Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-up
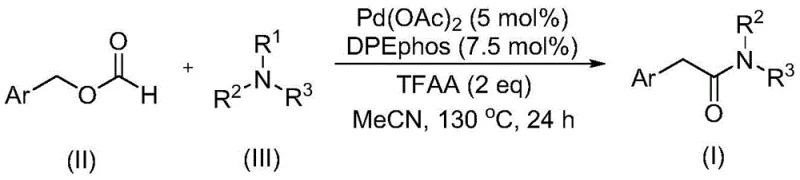
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for safer, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN111978194B, which discloses a novel preparation method for aryl acetamide compounds. This technology represents a paradigm shift from traditional amidation strategies, leveraging a sophisticated palladium-catalyzed carbonylation reaction. By utilizing benzyl formate as a dual-purpose reagent—acting as both a carbon monoxide source and a reactant—and employing tertiary amines as the nitrogen source, this method overcomes historical challenges associated with C-N bond activation. For R&D directors and procurement specialists seeking a reliable aryl acetamide supplier, understanding the mechanistic depth and commercial viability of this patent is crucial for optimizing supply chains and reducing manufacturing costs in the production of complex bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aryl acetamide scaffolds has relied heavily on the direct amidation of phenylacetic acid derivatives or transition metal-catalyzed carbonylation using gaseous carbon monoxide. While effective in some contexts, these conventional routes suffer from significant drawbacks that hinder large-scale pharmaceutical intermediates manufacturing. The direct use of phenylacetic acid often requires harsh activation conditions and generates substantial stoichiometric waste. Furthermore, traditional carbonylation strategies frequently necessitate the use of high-pressure CO gas, which poses severe safety hazards and requires specialized, expensive infrastructure. Additionally, activating the C-N bond of tertiary amines—a common structural motif in drug candidates—has historically been a formidable challenge, often requiring excessive amounts of toxic oxidants or explosive CO/O2 mixtures. These factors collectively increase the operational risk and environmental footprint, making legacy processes less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology described in CN111978194B introduces a streamlined and inherently safer alternative. This novel approach utilizes benzyl formate, a stable and commercially available liquid, which decomposes in situ to provide the necessary carbonyl unit without the logistical nightmares of gas handling. The reaction is mediated by a palladium catalyst system, specifically palladium acetate paired with the bidentate ligand DPEphos, in the presence of trifluoroacetic anhydride (TFAA). This combination facilitates the cleavage of the tertiary amine C-N bond under relatively mild thermal conditions (120-140°C) without the need for external oxidants. This innovation not only simplifies the reaction setup but also dramatically improves the atom economy and safety profile, offering a robust pathway for the commercial scale-up of complex aryl acetamides.
Mechanistic Insights into Pd-Catalyzed Carbonylation and C-N Activation
From a mechanistic perspective, this transformation is a tour de force of organometallic chemistry. The catalytic cycle likely initiates with the oxidative addition of the palladium(0) species into the benzylic C-O bond of the benzyl formate, activated by the Lewis acidic nature of TFAA. The wide bite angle of the DPEphos ligand plays a critical role here, stabilizing the palladium center and promoting the subsequent migratory insertion of the carbonyl moiety. Unlike traditional methods that struggle with steric hindrance, this system effectively activates the tertiary amine. The absence of external oxidants suggests a unique pathway where the formate itself or the TFAA assists in the C-N bond scission, preventing the formation of over-oxidized byproducts. This precise control over the reaction trajectory is vital for maintaining high-purity aryl acetamides, as it minimizes the generation of difficult-to-remove impurities that often plague amide syntheses involving aggressive oxidizing agents.
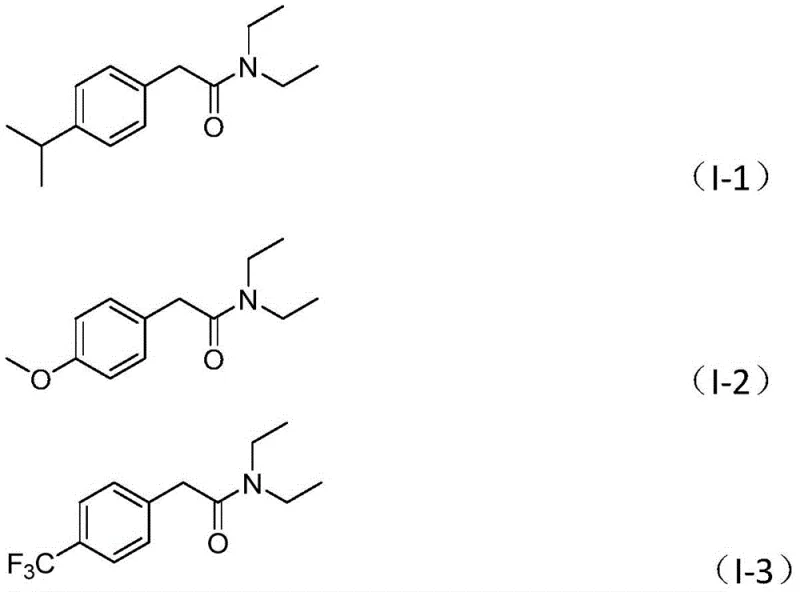
Furthermore, the substrate scope demonstrated in the patent highlights the versatility of this catalytic system. The reaction tolerates a wide array of electronic environments on the aryl ring, including electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as trifluoromethyl and cyano moieties. Heterocyclic substrates, including furan and thiophene derivatives, are also compatible, yielding the corresponding products in high efficiency. This broad tolerance indicates that the catalytic species is robust against potential catalyst poisons often found in heteroatom-rich drug intermediates. For process chemists, this means fewer failed batches and a more predictable outcome when scaling from gram to kilogram quantities, directly addressing the need for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Aryl Acetamide Efficiently
The operational simplicity of this method makes it highly attractive for industrial adoption. The procedure involves charging a reactor with the palladium catalyst, ligand, benzyl formate, tertiary amine, and TFAA in a solvent like acetonitrile. The mixture is then heated to 130°C for approximately 24 hours. Post-reaction workup is straightforward, involving filtration and standard purification techniques such as column chromatography. This ease of operation reduces the training burden on technical staff and minimizes the risk of operator error. For those interested in implementing this route, the detailed standardized synthesis steps are outlined below to ensure reproducibility and safety compliance.
- Combine palladium acetate catalyst, DPEphos ligand, benzyl formate, tertiary amine, and trifluoroacetic anhydride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed vessel and maintain stirring for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity aryl acetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits. The primary driver for cost reduction in pharmaceutical intermediates manufacturing lies in the raw material selection and process safety. Benzyl formate and tertiary amines are commodity chemicals with stable global supply chains, insulating manufacturers from the volatility associated with specialty reagents. Moreover, the elimination of high-pressure CO gas cylinders removes a significant capital expenditure barrier, allowing production to occur in standard glass-lined or stainless steel reactors without specialized high-pressure ratings. This flexibility enhances supply chain resilience, enabling faster response times to market demands.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted. By replacing hazardous gaseous CO with liquid benzyl formate, the facility saves on the costs associated with gas storage, monitoring, and safety compliance protocols. Additionally, the catalyst loading is relatively low (5 mol%), and the ligand DPEphos, while specialized, is used in catalytic quantities that do not disproportionately impact the bill of materials. The absence of expensive external oxidants further strips away unnecessary reagent costs. Qualitatively, this leads to a leaner cost structure per kilogram of product, allowing for more competitive pricing strategies in the global marketplace without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry. The reliance on widely available starting materials like benzyl alcohol derivatives and common tertiary amines ensures that production is not bottlenecked by the scarcity of exotic precursors. The robustness of the reaction conditions—tolerating various functional groups—means that raw material specifications can be slightly relaxed without compromising final product quality, providing procurement teams with greater flexibility in vendor selection. This redundancy in the supply base mitigates the risk of disruption, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental challenges, but this method is inherently greener. The avoidance of toxic oxidants and high-pressure gases reduces the facility's environmental liability and simplifies waste treatment protocols. The reaction produces fewer hazardous byproducts, aligning with increasingly stringent global environmental regulations. From a scalability standpoint, the use of standard solvents like acetonitrile and moderate temperatures facilitates a smooth transition from pilot plant to full commercial production, ensuring that volume requirements can be met reliably as demand grows.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the patent's specifics and its practical application. These insights are derived directly from the experimental data and claims within CN111978194B, providing a clear picture of what to expect during technology transfer. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production lines.
Q: What is the primary advantage of using benzyl formate in this synthesis?
A: Benzyl formate serves as a safe, liquid carbon monoxide source, eliminating the need for handling hazardous high-pressure CO gas while simultaneously acting as a reactant.
Q: Does this method require external oxidants for tertiary amine activation?
A: No, a key innovation of this patent is that the C-N bond cleavage of the tertiary amine occurs without the need for additional oxidants, simplifying the reaction profile and reducing impurity risks.
Q: What types of substrates are compatible with this catalytic system?
A: The system exhibits broad functional group tolerance, successfully processing aryl groups with electron-donating or withdrawing substituents, as well as heterocycles like furan and thiophene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
The technological potential of this palladium-catalyzed carbonylation route is immense, offering a pathway to high-value intermediates with superior efficiency. At NINGBO INNO PHARMCHEM, we pride ourselves on our ability to translate such innovative academic and patent literature into robust commercial realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, ensuring that every batch of aryl acetamide meets the exacting standards required by the global pharmaceutical industry.
We invite you to explore how this advanced synthesis method can optimize your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring your next generation of therapeutic agents to market faster and more economically.
