Advanced Photocatalytic Synthesis of 3-Acylquinoxalinones for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds that serve as the backbone for numerous bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN112409275B, which discloses a novel preparation method for 3-acylquinoxalinone compounds utilizing visible light photocatalysis. Quinoxalinones are critically important molecular skeletons in natural products and pharmaceuticals, exhibiting a wide spectrum of biological activities including antidepressant, antiviral, antibacterial, and protein kinase inhibitory properties. Many established drugs, such as panapron and opaverine, rely on this structural motif. The innovation presented in this patent addresses long-standing challenges in the functionalization of the quinoxalinone C3-H bond by employing a metal-free organic photocatalyst system. By leveraging simple and readily available aldehydes as acylating agents under mild visible light irradiation, this technology offers a transformative approach to generating high-purity pharmaceutical intermediates. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for evaluating its potential to streamline supply chains and reduce manufacturing costs in the production of advanced therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
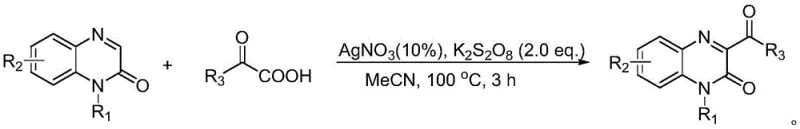
Historically, the acylation of quinoxalinones at the C3 position has been achieved through various methodologies, each carrying significant drawbacks that hinder large-scale commercial adoption. For instance, earlier research by Zeng's group in 2017 utilized silver nitrate (AgNO3) as a catalyst and potassium persulfate as an oxidant in acetonitrile at a high temperature of 100 °C for 3 hours. While effective, this method relies on expensive transition metals and requires energy-intensive heating conditions, which complicates process safety and waste management. Furthermore, it necessitates the use of benzoylformic acid derivatives as substrates, which are often more costly and less stable than their corresponding aldehydes. Similarly, Yuan's group reported a method in 2018 using tert-butyl hydroperoxide (TBHP) as an oxidant in dichloroethane at 70 °C. Although this avoided silver, it still required elevated temperatures and potentially hazardous peroxide reagents. Another approach by Bao's group in 2019 employed Acridine Red as a photosensitizer but still relied on benzoylformic acid原料 and required 8 hours of reaction time. These conventional routes collectively suffer from harsh reaction conditions, the necessity for toxic or precious metal reagents, and the reliance on less atom-economical keto-acid starting materials, creating bottlenecks in cost reduction and environmental compliance for industrial manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN112409275B introduces a remarkably mild and green synthetic strategy. The core innovation lies in the use of Eosin Y disodium salt as an organic photocatalyst combined with a persulfate oxidant under visible light irradiation at room temperature. This method fundamentally shifts the paradigm by enabling the direct use of simple aldehydes as the acyl source, bypassing the need for pre-oxidized keto acids. The reaction proceeds efficiently under nitrogen protection in common organic solvents like acetonitrile, with reaction times ranging from 16 to 24 hours under 20W blue LED light. This approach not only eliminates the contamination risks associated with heavy metal catalysts but also drastically reduces energy consumption by operating at ambient temperature. The versatility of the system is further enhanced by the compatibility with various organic dye photocatalysts such as Methylene Blue and Rose Bengal, providing flexibility in process optimization. By replacing harsh thermal conditions and toxic metals with benign visible light and organic dyes, this novel approach offers a superior pathway for the sustainable manufacturing of 3-acylquinoxalinone derivatives.

Mechanistic Insights into Eosin Y-Catalyzed C3 Acylation
The mechanistic elegance of this photocatalytic system provides R&D teams with a robust framework for understanding impurity profiles and reaction kinetics. The process initiates when the Eosin Y disodium salt photocatalyst absorbs visible light photons, transitioning from its ground state to an excited state. This excited photocatalyst then engages in a single electron transfer (SET) process with the persulfate oxidant, generating sulfate radical anions. These highly reactive radicals abstract a hydrogen atom from the aldehyde substrate, thereby generating an acyl radical species. This acyl radical subsequently attacks the electron-deficient C3 position of the quinoxalinone ring, forming a transient radical intermediate. The final step involves the oxidation of this intermediate by the oxidant system, followed by deprotonation to restore aromaticity and yield the desired 3-acylquinoxalinone product. This radical-mediated pathway is distinct from traditional ionic mechanisms, offering unique selectivity patterns that can be exploited to minimize side reactions. The mild nature of the radical generation ensures that sensitive functional groups on the aldehyde or quinoxalinone substrates remain intact, leading to cleaner reaction profiles and simplified downstream purification processes.
From an impurity control perspective, the absence of transition metals is a critical advantage. Traditional silver-catalyzed routes often leave trace metal residues that require rigorous and costly removal steps to meet stringent pharmaceutical purity specifications. In this photocatalytic method, the catalyst is an organic dye that can be easily separated or degraded, significantly reducing the burden on quality control laboratories. Furthermore, the use of room temperature conditions minimizes thermal degradation pathways that often lead to complex byproduct mixtures in high-temperature reactions. The stoichiometry can be finely tuned, with the patent suggesting a molar ratio of quinoxalinone to aldehyde between 1:1.5 and 1:5.0, and catalyst loading as low as 0.0001 to 0.05 equivalents. This precise control over reaction parameters allows for the optimization of yield while suppressing the formation of over-acylated or polymerized impurities. For process chemists, this mechanistic clarity translates to a more predictable and controllable manufacturing process, ensuring consistent batch-to-batch quality essential for regulatory compliance.
How to Synthesize 3-Acylquinoxalinone Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and yield. The general procedure involves charging a reaction vessel with the quinoxalinone substrate, the selected aldehyde, the Eosin Y disodium salt photocatalyst, and the ammonium persulfate oxidant. After purging the system with nitrogen to remove oxygen, an organic solvent such as acetonitrile is added, and the mixture is subjected to irradiation from a 20W blue LED lamp. Reaction monitoring is typically conducted via TLC or HPLC to determine the endpoint, usually achieved within 16 to 24 hours. Upon completion, the reaction mixture undergoes standard workup procedures involving extraction with ethyl acetate and water, followed by concentration and purification via silica gel column chromatography. Detailed standardized synthesis steps for specific derivatives are outlined below to guide technical teams in replicating these high-yielding results.
- Mix quinoxalinone substrate, aldehyde, Eosin Y disodium salt photocatalyst, and ammonium persulfate oxidant in a reaction vessel under nitrogen protection.
- Add organic solvent such as acetonitrile and irradiate the mixture with a 20W blue LED light source at room temperature for 16 to 24 hours.
- Monitor reaction progress via TLC, then perform extraction with ethyl acetate and water, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology presents compelling economic and logistical advantages over traditional synthesis routes. The shift from expensive transition metal catalysts to abundant organic dyes directly impacts the bill of materials, removing the volatility associated with precious metal pricing. Moreover, the ability to utilize simple aldehydes instead of specialized keto acids simplifies the raw material supply chain, as aldehydes are commodity chemicals available from a vast network of global suppliers. This diversification of supply sources enhances supply chain resilience, reducing the risk of disruptions caused by the scarcity of niche reagents. The mild reaction conditions also translate to lower utility costs, as there is no need for energy-intensive heating systems or specialized high-pressure equipment. These factors collectively contribute to a more sustainable and cost-efficient manufacturing model that aligns with modern green chemistry principles and corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of silver nitrate and other transition metal catalysts removes the significant cost burden associated with purchasing, recovering, and disposing of heavy metals. Additionally, the substitution of costly benzoylformic acid derivatives with inexpensive and widely available aldehydes drastically lowers the raw material input costs. The mild room temperature operation further reduces energy expenditures related to heating and cooling, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates. The simplified purification process, necessitated by the absence of metal residues, also reduces solvent consumption and labor time during the isolation of the final product.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like aldehydes and common organic solvents, manufacturers can mitigate the risks associated with sourcing specialized reagents that may have limited suppliers or long lead times. The robustness of the photocatalytic system allows for flexibility in raw material selection, as various aldehydes with different electronic properties can be accommodated without major process changes. This flexibility ensures continuous production capabilities even when specific raw material markets experience fluctuations. Furthermore, the stability of the organic photocatalysts ensures consistent performance over time, reducing the variability in production schedules and enhancing the reliability of delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green nature of this synthesis method, characterized by the absence of toxic metals and the use of visible light energy, positions it favorably for regulatory approval and environmental compliance. Scaling up photochemical reactions has historically been challenging, but advancements in flow chemistry and LED reactor design now make this feasible for commercial production. The reduced generation of hazardous waste streams simplifies wastewater treatment and disposal, lowering the environmental footprint of the manufacturing facility. This alignment with strict environmental regulations facilitates smoother permitting processes and reduces the liability associated with hazardous chemical handling, making it an attractive option for long-term capacity expansion in the fine chemical sector.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications provided in patent CN112409275B, offering clarity on catalyst selection, reaction conditions, and substrate scope. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing workflows. The responses highlight the versatility of the method across different substrates and its robustness under varying conditions, providing confidence in its applicability for diverse pharmaceutical intermediate projects.
Q: What are the primary advantages of this photocatalytic method over traditional silver-catalyzed routes?
A: This method eliminates the need for toxic and expensive transition metal catalysts like silver nitrate, operates at room temperature instead of high heat (100°C), and utilizes cheaper, more readily available aldehyde substrates instead of complex keto acids.
Q: Which photocatalysts are compatible with this synthesis protocol?
A: The patent specifies several organic dyes including Eosin Y disodium salt, Eosin Y sodium salt, Eosin B, Methylene Blue, Acridine Red, Rose Bengal, and Rose Bengal B, with Eosin Y disodium salt being the preferred embodiment.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (room temperature, visible light) and the use of non-toxic organic catalysts significantly enhance safety and environmental compliance, making it highly scalable for industrial manufacturing compared to harsh thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylquinoxalinone Supplier
The technological advancements described in patent CN112409275B underscore the immense potential of visible-light photocatalysis in modern organic synthesis, particularly for the production of complex heterocyclic intermediates. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent technologies into robust commercial realities. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry approach are realized at an industrial level. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs designed to meet the exacting standards of the global pharmaceutical industry. We understand that the transition from bench-scale discovery to commercial supply requires not just chemical expertise but also deep process engineering capabilities, which our team delivers with precision.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this metal-free photocatalytic method can optimize your specific supply chain economics. We encourage you to contact our technical procurement team to request specific COA data for relevant intermediates and comprehensive route feasibility assessments. Let us help you navigate the complexities of modern chemical manufacturing, ensuring a reliable supply of high-quality 3-acylquinoxalinone derivatives that drive your drug development programs forward efficiently and sustainably.
