Advanced Photocatalytic Synthesis of 3-Acylquinoxalinone Intermediates for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access bioactive heterocyclic scaffolds. Patent CN112409275B, published in October 2022, introduces a groundbreaking preparation method for 3-acylquinoxalinone compounds that leverages visible light photocatalysis to overcome significant limitations of prior art. Quinoxalinone derivatives are critical molecular backbones found in numerous therapeutic agents, exhibiting potent biological activities ranging from antidepressant and antiviral properties to protein kinase inhibition. The innovation disclosed in this patent utilizes eosin Y disodium salt as an organic photocatalyst and persulfate as an oxidant to facilitate the direct C3-acylation of quinoxalinones using simple, commercially available aldehydes. This approach represents a paradigm shift from traditional thermal or metal-catalyzed processes, offering a greener, safer, and potentially more cost-effective route for the production of high-purity pharmaceutical intermediates. By operating under mild room temperature conditions with visible light irradiation, this technology aligns perfectly with modern green chemistry principles while maintaining high synthetic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
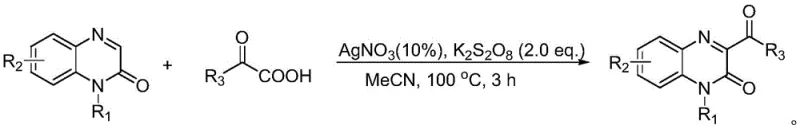
Historically, the functionalization of the quinoxalinone core at the C3 position has relied heavily on methods that pose significant challenges for large-scale commercial manufacturing. For instance, earlier research by Zeng's group in 2017 utilized silver nitrate (AgNO3) as a catalyst and potassium persulfate as an oxidant in acetonitrile at a high temperature of 100°C for 3 hours. While effective, the requirement for stoichiometric or near-stoichiometric amounts of precious silver salts introduces substantial raw material costs and creates complex waste streams containing heavy metals that require expensive removal processes to meet pharmaceutical purity standards. Furthermore, the elevated reaction temperature of 100°C demands significant energy input and poses safety risks associated with heating organic solvents under pressure.
Subsequent developments attempted to mitigate some issues but introduced new constraints. In 2018, Yuan's group reported a method using tert-butyl hydroperoxide (TBHP) as an oxidant in dichloroethane at 50°C. Although the temperature was lower, TBHP is a hazardous peroxide that requires careful handling and storage, complicating supply chain logistics and increasing safety compliance costs. Additionally, many of these conventional protocols rely on alpha-keto acids as acylating agents, which are often more expensive and less stable than simple aldehydes. The reliance on transition metals, harsh oxidants, and elevated temperatures collectively creates a bottleneck for the cost reduction in pharmaceutical intermediate manufacturing, driving the urgent need for a milder, metal-free alternative.
The Novel Approach
The methodology described in patent CN112409275B fundamentally reimagines this transformation by employing visible light photoredox catalysis. As illustrated in the general reaction scheme below, the process combines a quinoxalinone substrate with an aldehyde in the presence of an organic photocatalyst and a persulfate oxidant under nitrogen atmosphere. The reaction proceeds at room temperature under the irradiation of a visible light source, typically a 20W blue LED lamp, for a duration of 16 to 24 hours. This eliminates the need for external heating, drastically reducing energy consumption and improving operational safety. By replacing toxic silver catalysts with inexpensive organic dyes like eosin Y disodium salt, the process not only lowers the bill of materials but also simplifies the downstream purification workflow by removing the necessity for heavy metal scavenging steps.

Moreover, the use of simple aldehydes as the acyl source is a strategic advantage for procurement teams. Aldehydes are commodity chemicals available in vast structural diversity and at low cost compared to specialized keto-acids. The reaction conditions are remarkably mild and stable, allowing for a broader tolerance of functional groups on both the quinoxalinone and the aldehyde components. This robustness ensures that the synthesis of complex 3-acylquinoxalinone derivatives can be achieved with high reliability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates. The combination of ambient temperature, visible light energy, and earth-abundant reagents establishes a new standard for sustainable chemical manufacturing in this sector.
Mechanistic Insights into Eosin Y-Catalyzed Photoredox Acylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for process development. The reaction initiates when the eosin Y disodium salt photocatalyst absorbs photons from the blue LED light source, transitioning from its ground state to an excited state. This excited photocatalyst acts as a potent single-electron transfer agent, interacting with the persulfate oxidant (such as ammonium persulfate or potassium persulfate) to generate highly reactive sulfate radical anions. These sulfate radicals are capable of abstracting a hydrogen atom from the formyl group of the aldehyde substrate, thereby generating an acyl radical species. This acyl radical is the key nucleophilic intermediate that attacks the electron-deficient C3 position of the quinoxalinone ring system.
Following the radical addition, the resulting intermediate undergoes a subsequent oxidation and deprotonation sequence to restore aromaticity, yielding the final 3-acylquinoxalinone product. The elegance of this mechanism lies in its ability to generate reactive radical species under extremely mild conditions without the need for thermal initiation or metal mediation. From an impurity control perspective, the specificity of the radical generation minimizes side reactions often seen in thermal processes, such as over-oxidation or polymerization of the aldehyde. The use of eosin Y, a well-characterized organic dye, ensures that the catalyst itself does not introduce metallic impurities into the product stream. This clean reaction profile significantly reduces the burden on analytical quality control labs, as the impurity spectrum is simpler and easier to manage compared to metal-catalyzed counterparts, ensuring consistent high-purity pharmaceutical intermediate batches.

How to Synthesize 3-Acylquinoxalinone Efficiently
The practical implementation of this photocatalytic protocol is straightforward and relies on standard laboratory equipment that can be readily adapted for pilot and production scales. The procedure involves charging a reaction vessel with the quinoxalinone substrate, the chosen aldehyde, the eosin Y photocatalyst, and the persulfate oxidant in a suitable organic solvent such as acetonitrile. After purging the system with nitrogen to exclude oxygen, the mixture is stirred under blue LED irradiation at room temperature. Reaction progress is monitored via thin-layer chromatography (TLC) until completion, typically within 16 to 24 hours. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below.
- Mix quinoxalinone compound, aldehyde derivative, eosin Y disodium salt photocatalyst, and ammonium persulfate oxidant in a reaction vessel under nitrogen protection.
- Add organic solvent such as acetonitrile and irradiate the mixture with a 20W blue LED light source at room temperature for 16 to 24 hours.
- Monitor reaction progress via TLC, then perform extraction with ethyl acetate and water, followed by silica gel column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling strategic advantages that extend beyond mere technical novelty. The shift from metal-based catalysis to organophotocatalysis directly addresses several pain points associated with the sourcing and cost management of complex chemical intermediates. By eliminating the dependency on precious metals like silver, manufacturers can insulate their supply chains from the volatility of metal markets and reduce the regulatory burden associated with heavy metal limits in drug substances. Furthermore, the use of commodity aldehydes instead of specialized keto-acids simplifies the vendor qualification process and enhances supply security.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive reagents with low-cost alternatives. Replacing silver nitrate with eosin Y disodium salt results in a drastic reduction in catalyst costs, as organic dyes are significantly cheaper than precious metal salts on a molar basis. Additionally, the elimination of high-temperature heating requirements leads to substantial energy savings over the course of long production campaigns. The simplified workup procedure, which avoids complex metal scavenging resins or extensive washing steps to remove metal residues, further reduces the consumption of auxiliary materials and labor hours, contributing to overall cost optimization in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on simple aromatic aldehydes as starting materials ensures a robust and diversified supply base. Unlike specialized acylating agents that may have limited suppliers or long lead times, benzaldehyde derivatives are produced globally in massive quantities for various industries. This abundance guarantees consistent availability and competitive pricing, reducing the risk of production delays due to raw material shortages. Moreover, the stability of the reagents under ambient storage conditions simplifies inventory management and reduces the need for specialized cold-chain logistics or hazardous material handling protocols required for peroxides like TBHP.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this photochemical process is highly favorable. The reaction operates at room temperature and atmospheric pressure, minimizing the engineering controls required for high-pressure reactors or high-temperature heating systems. This inherent safety profile facilitates easier scale-up from gram to kilogram and ton scales. Environmentally, the avoidance of toxic heavy metals and hazardous peroxides aligns with increasingly stringent global environmental regulations. The waste streams generated are less toxic and easier to treat, lowering the costs associated with waste disposal and environmental compliance, thereby supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability for potential partners and licensees.
Q: What are the primary advantages of this photocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for toxic transition metal catalysts like silver nitrate and avoids harsh high-temperature conditions (100°C), utilizing instead mild room temperature visible light irradiation and inexpensive organic dyes.
Q: Can this synthesis protocol accommodate diverse aldehyde substrates?
A: Yes, the protocol demonstrates excellent substrate scope, successfully reacting with various aromatic aldehydes including those with electron-withdrawing groups like bromine and chlorine, as well as electron-donating groups.
Q: Is the purification process complex for large-scale production?
A: The purification involves standard industrial unit operations including liquid-liquid extraction and silica gel column chromatography, which are well-established and easily scalable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylquinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis method disclosed in patent CN112409275B for the production of high-value quinoxalinone derivatives. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry approach are realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-acylquinoxalinone intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this metal-free route can optimize your budget. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a seamless transition from discovery to commercial supply.
