Advanced Asymmetric Hydrogen Transfer for Commercial Scale-up of Chiral Purine Nucleosides
The pharmaceutical industry's relentless pursuit of potent antiviral agents has placed chiral carbocyclic purine nucleosides at the forefront of medicinal chemistry research. Compounds such as Abacavir and Entecavir, which are critical for treating HIV and HBV respectively, rely heavily on the precise stereochemical configuration of their carbocyclic rings to exhibit biological activity. A groundbreaking approach detailed in patent CN109761984B introduces a highly efficient method for synthesizing these vital intermediates via asymmetric hydrogen transfer. This technology represents a paradigm shift from hazardous high-pressure hydrogenation to a safer, more controllable catalytic system. By utilizing a specialized chiral ruthenium catalyst in conjunction with a formic acid and triethylamine hydrogen donor system, the process achieves dynamic kinetic resolution of racemic alpha-purine substituted cyclopentanones. This innovation not only simplifies the synthetic route but also ensures the production of optically pure materials essential for regulatory compliance in drug manufacturing.
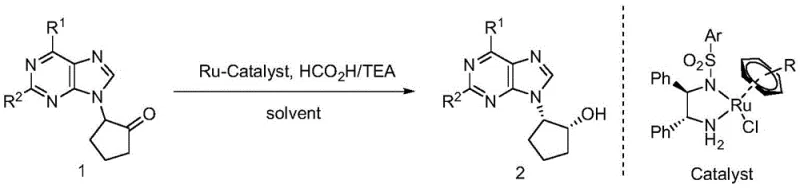
The implementation of this asymmetric hydrogen transfer protocol offers a robust solution for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. The ability to generate two chiral centers simultaneously with high fidelity addresses a long-standing challenge in nucleoside analogue synthesis. Furthermore, the mild reaction conditions and the use of readily available starting materials position this methodology as a cornerstone for cost reduction in API manufacturing. As global demand for antiviral therapeutics continues to rise, mastering this synthetic pathway provides a strategic advantage in securing supply chains for next-generation nucleoside drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral carbocyclic nucleosides has been plagued by significant operational hazards and inefficiencies inherent to traditional reduction techniques. The first conventional route typically involves asymmetric hydrogenation using molecular hydrogen gas under high pressure. This approach necessitates expensive high-pressure reactors and rigorous safety protocols to mitigate the risks of hydrogen leakage and potential explosions, creating a substantial barrier to entry for many manufacturing facilities. Moreover, the second conventional route, which employs asymmetric hydrogen transfer, has often suffered from poor catalytic efficiency and inadequate stereocontrol. Previous catalysts frequently failed to distinguish effectively between the enantiomers of the racemic starting material, leading to low yields of the desired isomer and requiring cumbersome purification steps to remove unwanted diastereomers. These limitations result in increased production costs, extended lead times, and a larger environmental footprint due to solvent waste and energy consumption associated with high-pressure operations.
The Novel Approach
In stark contrast, the novel methodology described in the patent data leverages a sophisticated chiral ruthenium catalyst system to overcome these historical bottlenecks. By employing a specific ruthenium complex featuring a chiral diamine ligand and an arene group, the reaction proceeds via a dynamic kinetic resolution mechanism that is both rapid and highly selective. The use of a formic acid and triethylamine mixture as the hydrogen source eliminates the need for gaseous hydrogen entirely, allowing the reaction to proceed safely at atmospheric pressure and near-ambient temperatures (around 27°C). This transition from high-pressure gas to liquid reagents drastically simplifies the engineering requirements for the reactor setup. Additionally, the new catalyst design ensures that the racemization of the starting ketone is faster than the reduction step, theoretically allowing for a 100% yield of the chiral product from a racemic starting material. This efficiency translates directly into reduced raw material costs and minimized waste generation, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogen Transfer
The success of this synthetic strategy hinges on the precise interaction between the chiral ruthenium catalyst and the substrate during the hydride transfer event. The catalyst, specifically variants like Catalyst D shown in the structural analysis, operates through an outer-sphere mechanism where the metal center does not directly coordinate to the carbonyl oxygen of the ketone substrate. Instead, the activation occurs through a concerted six-membered transition state involving the ruthenium hydride species and the amine proton of the ligand. This bifunctional activation allows for the simultaneous delivery of a hydride ion and a proton to the carbonyl group with exceptional stereochemical precision. The bulky substituents on the chiral ligand, such as the pentafluorophenyl group in Catalyst D, create a steric environment that strongly favors the formation of one enantiomer over the other. This steric differentiation is crucial for achieving the reported enantiomeric excess values of up to 99%, ensuring that the final product meets the stringent purity requirements for pharmaceutical applications.
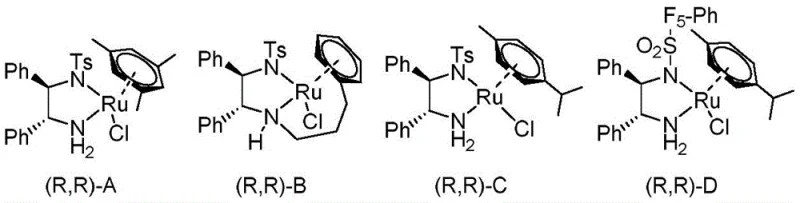
Furthermore, the dynamic kinetic resolution aspect of this mechanism is vital for maximizing atom economy. In a standard kinetic resolution, the maximum theoretical yield is limited to 50% because only one enantiomer of the racemic starting material reacts. However, in this system, the basic conditions provided by triethylamine facilitate the rapid racemization of the unreacted ketone enantiomer. This continuous interconversion ensures that the entire pool of starting material is eventually funneled into the productive catalytic cycle that generates the desired chiral alcohol. The result is a process that combines the benefits of resolution with the efficiency of asymmetric synthesis. Understanding this mechanistic nuance is critical for process chemists aiming to scale this reaction, as maintaining the correct balance of base and hydrogen donor is essential to sustain the racemization rate without deactivating the catalyst or promoting side reactions.
How to Synthesize Chiral Carbocyclic Purine Nucleosides Efficiently
Executing this synthesis requires careful attention to the preparation of the catalytic system and the control of reaction parameters to ensure reproducibility on a commercial scale. The process begins with the dissolution of the racemic alpha-purine substituted cyclopentanone in a suitable organic solvent, with dioxane identified as the optimal medium for balancing solubility and reaction rate. The chiral ruthenium catalyst is then introduced at a loading of approximately 1 mol%, followed by the addition of the formic acid and triethylamine azeotrope. Maintaining the reaction temperature at 27°C is critical; while higher temperatures might accelerate the rate, they could compromise the enantioselectivity, whereas lower temperatures might stall the racemization equilibrium necessary for dynamic kinetic resolution. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining racemic alpha-purine substituted cyclopentanone, chiral ruthenium catalyst (e.g., Catalyst D), and dioxane solvent in a reaction vessel.
- Add the hydrogen source mixture consisting of formic acid and triethylamine (typically in a 1: 1 molar ratio) to the reaction system under inert atmosphere.
- Stir the reaction mixture at a mild temperature of 27°C for approximately 24 hours to allow dynamic kinetic resolution and reduction to proceed.
- Upon completion, concentrate the organic phase under vacuum and purify the resulting chiral nucleoside via column chromatography to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric hydrogen transfer technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of high-pressure hydrogen infrastructure represents a significant capital expenditure saving, as facilities do not need to invest in specialized autoclaves or extensive safety monitoring systems for flammable gases. This reduction in equipment complexity lowers the barrier for contract manufacturing organizations to offer this service, thereby increasing the number of potential suppliers and enhancing supply chain resilience. Additionally, the use of formic acid and triethylamine as reagents leverages commodity chemicals that are inexpensive and readily available in bulk quantities globally, insulating the production process from the volatility associated with specialized reagents or rare metal precursors.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the high atom efficiency and the ability to utilize racemic starting materials. Since the dynamic kinetic resolution converts the entire racemic mixture into the desired chiral product, there is no need to discard 50% of the starting material as is common in classical resolutions. This doubling of effective yield from the starting ketone significantly lowers the cost of goods sold. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, and the high selectivity minimizes the need for expensive and solvent-intensive chromatographic purifications, leading to substantial cost savings in downstream processing.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system contributes to more predictable production schedules. Unlike sensitive enzymatic processes that may require strict pH and temperature controls, this chemical catalytic system tolerates a reasonable range of operating conditions without significant loss of performance. The availability of diverse substrate scopes, as demonstrated by the successful synthesis of various purine derivatives with different substituents, means that a single platform technology can be adapted to produce a wide array of nucleoside analogues. This flexibility allows manufacturers to respond quickly to changes in market demand for different antiviral candidates without retooling entire production lines.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the absence of gas-liquid mass transfer limitations that typically hinder high-pressure hydrogenations. The homogeneous nature of the reaction mixture ensures consistent heat and mass transfer, reducing the risk of hot spots or runaway reactions. From an environmental perspective, the process generates fewer hazardous byproducts compared to stoichiometric reduction methods using borohydrides or aluminum hydrides. The primary byproduct is carbon dioxide from the decomposition of formic acid, which is easily managed. This cleaner profile simplifies waste treatment protocols and helps manufacturing sites maintain compliance with increasingly stringent environmental regulations regarding solvent and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this technology for integration into their existing workflows. Understanding these specifics is crucial for assessing the feasibility of adopting this route for specific target molecules.
Q: What are the safety advantages of this asymmetric hydrogen transfer method compared to traditional hydrogenation?
A: Unlike traditional methods that require high-pressure hydrogen gas which poses significant explosion risks and requires specialized equipment, this method utilizes a formic acid/triethylamine azeotrope as a safe, liquid hydrogen source under atmospheric pressure and mild temperatures.
Q: What level of stereoselectivity can be achieved with this ruthenium-catalyzed process?
A: The process demonstrates exceptional stereocontrol, achieving enantiomeric excess (ee) values of up to 99% and diastereomeric ratios (dr) greater than 99:1, specifically yielding the desired cis-configured products exclusively through dynamic kinetic resolution.
Q: Can the synthesized chiral nucleosides be further derivatized for drug discovery?
A: Yes, the resulting hydroxyl group on the cyclopentane ring serves as a versatile handle for further functionalization, allowing conversion into mesylates, azides, and subsequently 1,2,3-triazole derivatives via click chemistry, expanding the chemical space for antiviral screening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Carbocyclic Purine Nucleosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antiviral medications. Our technical team has extensively analyzed the potential of the asymmetric hydrogen transfer pathway described in CN109761984B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm enantiomeric excess values exceeding 99% as required for clinical grade materials.
We invite pharmaceutical partners to collaborate with us to optimize this synthetic route for their specific pipeline candidates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this catalytic method for your specific project. We encourage you to contact us to obtain specific COA data for similar nucleoside intermediates and to discuss route feasibility assessments tailored to your volume requirements. Let us be your strategic partner in delivering high-purity chiral building blocks efficiently and reliably.
