Advanced Metal-Free Synthesis of 3-Acylquinoxalinone Derivatives for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 3-Acylquinoxalinone Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical pharmacophores in drug discovery. A significant breakthrough in this domain is detailed in Chinese Patent CN108033922B, which discloses a novel preparation method for 3-acylquinoxalinone derivatives. This technology represents a paradigm shift from traditional multi-step or metal-catalyzed processes to a streamlined, metal-free oxidative acylation strategy. By utilizing substituted quinoxalin-2-one derivatives and simple aldehydes or benzyl alcohols as starting materials, this invention achieves direct functionalization at the C3 position of the quinoxalinone core. The significance of this development cannot be overstated for R&D teams focusing on antitumor agents, antibacterial compounds, and HIV-I reverse transcriptase inhibitors, where the 3-acylquinoxalinone motif is a prevalent structural element. The method employs mass percent 70% tert-butyl hydroperoxide (TBHP) as a green oxidant, operating effectively under air conditions without the need for inert gas protection or precious metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 3-acylquinoxalinone derivatives was often plagued by inefficiencies that hindered large-scale manufacturing and increased overall production costs. Historical literature describes routes involving cyclization followed by oxidation, typically requiring two distinct reaction steps. For instance, one common approach involves the reaction of o-phenylenediamine with bromo-ketoesters to form 3-bromobenzylquinoxalinone intermediates, which subsequently undergo debromination and oxidation in DMSO. 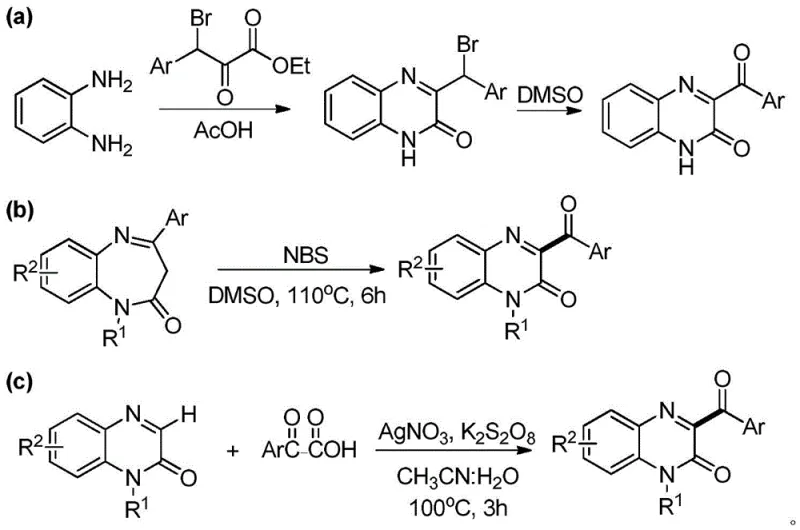 Another documented method utilizes 1,5-benzodiazepinones reacting with N-bromosuccinimide (NBS) at elevated temperatures of 110°C for extended periods, leading to poor atom economy and significant energy consumption. Furthermore, transition metal-catalyzed pathways, such as those employing silver nitrate (AgNO3) with potassium persulfate, introduce the complication of heavy metal contamination. For procurement managers and supply chain heads, these legacy methods present substantial risks, including the high cost of noble metal catalysts, the logistical burden of sourcing specialized reagents, and the stringent regulatory requirements for removing trace metals from final Active Pharmaceutical Ingredients (APIs).
Another documented method utilizes 1,5-benzodiazepinones reacting with N-bromosuccinimide (NBS) at elevated temperatures of 110°C for extended periods, leading to poor atom economy and significant energy consumption. Furthermore, transition metal-catalyzed pathways, such as those employing silver nitrate (AgNO3) with potassium persulfate, introduce the complication of heavy metal contamination. For procurement managers and supply chain heads, these legacy methods present substantial risks, including the high cost of noble metal catalysts, the logistical burden of sourcing specialized reagents, and the stringent regulatory requirements for removing trace metals from final Active Pharmaceutical Ingredients (APIs).
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the method disclosed in CN108033922B offers a direct, one-step synthesis that dramatically simplifies the operational workflow. The core innovation lies in the direct oxidative coupling of quinoxalin-2-one derivatives with aldehydes or benzyl alcohols using peroxide oxidants. 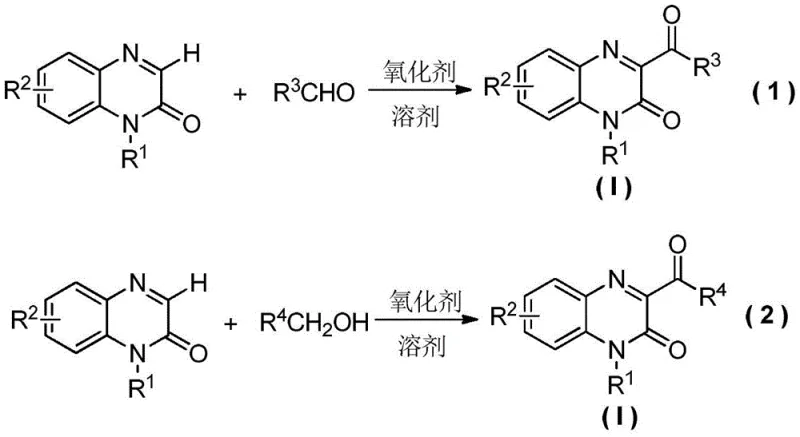 This novel approach bypasses the need for pre-functionalized halogenated intermediates or expensive transition metal catalysts entirely. The reaction proceeds smoothly in common organic solvents such as acetonitrile, acetone, or 1,2-dichloroethane at moderate temperatures ranging from 60°C to 90°C. By eliminating the multi-step purification of intermediates and avoiding the use of silver or other heavy metals, this process inherently reduces the environmental footprint and operational complexity. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates to a more resilient supply chain, as the raw materials—substituted quinoxalinones and commodity aldehydes—are readily available and cost-effective. The ability to perform the reaction under air atmosphere further lowers the barrier to entry for industrial implementation, removing the need for specialized anhydrous or anaerobic equipment.
This novel approach bypasses the need for pre-functionalized halogenated intermediates or expensive transition metal catalysts entirely. The reaction proceeds smoothly in common organic solvents such as acetonitrile, acetone, or 1,2-dichloroethane at moderate temperatures ranging from 60°C to 90°C. By eliminating the multi-step purification of intermediates and avoiding the use of silver or other heavy metals, this process inherently reduces the environmental footprint and operational complexity. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates to a more resilient supply chain, as the raw materials—substituted quinoxalinones and commodity aldehydes—are readily available and cost-effective. The ability to perform the reaction under air atmosphere further lowers the barrier to entry for industrial implementation, removing the need for specialized anhydrous or anaerobic equipment.
Mechanistic Insights into Metal-Free Radical Oxidative Acylation
Understanding the underlying chemical mechanism is crucial for R&D directors evaluating the feasibility and impurity profile of this synthesis. The reaction operates via a free radical pathway initiated by the thermal decomposition of the peroxide oxidant, such as tert-butyl hydroperoxide (TBHP). Upon heating, TBHP homolytically cleaves to generate tert-butoxy radicals and hydroxyl radicals, which serve as the primary initiators for the catalytic cycle. The benzaldehyde substrate reacts with the tert-butoxy radical to abstract a hydrogen atom, forming a reactive benzoyl radical species while releasing tert-butanol as a byproduct. This benzoyl radical then acts as a nucleophilic radical attacker, selectively targeting the electron-deficient C3 position of the quinoxalin-2-one ring system. 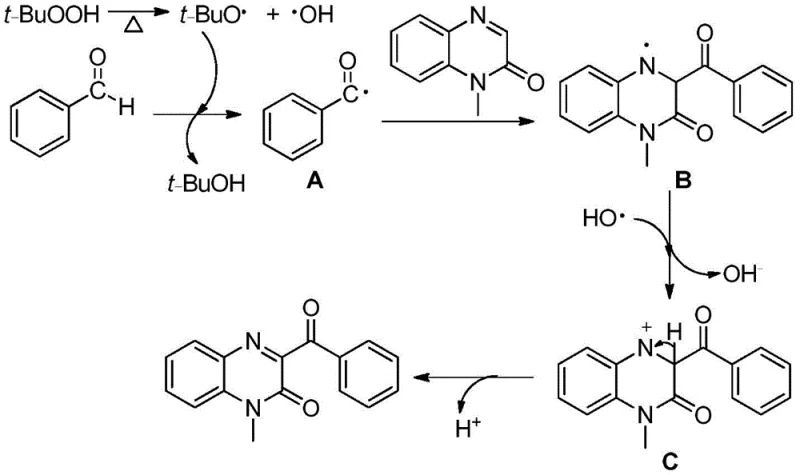 The resulting intermediate is an N-centered radical, which subsequently undergoes single-electron transfer (SET) oxidation to form a cationic intermediate. Finally, the loss of a proton from this cation restores aromaticity and yields the desired 3-acylquinoxalinone product. This mechanistic clarity is vital for impurity control; since the reaction avoids metal coordination complexes, the impurity profile is dominated by organic byproducts rather than inorganic metal residues, simplifying downstream purification.
The resulting intermediate is an N-centered radical, which subsequently undergoes single-electron transfer (SET) oxidation to form a cationic intermediate. Finally, the loss of a proton from this cation restores aromaticity and yields the desired 3-acylquinoxalinone product. This mechanistic clarity is vital for impurity control; since the reaction avoids metal coordination complexes, the impurity profile is dominated by organic byproducts rather than inorganic metal residues, simplifying downstream purification.
From a process chemistry perspective, the radical nature of this transformation ensures high regioselectivity for the C3 position, minimizing the formation of isomeric byproducts that often complicate chromatographic separation. The use of aqueous TBHP (70%) provides a controlled release of radicals, preventing runaway exotherms that can occur with more volatile initiators. Furthermore, the tolerance of the reaction to various substituents on the quinoxalinone ring (such as methyl, chloro, or methoxy groups) and the versatility in accepting both aromatic and aliphatic aldehydes demonstrate the robustness of this radical manifold. For quality control teams, this predictability means that method validation is more straightforward, as the reaction does not suffer from the batch-to-batch variability often associated with heterogeneous metal catalysis. The mechanism also explains why benzyl alcohols can serve as effective precursors; they are likely oxidized in situ to the corresponding aldehydes or acyl radicals under the strong oxidative conditions, expanding the scope of accessible chemical space for medicinal chemists.
How to Synthesize 3-Acylquinoxalinone Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The standard protocol involves dissolving the substituted quinoxalin-2-one derivative and the aldehyde or benzyl alcohol substrate in a selected solvent, followed by the addition of the oxidant. The mixture is then heated to the specified temperature range, typically between 60°C and 90°C, and stirred for a duration of 5.0 to 9.0 hours. Workup procedures are conventional, involving solvent removal under reduced pressure and extraction with ethyl acetate, followed by washing with brine and drying over anhydrous sodium sulfate. The crude product is usually purified via column chromatography using standard eluent systems like ethyl acetate and petroleum ether.
- Mix substituted quinoxalin-2-one derivative with aldehyde or benzyl alcohol in a suitable solvent such as 1,2-dichloroethane or acetonitrile.
- Add an oxidant such as 70% aqueous TBHP, DTBP, or TBPB to the reaction mixture under air atmosphere.
- Heat the reaction mixture to 60-90°C for 5.0-9.0 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic advantages that directly impact the bottom line and operational reliability. The most significant benefit is the drastic simplification of the supply chain regarding raw material sourcing. By eliminating the dependency on transition metal catalysts like silver nitrate or specialized brominated reagents, manufacturers can mitigate the risks associated with price volatility and supply shortages of these niche chemicals. Moreover, the removal of heavy metals from the process flow means that the costly and time-consuming steps required for metal scavenging and residual metal testing are no longer necessary. This reduction in processing steps leads to substantial cost savings in terms of both consumables and labor hours, enhancing the overall economic viability of producing high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as noble metals are subject to significant market fluctuations and high unit prices. Additionally, the one-step nature of the reaction reduces solvent consumption and energy usage compared to multi-step legacy methods that require isolation and purification of intermediates. The avoidance of specialized reagents like N-bromosuccinimide further lowers the input costs, allowing for a more competitive pricing structure for the final API intermediate. This efficiency gain is compounded by the higher atom economy of the direct oxidative coupling, which minimizes waste generation and the associated disposal costs.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as benzaldehyde derivatives and aqueous TBHP ensures a stable and continuous supply of raw materials, as these are produced globally in large volumes. The robustness of the reaction conditions, which tolerate air and moisture to a certain extent, reduces the risk of batch failures due to environmental factors, thereby improving delivery consistency. For supply chain planners, this reliability translates to reduced safety stock requirements and shorter lead times for high-purity pharmaceutical intermediates, enabling a more agile response to market demand fluctuations.
- Scalability and Environmental Compliance: The mild reaction temperatures (60-90°C) and the use of non-hazardous solvents make this process inherently safer and easier to scale from kilogram to tonnage production. The absence of heavy metal waste streams simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge and hazardous waste management. This green chemistry profile not only reduces the regulatory burden but also aligns with the sustainability goals of modern pharmaceutical companies, potentially facilitating faster regulatory approval for new drug applications that utilize this manufacturing route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this metal-free synthesis over traditional methods?
A: This method eliminates the need for expensive transition metal catalysts like silver salts, thereby removing the requirement for complex heavy metal removal steps and reducing environmental waste.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction operates under mild conditions (60-90°C) in air atmosphere using commercially available oxidants, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the substrate scope for this acylation reaction?
A: The method demonstrates broad applicability, tolerating various substituents on the quinoxalinone ring and accepting both aromatic aldehydes and benzyl alcohols as acyl sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylquinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis described in CN108033922B for the production of high-value quinoxalinone derivatives. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-acylquinoxalinone intermediate adheres to the highest industry standards. We understand that transitioning to a new synthetic route requires confidence in the supplier's technical capability, and our team is prepared to validate this metal-free process to meet your specific project timelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your project scale. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence. Let us collaborate to bring your next-generation pharmaceutical candidates to market faster and more efficiently through our commitment to innovation and quality excellence.
