Advanced Catalytic Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved biological profiles and synthetic accessibility, particularly in the realm of oncology therapeutics. A groundbreaking development in this sector is detailed in patent CN114524701B, which discloses a robust and highly enantioselective method for synthesizing N-N axis chiral pyrrole derivatives. These compounds are not merely academic curiosities; they have demonstrated potent cytotoxic activity against QGP-1 tumor cells, marking them as high-value candidates for anticancer drug discovery. The core innovation lies in the utilization of chiral phosphoric acid organocatalysts to facilitate the condensation of indoleamines or pyrroleamines with 1,4-diketone derivatives. This approach bypasses the limitations of traditional dynamic kinetic resolution, offering a direct, atom-economical route to complex chiral architectures. As a leading entity in fine chemical manufacturing, we recognize the immense potential of this technology to streamline the supply chain for high-purity pharmaceutical intermediates.
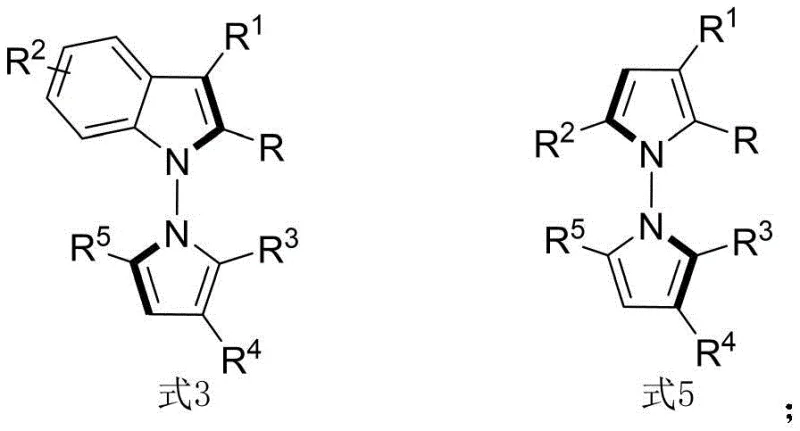
The structural diversity achievable through this methodology is substantial, allowing for the modulation of R groups to fine-tune pharmacokinetic properties. The general structures, depicted as Formula 3 and Formula 5 in the patent documentation, highlight the versatility of the N-N axial chirality motif. By varying substituents such as alkyl, aryl, ester, and halogen groups, medicinal chemists can generate extensive libraries for structure-activity relationship (SAR) studies. This capability is crucial for modern drug discovery programs where rapid iteration is key to identifying lead compounds with optimal efficacy and safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-N axis chiral heterocycles has been fraught with challenges, primarily relying on dynamic kinetic resolution or desymmetrization strategies that often suffer from limited substrate scope and moderate yields. Conventional methods frequently require harsh reaction conditions, expensive transition metal catalysts, or complex multi-step sequences that hinder scalability. Furthermore, prior art lacked efficient protocols for constructing these specific N-N linked pyrrole-indole or pyrrole-pyrrole systems via in-situ ring formation. The reliance on racemic mixtures necessitates difficult downstream separation processes, significantly increasing the cost of goods and extending lead times for clinical material. These inefficiencies create bottlenecks in the supply chain for reliable pharmaceutical intermediate suppliers aiming to deliver complex molecules.
The Novel Approach
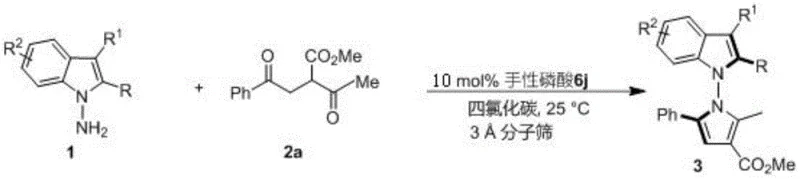
The methodology described in the patent represents a paradigm shift by employing a direct condensation strategy catalyzed by chiral phosphoric acids. This novel approach utilizes readily available starting materials—indoleamines or pyrroleamines and 1,4-diketone derivatives—to construct the target scaffolds in a single operational step. The reaction proceeds under remarkably mild conditions, typically at room temperature (25°C) in carbon tetrachloride, which simplifies process control and enhances safety. By leveraging the power of asymmetric organocatalysis, this method achieves exceptional enantioselectivity, with some examples reaching up to 96% ee. This high level of stereocontrol eliminates the need for costly chiral resolution steps, directly addressing the pain points of cost reduction in API manufacturing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
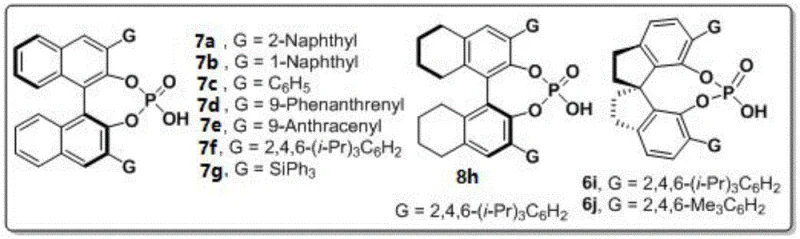
The success of this synthesis hinges on the precise interaction between the chiral phosphoric acid catalyst and the substrates. The catalyst, typically derived from binaphthyl, octahydrobinaphthyl, or spiro backbones, acts as a bifunctional activator. It simultaneously activates the electrophilic 1,4-diketone and the nucleophilic amine through a network of hydrogen bonds, organizing the transition state within a well-defined chiral pocket. This rigid environment dictates the facial selectivity of the attack, ensuring the formation of the desired N-N axis configuration with high fidelity. The patent highlights various catalyst structures, such as those shown in Formulas 6, 7, and 8, where the steric bulk of the substituents (G groups) at the 3,3'-positions plays a critical role in shielding one face of the substrate.
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The high stereoselectivity inherently minimizes the formation of unwanted diastereomers and enantiomers, resulting in a cleaner crude reaction profile. This reduces the burden on downstream purification processes, such as silica gel column chromatography, and improves the overall mass balance of the process. Furthermore, the use of 3A molecular sieves as an additive helps to sequester water generated during the condensation, driving the equilibrium towards product formation and preventing hydrolysis of sensitive intermediates. This attention to mechanistic detail ensures that the process is not only scientifically elegant but also practically robust for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to specific stoichiometric ratios and environmental controls outlined in the experimental examples. The standard protocol involves dissolving the amine substrate and the 1,4-diketone in anhydrous carbon tetrachloride, followed by the addition of the chiral catalyst and activated molecular sieves. Maintaining anhydrous conditions is paramount to prevent catalyst deactivation and side reactions. The reaction is typically monitored by thin-layer chromatography (TLC) until completion, after which simple filtration removes the solid additives.
- Prepare the reaction mixture by combining indoleamine or pyrroleamine substrates with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of the optimized chiral phosphoric acid catalyst (such as compound 6j).
- Stir the reaction at room temperature (25°C) monitoring progress via TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The elimination of precious metal catalysts removes a significant cost driver and mitigates the risk of heavy metal contamination, which is a stringent regulatory requirement for pharmaceutical ingredients. Additionally, the use of commodity chemicals as starting materials ensures a stable and resilient supply base, reducing the risk of raw material shortages that can disrupt production schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metals and complex chiral auxiliaries. The high yields and enantioselectivity mean that less raw material is wasted on off-target isomers, directly improving the atom economy. Furthermore, the mild reaction temperatures reduce energy consumption associated with heating or cooling large reactors, contributing to lower operational expenditures.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route that relies on widely available indole and pyrrole derivatives, manufacturers can diversify their supplier base and avoid single-source dependencies. The simplicity of the workup procedure, involving basic filtration and concentration, allows for faster batch turnover times. This agility enables suppliers to respond more rapidly to fluctuating market demands and urgent clinical trial material requests.
- Scalability and Environmental Compliance: The reaction conditions are inherently safe and scalable, avoiding the use of pyrophoric reagents or extreme pressures that complicate plant operations. The solvent system, while requiring careful handling, is standard in the industry and can be efficiently recovered and recycled. This aligns with green chemistry principles by minimizing waste generation and facilitating easier compliance with increasingly strict environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chiral synthesis platform. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines.
Q: What is the optimal catalyst loading for this asymmetric synthesis?
A: According to the patent data, a catalyst loading of 10 mol% is generally sufficient for high conversion, though specific substrates may require up to 20 mol% to achieve optimal enantioselectivity.
Q: What biological activity do these N-N axis chiral pyrroles exhibit?
A: Biological testing indicates that these derivatives possess strong cytotoxic activity against QGP-1 pancreatic tumor cells, suggesting significant potential for oncology drug development.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes mild reaction conditions (25°C), common solvents like carbon tetrachloride, and avoids expensive transition metals, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with robust manufacturing capabilities to bring cutting-edge chemical innovations to the global market. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-N axis chiral pyrrole derivatives meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain strategy and accelerate your time to market.
