Advanced Carbonylation Strategy for Scalable Thioester Production in Pharma Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharma Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, where efficiency, safety, and cost-effectiveness are paramount. A significant breakthrough in this domain is detailed in patent CN113004181B, which discloses a novel method for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. Thioesters are critical structural motifs serving as stable acyl donors in the synthesis of complex aldehydes, ketones, esters, and amides, and they play indispensable roles in biological processes such as natural chemical ligation. However, traditional synthetic routes often rely on hazardous reagents that complicate scale-up. This new methodology addresses these challenges by utilizing inexpensive and readily available benzyl chloride compounds and sulfonyl chlorides as starting materials, offering a streamlined pathway for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation of thiols with carboxylic acids or their derivatives, a process fraught with significant operational and safety drawbacks. Thiols are notoriously malodorous, possessing a repulsive smell that poses severe challenges for worker safety and environmental compliance in industrial settings. Furthermore, sulfur-containing compounds like thiols are potent catalyst poisons; they can irreversibly bind to transition metal centers, deactivating the catalyst and leading to poor reaction yields or requiring excessive catalyst loading. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from limited substrate scope or require harsh reaction conditions that are incompatible with sensitive functional groups commonly found in drug molecules. These limitations necessitate a paradigm shift towards safer, more robust synthetic strategies that can be reliably implemented in commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The innovative approach described in the patent circumvents these traditional pitfalls by employing a palladium-catalyzed carbonylation reaction that utilizes sulfonyl chlorides as the sulfur source instead of thiols. This strategic substitution is transformative, as sulfonyl chlorides are odorless, stable, and commercially abundant, effectively eliminating the safety hazards associated with volatile thiols. The reaction proceeds efficiently under relatively mild conditions, typically involving heating at 100°C in an organic solvent like acetonitrile. Crucially, the system employs tungsten carbonyl, which acts dually as the carbonyl source and the reducing agent, simplifying the reagent profile by removing the need for additional external reducers. This method exhibits exceptional functional group tolerance, accommodating various substituents including halogens, alkyl, and alkoxy groups, thereby providing a versatile platform for synthesizing diverse thioester derivatives required in medicinal chemistry.
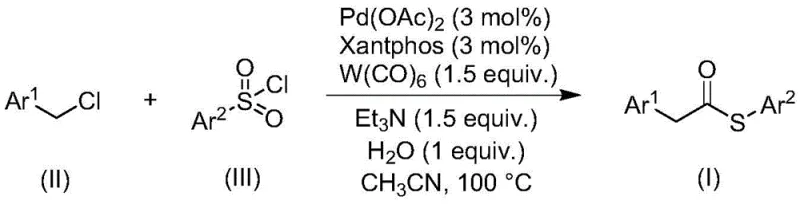
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this synthetic advancement lies in the sophisticated interplay between the palladium catalyst, the Xantphos ligand, and the tungsten carbonyl reagent. The reaction initiates with the oxidative addition of the benzyl chloride to the palladium(0) species generated in situ, forming a benzyl-palladium(II) intermediate. The presence of the bulky and electron-rich Xantphos ligand stabilizes this intermediate and facilitates the subsequent insertion of carbon monoxide, which is released from the tungsten carbonyl precursor. This step forms an acyl-palladium complex, a key transient species in the catalytic cycle. Unlike traditional carbonylations that require high-pressure CO gas, this system generates CO internally, enhancing safety and operational simplicity. The sulfonyl chloride then interacts with the acyl-palladium species, likely undergoing a reduction and sulfur transfer process mediated by the tungsten species, ultimately releasing the thioester product and regenerating the active palladium catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes. In conventional methods, thiols are prone to oxidation, leading to disulfide byproducts that are difficult to separate and can contaminate the final API intermediate. By avoiding free thiols entirely, the carbonylation method significantly reduces the formation of sulfur-based oxidative impurities. Additionally, the use of a well-defined palladium/Xantphos system ensures high chemoselectivity, minimizing side reactions such as homocoupling of the benzyl chloride or hydrolysis of the acid chloride intermediates. The result is a cleaner reaction profile that simplifies downstream purification, allowing for the isolation of products with stringent purity specifications through standard workup procedures like filtration and column chromatography, which is vital for maintaining quality control in regulated industries.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and reproducibility. The process begins by combining the benzyl chloride substrate and the sulfonyl chloride derivative with the catalytic system in a suitable organic solvent. The choice of solvent is critical; while several aprotic solvents were screened, acetonitrile was identified as the optimal medium due to its ability to effectively dissolve all reagents and promote high conversion rates. The reaction mixture is then heated to 100°C and maintained for approximately 24 hours. This duration strikes a balance between ensuring complete consumption of the starting materials and minimizing energy costs associated with prolonged heating. Following the reaction, the workup involves simple filtration to remove metal residues, followed by purification techniques common in the field to isolate the target thioester. For a detailed breakdown of the standardized synthesis steps, please refer to the guide below.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten carbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 24 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this carbonylation technology presents compelling economic and logistical benefits that directly impact the bottom line. The primary driver for cost reduction is the substitution of expensive and hazardous thiols with cheap, commodity-grade sulfonyl chlorides and benzyl chlorides. These starting materials are widely available in the global chemical market, ensuring a stable supply chain that is less susceptible to the volatility often seen with specialized sulfur reagents. Furthermore, the elimination of malodorous thiols drastically simplifies facility requirements; manufacturers do not need specialized scrubbing systems or containment protocols for noxious gases, leading to substantial capital expenditure savings and lower operational overheads. This translates into a more competitive pricing structure for the final thioester intermediates without compromising on quality or performance.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing tungsten carbonyl as a dual-purpose reagent, serving as both the carbonyl source and the reducing agent. This innovation removes the necessity for purchasing and handling separate reducing agents, thereby streamlining the bill of materials. Additionally, the high reaction efficiency and broad substrate tolerance mean that fewer batches are rejected due to poor conversion, maximizing raw material utilization. The simplified post-treatment process, which avoids complex extraction steps often needed to remove thiol odors, further reduces labor and utility costs, contributing to overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: Reliance on benzyl chlorides and sulfonyl chlorides enhances supply chain resilience because these are high-volume industrial chemicals produced by numerous suppliers worldwide. This diversity of supply sources mitigates the risk of shortages that can occur with niche reagents. Moreover, the stability of these starting materials allows for longer storage times and easier transportation compared to sensitive thiols or pressurized carbon monoxide cylinders. This robustness ensures consistent production schedules and reliable delivery timelines for downstream customers, which is critical for maintaining continuous pharmaceutical manufacturing operations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this method is vastly superior to traditional routes. The absence of volatile, toxic thiols reduces the environmental footprint of the manufacturing process and lowers the regulatory burden associated with waste disposal. The reaction operates at atmospheric pressure regarding CO gas (since it is generated in situ), eliminating the safety risks of high-pressure reactors. These factors make the process highly scalable from kilogram to multi-ton production levels, facilitating the commercial scale-up of complex pharmaceutical intermediates while adhering to increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries based on the patent data. These questions address the practical aspects of the reaction conditions, substrate compatibility, and purification requirements. Understanding these details is essential for R&D directors planning process development and for procurement teams assessing the feasibility of integrating this new intermediate into their supply chain. The following insights provide a clear picture of the operational parameters and the versatility of this synthetic method.
Q: What is the primary advantage of using sulfonyl chloride over thiols in this synthesis?
A: Using sulfonyl chloride eliminates the need for malodorous and toxic thiol compounds, which often poison catalysts and create significant safety and environmental hazards in large-scale manufacturing.
Q: What role does tungsten carbonyl play in the reaction mechanism?
A: Tungsten carbonyl serves a dual function as both the carbonyl source (providing the CO group) and the reducing agent, thereby simplifying the reagent system by removing the need for an additional external reducer.
Q: Is this method suitable for substrates with sensitive functional groups?
A: Yes, the method demonstrates wide substrate tolerance, successfully accommodating various substituents such as halogens, alkyl groups, and alkoxy groups on both the benzyl chloride and sulfonyl chloride components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value thioester intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thioester compound delivered meets the highest standards required for pharmaceutical applications. We are committed to leveraging advanced synthetic methodologies like this to drive innovation and efficiency in the global supply chain.
We invite you to collaborate with us to explore how this cost-effective synthesis route can benefit your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and economically advantageous source for your thioester intermediates, empowering your organization to accelerate drug development and optimize manufacturing costs.
