downstream purification steps.
How to Synthesize Fluorine-Containing Alkylthio Substituted Olefin Derivatives Efficiently
To implement this cutting-edge technology in a laboratory or pilot plant setting, the process begins with the preparation of the key alkenyl sulfonium salt precursor, which can be synthesized from readily available olefins and sulfolane derivatives using trifluoromethanesulfonic anhydride. Once the precursor is secured, the fluorination step is executed by combining the sulfonium salt with a fluoride source, such as CsF, in the presence of a palladium promoter within a suitable reaction vessel. The detailed standardized synthesis steps for this procedure are outlined in the guide below, providing a clear roadmap for technical teams to replicate the high yields reported in the patent literature. Adhering to these optimized conditions ensures consistent product quality and maximizes the efficiency of the transformation, making it an ideal candidate for technology transfer and process validation.
- Prepare the alkenyl sulfonium salt precursor (II) by reacting an olefin with sulfolane and trifluoromethanesulfonic anhydride in DCM at -78°C.
- Combine the alkenyl sulfonium salt (II), a fluoride salt (preferably CsF), and a palladium catalyst (preferably Pd(OAc)2) in an aprotic solvent like 1,4-dioxane.
- Heat the reaction mixture to 80-100°C under an inert atmosphere for 5-12 hours to facilitate the nucleophilic substitution, followed by standard purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed fluorination method offers transformative benefits for procurement and supply chain operations, primarily driven by the simplification of raw material sourcing and process safety. The shift away from exotic, high-cost fluorinating reagents to commodity chemicals like cesium fluoride and palladium acetate significantly lowers the barrier to entry for producing these valuable intermediates. This change in reagent profile not only reduces direct material costs but also mitigates supply chain risks associated with the availability of specialized fluorinating agents, which are often subject to market volatility and strict regulatory controls. For supply chain heads, this translates into enhanced supply chain reliability and the ability to secure long-term contracts for raw materials at stable prices, ensuring uninterrupted production schedules for critical pharmaceutical projects.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous fluorinating reagents directly impacts the bottom line by reducing the cost of goods sold. By utilizing relatively non-toxic and inexpensive palladium salts as promoters, the process avoids the need for costly heavy metal removal steps that are often required with other transition metal catalysts, further streamlining the purification workflow. Additionally, the high reaction efficiency and yields reported, reaching up to 90% in optimized examples, mean that less raw material is wasted, maximizing the output per batch and improving overall resource utilization. These factors collectively contribute to substantial cost savings in fluorine-containing olefin derivatives manufacturing, making the final intermediates more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to disruptions. Fluoride salts and palladium catalysts are widely produced and stocked by multiple global suppliers, reducing the risk of single-source dependency. Furthermore, the mild reaction conditions reduce the wear and tear on manufacturing equipment and lower the energy consumption required for heating and cooling, contributing to a more sustainable and reliable production infrastructure. This stability is crucial for meeting the tight delivery timelines demanded by multinational pharmaceutical companies, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality rather than just a goal.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of common solvents like 1,4-dioxane facilitate easy scale-up from gram to ton quantities without significant process redesign. The reduced toxicity of the reagents compared to traditional fluorination methods aligns with increasingly stringent environmental regulations, minimizing the generation of hazardous waste and simplifying disposal protocols. This environmental compliance is a key factor for modern chemical manufacturers who must adhere to green chemistry principles while maintaining high production volumes. The ability to scale this process efficiently supports the commercial scale-up of complex polymer additives and fine chemicals, positioning manufacturers to capture emerging market opportunities in advanced materials and life sciences.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology, based on the detailed data provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for your specific manufacturing needs. By understanding the nuances of the reaction conditions and substrate scope, decision-makers can better evaluate the fit of this technology within their existing production portfolios.
Q: What are the key advantages of using alkenyl sulfonium salts for fluorination compared to traditional methods?
A: The use of alkenyl sulfonium salts allows for mild reaction conditions and avoids the need for hazardous, expensive fluorinating reagents like DAST. The method utilizes commercially available fluoride salts such as CsF, significantly reducing raw material costs and improving operational safety.
Q: What is the typical yield and purity profile for this Pd-catalyzed fluorination process?
A: According to patent data, the reaction yields range from 45% to 90%, with optimal conditions achieving up to 90% yield. The products exhibit good stereoselectivity and functional group tolerance, making them suitable for high-purity pharmaceutical intermediate applications.
Q: Is this synthesis method scalable for industrial production of fluorine-containing intermediates?
A: Yes, the method is designed for scalability. It uses relatively non-toxic palladium promoters and common solvents like 1,4-dioxane. The simple operation and high efficiency support the commercial scale-up of complex fluorine-containing olefin derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Olefin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods disclosed in CN114369045B and are fully equipped to leverage this technology for our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorine-containing intermediates meets the highest international standards, providing our partners with the confidence they need to advance their drug development programs. Our commitment to quality and consistency makes us a trusted partner for companies seeking reliable pharmaceutical intermediates suppliers who can deliver on complex technical requirements.
We invite you to engage with our technical procurement team to discuss how this advanced fluorination strategy can be integrated into your supply chain to drive innovation and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project volume and timeline. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your journey from concept to commercial success with precision and reliability.
