Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles, particularly those incorporating fluorine motifs that enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN116253692A, which discloses a novel preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds. These scaffolds are increasingly recognized for their potent biological activities, ranging from anticancer and antifungal properties to antihypertensive effects. The disclosed technology represents a paradigm shift from traditional transition-metal catalyzed processes to a greener, base-promoted cycloaddition strategy. By leveraging cheap and readily available starting materials like chlorohydrazones and trifluoroacetyl sulfur ylides, this method addresses critical pain points in modern medicinal chemistry, specifically the need for operationally simple, air-tolerant, and cost-effective routes to complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied heavily on condensation reactions between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical approaches are well-documented, they suffer from inherent inefficiencies that hinder large-scale application. Conventional protocols often necessitate harsh reaction conditions, including elevated temperatures and strictly anhydrous environments, which increase energy consumption and operational complexity. Furthermore, many existing methods exhibit poor structural diversity, limiting the ability of chemists to rapidly explore structure-activity relationships (SAR) around the triazine core. The reliance on specific, sometimes difficult-to-synthesize substrates further exacerbates supply chain bottlenecks, making the production of diverse trifluoromethyl-substituted derivatives economically challenging for commercial manufacturers.
The Novel Approach
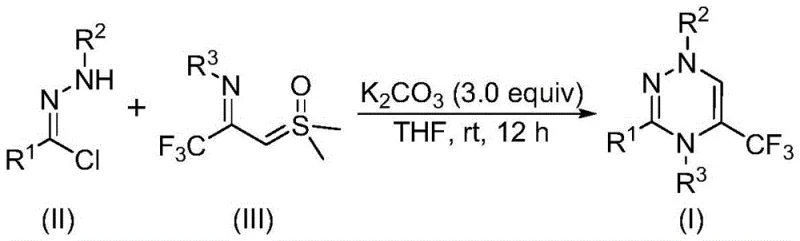
In stark contrast to legacy techniques, the methodology described in CN116253692A introduces a streamlined [3+3] cycloaddition strategy that operates under exceptionally mild conditions. The process utilizes potassium carbonate as a benign promoter in common organic solvents like tetrahydrofuran, eliminating the need for expensive ligands or toxic heavy metal catalysts. This innovation allows the reaction to proceed efficiently at room temperature (20-40°C) even in the presence of air, drastically reducing the engineering controls required for manufacturing. The versatility of this approach is demonstrated by its compatibility with a wide array of functional groups, enabling the synthesis of diverse derivatives with high yields. By simplifying the reaction setup and workup procedures, this novel route offers a compelling solution for the rapid generation of high-purity triazine intermediates.
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The mechanistic pathway of this transformation is both elegant and efficient, driven by the in situ generation of reactive intermediates under basic conditions. Initially, the chlorohydrazone precursor undergoes dehydrohalogenation promoted by potassium carbonate to generate a nitrile imine intermediate. This highly reactive 1,3-dipole then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl sulfur ylide. The reaction proceeds through a concerted mechanism that constructs the six-membered triazine ring while simultaneously eliminating a molecule of dimethyl sulfoxide (DMSO). Alternatively, the process may occur via a stepwise sequence involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution. This dual-pathway flexibility ensures high conversion rates and minimizes the formation of polymeric byproducts, which is a common issue in dipolar cycloadditions.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product quality. Traditional methods often lead to decomposition of sensitive functional groups or over-reaction due to high thermal energy input. In this potassium carbonate-mediated system, the ambient temperature profile preserves the integrity of labile substituents such as esters, halogens, and ethers. The absence of transition metals also precludes the formation of metal-complexed impurities, which are notoriously difficult to remove and can pose significant regulatory hurdles in pharmaceutical development. Consequently, the crude reaction mixture typically exhibits a cleaner profile, facilitating simpler purification workflows and higher overall recovery of the target trifluoromethyl-substituted 1,2,4-triazine.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemistry teams aiming to scale up production. The standard protocol involves charging a reactor with the chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate in a suitable aprotic solvent such as THF. The mixture is stirred at room temperature for approximately 10 to 14 hours, after which the reaction is deemed complete. Workup is straightforward, typically involving filtration to remove inorganic salts followed by standard chromatographic purification. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as tetrahydrofuran.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Filter the reaction mixture, mix the residue with silica gel, and purify via column chromatography to isolate the target trifluoromethyl-substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers substantial strategic advantages regarding cost structure and supply reliability. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk associated with the volatility of metal prices. Furthermore, the use of commodity chemicals like potassium carbonate and common solvents ensures that the raw material supply chain is robust and less susceptible to geopolitical disruptions. The ability to run the reaction under air atmosphere rather than inert gas significantly reduces utility costs and simplifies reactor requirements, translating to lower capital expenditure for manufacturing facilities.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete avoidance of expensive transition metal catalysts and specialized ligands. In traditional cross-coupling or cyclization reactions, the cost of palladium or copper catalysts, combined with the subsequent need for metal scavenging resins to meet strict ppm limits, can be prohibitive. This new method bypasses those costs entirely. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup reduces solvent usage and waste disposal fees, leading to a significantly leaner cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically chlorohydrazones and sulfur ylides, are derived from widely available bulk chemicals such as acyl chlorides, hydrazines, and trifluoroacetic acid derivatives. This reliance on commodity feedstocks ensures a stable supply base with multiple potential vendors, reducing the risk of single-source dependency. The operational robustness of the reaction, which tolerates air and moisture better than many organometallic processes, also means that production schedules are less likely to be disrupted by minor environmental fluctuations or equipment failures, ensuring consistent on-time delivery for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for expansion. The exothermic profile is manageable at room temperature, reducing the need for complex cryogenic systems or aggressive cooling jackets. From an environmental standpoint, the process generates minimal hazardous waste; the primary byproduct is dimethyl sulfoxide, which is relatively benign and easily managed compared to heavy metal sludge. This aligns perfectly with modern green chemistry principles and helps manufacturing partners maintain compliance with increasingly stringent environmental regulations without costly retrofitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the process capabilities and limitations for potential licensees or manufacturing partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process explicitly avoids the use of heavy metal catalysts. It utilizes inexpensive and non-toxic potassium carbonate as a promoter, which significantly simplifies downstream purification and eliminates the risk of heavy metal contamination in the final API intermediate.
Q: What are the reaction conditions required for this cyclization?
A: The reaction operates under remarkably mild conditions, proceeding efficiently at room temperature (20-40°C) in an air atmosphere. This eliminates the need for rigorous inert gas protection or high-energy heating, making it highly suitable for large-scale manufacturing.
Q: Is the substrate scope limited for this triazine synthesis?
A: The method demonstrates excellent structural diversity. It tolerates various substituents on the phenyl rings, including alkyl, alkoxy, halogen, nitro, and trifluoromethyl groups at ortho, meta, or para positions, allowing for the design of diverse libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in accelerating drug discovery and development pipelines. Our technical team has extensively evaluated the methodology described in CN116253692A and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from gram-scale research to industrial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl-substituted 1,2,4-triazine meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Whether you require custom synthesis of specific analogs or bulk supply of standard intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations that demonstrate how we can optimize your supply chain while maintaining uncompromised quality.
