Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved biological profiles and synthetic accessibility, particularly in the realm of oncology. Patent CN114524701B introduces a groundbreaking methodology for the construction of N-N axis chiral pyrrole derivatives, a class of compounds that has historically been difficult to access with high stereochemical fidelity. This innovation addresses a critical gap in medicinal chemistry by providing a robust route to indole-pyrrole and pyrrole-pyrrole architectures that demonstrate potent cytotoxicity against QGP-1 pancreatic tumor cells. Unlike previous approaches that relied heavily on dynamic kinetic resolution or desymmetrization with limited substrate scope, this technology employs an ingenious in-situ ring formation strategy. The core breakthrough lies in the utilization of chiral phosphoric acid organocatalysts to drive the condensation of indoleamines or pyrroleamines with 1,4-diketone derivatives. This approach not only expands the chemical space available for drug discovery but also offers a reliable pharmaceutical intermediate supplier with a pathway to complex chiral molecules that were previously inaccessible or economically unviable to produce.
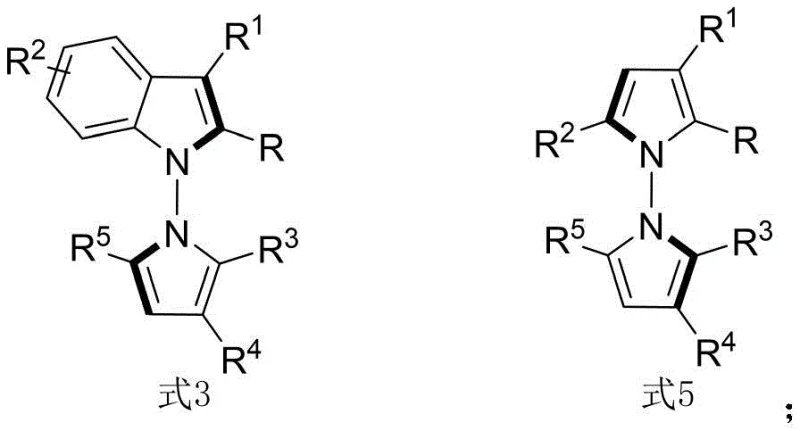
The structural diversity achievable through this method is substantial, allowing for variations in R-groups including alkyl, ester, aryl, and halogen substituents. This flexibility is paramount for lead optimization campaigns where fine-tuning electronic and steric properties is necessary to enhance binding affinity and metabolic stability. The ability to synthesize these motifs with such precision positions this technology as a key asset for developing next-generation anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral heterocycles has been plagued by significant inefficiencies that hinder both research and commercial production. Traditional methods often depend on the resolution of racemic mixtures, a process that inherently caps the maximum theoretical yield at 50% unless dynamic processes are employed, which are themselves complex to control. Furthermore, existing strategies for N-N axis chirality are frequently restricted to specific substrate classes, lacking the generality required for diverse drug discovery libraries. Many conventional routes require harsh reaction conditions, such as extreme temperatures or the use of sensitive transition metal catalysts that necessitate rigorous exclusion of air and moisture. These factors contribute to high operational costs, complicated waste streams containing heavy metals, and significant challenges in purification. For a procurement manager, these limitations translate into volatile supply chains, higher raw material costs, and extended lead times due to the difficulty in sourcing specialized chiral ligands or resolving agents. Additionally, the low atom economy associated with resolution strategies generates substantial chemical waste, conflicting with modern green chemistry mandates and increasing environmental compliance burdens.
The Novel Approach
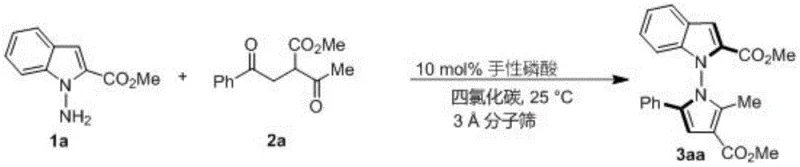
The methodology described in patent CN114524701B represents a paradigm shift by utilizing a direct, catalytic asymmetric synthesis that bypasses the need for resolution entirely. The process involves the reaction of readily available indoleamine or pyrroleamine precursors with 1,4-diketone derivatives in the presence of a chiral phosphoric acid catalyst. As illustrated in the specific synthesis of compound 3aa, the reaction proceeds smoothly in carbon tetrachloride at a mild temperature of 25°C. This mildness is a distinct advantage, preserving sensitive functional groups that might degrade under harsher conditions. The use of 3A molecular sieves as an additive further drives the equilibrium towards product formation by sequestering water generated during the condensation. The result is a highly efficient transformation that delivers products with exceptional yields, often exceeding 90%, and outstanding enantiomeric excess (ee) values reaching up to 96%. This level of performance drastically simplifies the downstream processing, as the high stereoselectivity reduces the burden on chiral separation technologies. For stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing, this route offers a compelling value proposition by maximizing material throughput and minimizing purification steps.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
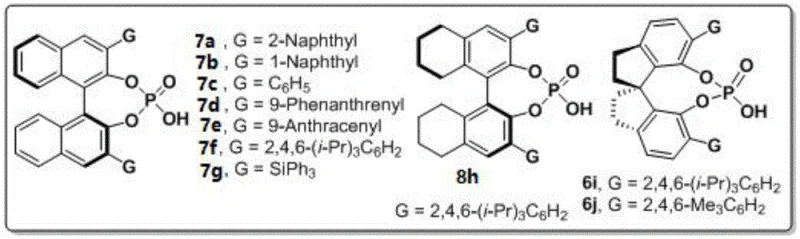
The success of this synthetic strategy hinges on the precise stereochemical control exerted by the chiral phosphoric acid catalyst. The mechanism likely involves a dual hydrogen-bonding activation mode, where the acidic proton of the phosphate activates the electrophilic carbonyl of the 1,4-diketone, while the phosphoryl oxygen interacts with the nucleophilic amine nitrogen. This bifunctional activation brings the reactants into a specific spatial orientation within the chiral pocket of the catalyst. The patent highlights the superiority of spiro-backbone derivatives, specifically catalyst 6j bearing 2,4,6-trimethylphenyl groups. The steric bulk of these mesityl groups creates a confined environment that effectively discriminates between the prochiral faces of the reacting species, thereby enforcing the formation of a single atropisomer. This rigorous control over the transition state geometry is what enables the observed high enantioselectivity. From an R&D perspective, understanding this mechanism is crucial for troubleshooting and further optimizing the reaction for novel substrates. The robustness of the catalyst ensures that side reactions, such as polymerization of the diketone or non-selective background reactions, are minimized. This leads to a cleaner crude reaction profile, which is essential for maintaining high purity standards in API intermediate production.
Furthermore, the impurity profile is managed effectively by the mild reaction conditions. Since the reaction occurs at room temperature, thermal degradation pathways are suppressed. The use of molecular sieves prevents hydrolysis of the product or intermediates, ensuring that the final isolated material meets stringent quality specifications. This mechanistic elegance translates directly into process reliability, allowing for consistent batch-to-batch reproducibility which is a critical metric for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
The practical implementation of this synthesis is designed to be straightforward and scalable, making it accessible for both laboratory research and pilot plant operations. The protocol utilizes standard laboratory equipment and does not require specialized high-pressure or cryogenic apparatus. The general procedure involves dissolving the amine and diketone substrates in carbon tetrachloride, adding the activated molecular sieves, and introducing the chiral catalyst. The reaction progress is easily monitored using thin-layer chromatography (TLC), allowing operators to quench the reaction precisely upon completion to prevent over-reaction or decomposition. Following the reaction, the workup is remarkably simple, involving filtration to remove the solid sieves and catalyst residues, followed by concentration and standard silica gel column chromatography. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining indoleamine or pyrroleamine substrates with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of the optimized chiral phosphoric acid catalyst (specifically spiro-type catalyst 6j).
- Stir the reaction at room temperature (25°C) until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers transformative benefits for the supply chain and procurement functions within a pharmaceutical organization. The shift from resolution-based methods to direct asymmetric catalysis fundamentally alters the cost structure and risk profile of producing these valuable intermediates. By eliminating the 50% yield loss inherent in resolution, the effective cost of goods sold (COGS) is drastically reduced, even before accounting for savings in solvent and energy usage. The reliance on organocatalysis rather than transition metals removes the need for expensive metal scavengers and the associated analytical testing for residual heavy metals, streamlining the quality control workflow. This simplification allows for faster release of batches and reduces the inventory holding time.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by its high atom economy and the use of commercially available starting materials. The catalyst loading is low (10 mol%), and the catalyst itself can potentially be recovered or used in lower quantities upon further optimization. The elimination of cryogenic cooling (reactions run at 25°C) results in substantial energy savings compared to traditional low-temperature organometallic reactions. Furthermore, the simplified purification process reduces the consumption of silica gel and eluents, lowering waste disposal costs. These factors combine to create a leaner, more cost-effective manufacturing process that enhances margin potential for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of raw materials. Indoleamines, pyrroleamines, and 1,4-diketones are commodity chemicals or easily synthesized from bulk precursors, reducing dependency on niche suppliers. The tolerance of the reaction to various functional groups means that a single platform technology can be used to generate a wide array of derivatives, allowing for flexible production scheduling. This versatility mitigates the risk of supply disruptions for specific analogs, ensuring continuity of supply for clinical and commercial programs. The stability of the reagents also simplifies logistics, as there is no need for specialized cold-chain shipping or inert atmosphere handling during transport.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and ton scales is facilitated by the absence of exothermic hazards typically associated with strong bases or reactive metals. The reaction can be safely performed in standard glass-lined or stainless steel reactors. From an environmental standpoint, the process aligns well with green chemistry principles by avoiding toxic heavy metals and reducing solvent usage through higher concentrations and yields. The waste stream is primarily organic and free of persistent metal contaminants, simplifying treatment and disposal. This environmental profile supports corporate sustainability goals and eases the regulatory burden associated with environmental permits and audits.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the synthesis and properties of these N-N axis chiral pyrrole derivatives. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for assessing the feasibility of integrating this chemistry into your existing development pipelines.
Q: What is the primary advantage of this synthesis method over traditional resolution techniques?
A: Unlike traditional dynamic kinetic resolution which is limited in scope, this method utilizes an in-situ ring formation strategy catalyzed by chiral phosphoric acids, achieving significantly higher enantioselectivity (up to 96% ee) and broader substrate tolerance under mild conditions.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological testing indicates that these derivatives possess strong cytotoxic activity against QGP-1 pancreatic tumor cells, with specific compounds showing high sensitivity and low IC50 values, making them promising candidates for anticancer drug development.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at room temperature (25°C) using conventional solvents and stable organocatalysts, eliminating the need for cryogenic conditions or expensive transition metals, which facilitates safe and cost-effective scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
The technological advancements detailed in patent CN114524701B represent a significant opportunity for the development of novel anticancer therapeutics, and NINGBO INNO PHARMCHEM is uniquely positioned to support your journey from discovery to commercialization. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via HPLC, to guarantee that every batch meets the highest industry standards. We understand the critical nature of API intermediates and are committed to delivering materials that facilitate your regulatory filings and clinical trials without delay.
We invite you to collaborate with our technical team to explore the full potential of this chiral pyrrole synthesis platform. Whether you require custom synthesis of specific analogs or scale-up of the lead candidate, we are ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us help you accelerate your drug development timeline with our superior manufacturing capabilities and unwavering commitment to quality.
