Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
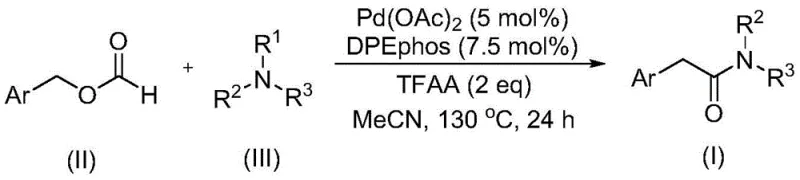
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing the amide bond, a ubiquitous motif found in countless bioactive molecules. Patent CN111978194B introduces a transformative approach to synthesizing aryl acetamide compounds, addressing long-standing challenges in transition metal-catalyzed carbonylation. This innovation leverages a palladium-catalyzed system that utilizes benzyl formate not merely as a solvent or additive, but as a dual-function reagent serving as both a carbon monoxide source and a reactant. By circumventing the need for hazardous gaseous carbon monoxide and avoiding the use of excessive external oxidants for tertiary amine activation, this technology offers a safer, more sustainable pathway for producing high-value intermediates. For R&D teams and procurement strategists alike, this represents a significant leap forward in process chemistry, enabling the efficient assembly of complex molecular architectures with enhanced functional group tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamides has relied heavily on the direct amidation of phenylacetic acid derivatives or traditional carbonylation reactions involving aryl halides. These conventional routes are fraught with significant logistical and safety hurdles that complicate commercial manufacturing. Direct amidation often necessitates harsh coupling reagents and elevated temperatures, which can lead to poor atom economy and difficult purification processes. Furthermore, traditional transition metal-catalyzed carbonylation typically demands the handling of toxic carbon monoxide gas, often in combination with oxygen, creating explosive mixtures that require specialized high-pressure equipment and rigorous safety protocols. Additionally, the activation of tertiary amines for C-N bond cleavage in these contexts remains a formidable chemical challenge, frequently resulting in low conversion rates or requiring stoichiometric amounts of expensive and waste-generating oxidants.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by introducing benzyl formate as a stable, liquid surrogate for carbon monoxide. This strategic substitution eliminates the immediate dangers associated with high-pressure CO gas cylinders, allowing reactions to proceed in standard sealed vessels under much safer conditions. Crucially, the system demonstrates an unprecedented ability to cleave the C-N bonds of tertiary amines without the addition of external oxidants, a feat that significantly streamlines the reaction profile. By employing a palladium catalyst in conjunction with specific ligands like DPEphos and trifluoroacetic anhydride, the process achieves high reaction efficiency and broad substrate scope. This novel approach not only simplifies the operational workflow but also enhances the designability of the final products, allowing chemists to incorporate diverse functional groups that might be incompatible with harsher traditional methods.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst, the phosphine ligand, and the unique reactivity of benzyl formate. The reaction mechanism likely involves the oxidative addition of the palladium species to the benzyl formate, facilitating the release of carbon monoxide in situ which then inserts into the metal-carbon bond. This generated acyl-palladium intermediate subsequently undergoes nucleophilic attack by the tertiary amine. What distinguishes this system is the role of trifluoroacetic anhydride (TFAA), which acts as a promoter to facilitate the difficult C-N bond cleavage of the tertiary amine substrate. The ligand, specifically bis(2-diphenylphosphinophenyl) ether (DPEphos), provides the necessary steric and electronic environment to stabilize the active catalytic species, ensuring high turnover numbers and preventing catalyst deactivation. This precise orchestration allows for the transformation to proceed smoothly at temperatures around 130°C, balancing kinetic energy with thermal stability.
From an impurity control perspective, this mechanism offers distinct advantages over oxidative carbonylation routes. Since no external oxidants like peroxides or oxygen gas are required to drive the C-N bond scission, the formation of over-oxidized byproducts is inherently minimized. The use of benzyl formate ensures a controlled release of the carbonyl unit, preventing the accumulation of reactive intermediates that could lead to polymerization or side reactions. Furthermore, the compatibility of the catalyst system with various substituents on the aryl ring—such as electron-donating methoxy groups or electron-withdrawing trifluoromethyl groups—demonstrates a robust tolerance that simplifies the purification of the final aryl acetamide. This mechanistic elegance translates directly into higher purity profiles, reducing the burden on downstream processing and quality control laboratories.
How to Synthesize Aryl Acetamide Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and reaction parameters to maximize yield and efficiency. The process begins with the precise combination of the palladium catalyst, ligand, and activators in a suitable aprotic solvent. While the patent outlines a general procedure applicable to a wide range of substrates, optimizing the molar ratios of benzyl formate to tertiary amine is critical for driving the equilibrium towards the desired product. The following guide summarizes the standardized operational steps derived from the patent examples, providing a clear roadmap for laboratory execution and subsequent scale-up efforts.
- Combine palladium acetate catalyst, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 120°C and 140°C, preferably 130°C, and maintain stirring for approximately 24 hours.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target aryl acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The shift away from hazardous gaseous reagents to stable liquid surrogates fundamentally alters the risk profile of the manufacturing process, potentially lowering insurance costs and reducing the need for specialized containment infrastructure. Moreover, the reliance on commercially abundant starting materials ensures a resilient supply chain that is less susceptible to the volatility often seen with specialty gases or exotic coupling reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and widely available raw materials such as benzyl formate, which can be derived from cheap formic acid and benzyl alcohol. By eliminating the need for expensive external oxidants and reducing the complexity of the catalyst system, the overall cost of goods sold is significantly optimized. The simplified post-treatment process, which involves standard filtration and chromatography rather than complex extraction or distillation sequences, further contributes to substantial cost savings by reducing labor hours and solvent consumption during the isolation phase.
- Enhanced Supply Chain Reliability: Sourcing stability is a paramount concern for any large-scale chemical operation, and this method excels by utilizing commodity chemicals that are readily accessible from multiple global suppliers. The avoidance of regulated toxic gases like carbon monoxide removes a major bottleneck in logistics and storage, allowing for more flexible manufacturing site selection. This flexibility ensures that production can be maintained even during regional supply disruptions, providing a reliable aryl acetamide supplier with the agility to respond to market fluctuations without compromising on output volume or quality standards.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental challenges, but this methodology is inherently greener due to its high atom economy and reduced waste generation. The absence of heavy metal oxidants and the use of a closed system for carbonylation minimize the release of volatile organic compounds and hazardous effluents. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the costs associated with waste disposal and environmental remediation, making the commercial scale-up of complex amide intermediates both environmentally responsible and economically sound.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on reaction conditions, substrate compatibility, and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve safety compared to traditional carbonylation?
A: Traditional carbonylation often requires hazardous carbon monoxide gas or explosive CO/O2 mixtures. This patented method utilizes benzyl formate as a safe, liquid carbon monoxide surrogate, significantly reducing operational risks and eliminating the need for high-pressure gas infrastructure.
Q: Can this process activate tertiary amines effectively?
A: Yes, unlike conventional methods that struggle with tertiary amine C-N bond activation, this palladium-catalyzed system efficiently cleaves the C-N bond of tertiary amines without requiring additional external oxidants, offering a distinct advantage for synthesizing complex amide structures.
Q: What represents the primary cost driver in this synthesis?
A: The process relies on commercially available and inexpensive starting materials such as benzyl formate, formic acid derivatives, and common tertiary amines. The use of standard palladium catalysts and simple post-treatment procedures further optimizes the cost structure for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
As the demand for high-quality pharmaceutical intermediates continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of modern chemical synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch, guaranteeing that the final aryl acetamide products meet the highest industry standards for potency and impurity profiles.
We invite you to collaborate with our technical procurement team to explore how this advanced palladium-catalyzed technology can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial reliability.
