Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Commercial Pharmaceutical Applications
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for complex chiral scaffolds in modern drug discovery. A significant breakthrough in this domain is documented in patent CN114524701B, which details a robust and highly stereoselective method for constructing N-N axis chiral pyrrole derivatives. This technology addresses a critical gap in organic chemistry, as N-N axis chiral compounds are notoriously difficult to synthesize due to the low rotational barrier of the N-N bond. The patent introduces a novel organocatalytic approach that not only expands the structural diversity of these valuable intermediates but also demonstrates potent biological activity against QGP-1 tumor cells. For pharmaceutical developers, this represents a pivotal advancement, offering a reliable pathway to access privileged structures that were previously limited to dynamic kinetic resolution strategies. The ability to generate these motifs with high enantiomeric excess under mild conditions positions this methodology as a cornerstone for next-generation anticancer agent development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral compounds containing an N-N bond has been fraught with synthetic challenges. Traditional approaches often rely on dynamic kinetic resolution or desymmetrization reactions, which inherently limit the scope of accessible substrates and often suffer from moderate yields. Furthermore, many existing protocols require harsh reaction conditions, expensive transition metal catalysts, or complex multi-step sequences that are ill-suited for large-scale manufacturing. The scarcity of efficient methods to directly construct the N-N axis through in-situ ring formation has hindered the exploration of this chemical space. Consequently, the availability of diverse N-N axis chiral pyrrole derivatives for biological screening has been severely restricted, slowing down the identification of potential drug candidates. These limitations underscore the urgent need for a more versatile and operationally simple synthetic strategy that can overcome the thermodynamic instability associated with N-N axial chirality.
The Novel Approach
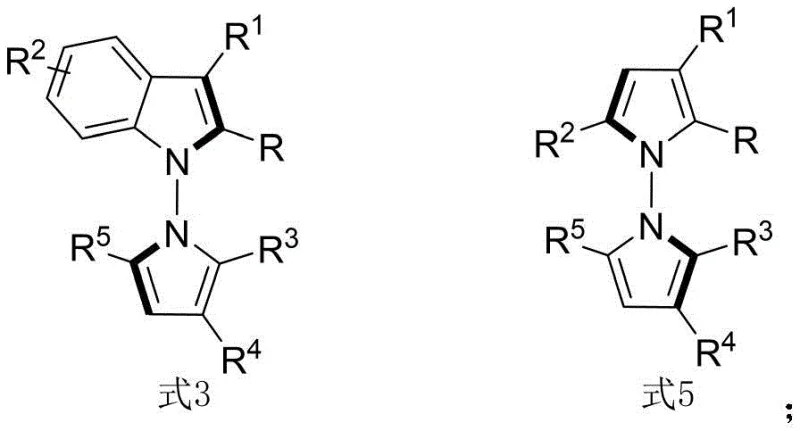
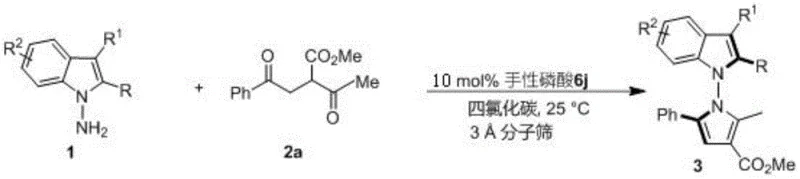
The methodology disclosed in the patent revolutionizes this field by employing a direct condensation and cyclization strategy catalyzed by chiral phosphoric acids. By utilizing indoleamines or pyrroleamines reacting with 1,4-diketone derivatives, the process achieves the formation of the N-N axis and the pyrrole ring in a single operational step. This approach eliminates the need for pre-functionalized precursors required in older methods, significantly streamlining the synthetic route. The reaction proceeds efficiently in carbon tetrachloride with 3A molecular sieves as an additive, ensuring water removal and driving the equilibrium towards product formation. Crucially, this method accommodates a wide range of substituents on both the amine and diketone components, allowing for the rapid generation of a diverse library of analogs. This versatility is essential for structure-activity relationship (SAR) studies in medicinal chemistry, enabling researchers to fine-tune the physicochemical properties of the final drug candidate.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
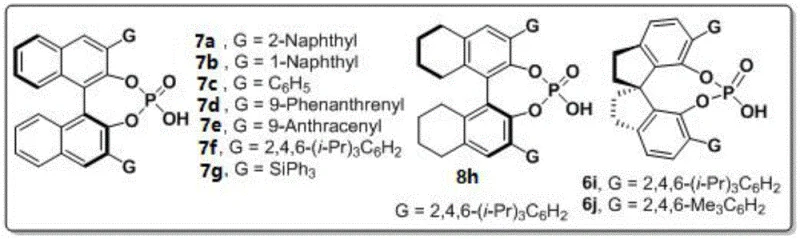
The success of this transformation hinges on the precise stereocontrol exerted by the chiral phosphoric acid catalyst. The mechanism likely involves a dual activation mode where the chiral phosphoric acid acts as a Brønsted acid to activate the carbonyl group of the 1,4-diketone while simultaneously engaging the amine nucleophile through hydrogen bonding interactions. This organized transition state restricts the conformational freedom of the reacting species, effectively locking the rotation around the forming N-N bond and directing the attack to one specific face of the prochiral center. Among the various catalysts screened, the spiro-backbone chiral phosphoric acid (specifically compound 6j with 2,4,6-trimethylphenyl groups) demonstrated superior performance. The rigid spiro structure provides a well-defined chiral pocket that enhances the discrimination between enantiomeric pathways, resulting in exceptional enantioselectivity. This level of control is critical for pharmaceutical applications, where the presence of the wrong enantiomer can lead to reduced efficacy or unwanted toxicity.
Furthermore, the choice of catalyst directly influences the impurity profile of the final product. High enantioselectivity minimizes the formation of the undesired enantiomer, thereby reducing the burden on downstream purification processes. In traditional metal-catalyzed reactions, trace metal contamination is a persistent concern that requires additional scavenging steps, adding cost and complexity. In contrast, this organocatalytic system is metal-free, inherently simplifying the purification workflow and ensuring a cleaner final product. The mild reaction temperature of 25°C further contributes to impurity control by suppressing side reactions such as polymerization or decomposition that are often accelerated by heat. This mechanistic elegance translates directly into process robustness, making the technology highly attractive for GMP manufacturing environments where consistency and purity are paramount.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
To implement this synthesis effectively, operators should follow a standardized protocol that ensures reproducibility and optimal yield. The process begins with the careful selection of high-purity starting materials, specifically the indoleamine or pyrroleamine substrates and the 1,4-diketone derivatives. The reaction is typically conducted in carbon tetrachloride, although solvent optimization may be explored for specific substrates. The addition of activated 3A molecular sieves is crucial to sequester water generated during the condensation, shifting the equilibrium towards the desired product. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical documentation below.
- Prepare the reaction mixture by combining indoleamine or pyrroleamine substrates with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of chiral phosphoric acid catalyst (preferably spiro-based catalyst 6j).
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthesis method offers compelling advantages that align with the strategic goals of cost reduction and operational efficiency. The reliance on organocatalysis rather than precious metal complexes significantly lowers the raw material costs associated with the catalyst. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive testing and validation to ensure residual metal levels meet ICH guidelines. This streamlined compliance pathway accelerates the time-to-market for new drug candidates developed using this technology. Additionally, the reaction conditions are remarkably mild, occurring at room temperature without the need for specialized high-pressure or cryogenic equipment. This reduces the energy consumption of the manufacturing process and allows for the utilization of standard reactor infrastructure, facilitating easier technology transfer between sites.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in purification steps lead to substantial cost savings. The high atom economy of the condensation reaction ensures that a greater proportion of the starting materials are incorporated into the final product, minimizing waste disposal costs. Furthermore, the use of readily available and inexpensive solvents like carbon tetrachloride contributes to a lower overall cost of goods sold (COGS). These economic benefits make the commercial production of these complex chiral intermediates financially viable, even for early-stage clinical programs with tight budgets.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining a stable supply of key intermediates. The broad substrate scope means that supply chain disruptions for specific starting materials can often be mitigated by switching to alternative analogs without re-optimizing the entire process. The simplicity of the workup procedure, involving basic filtration and chromatography, reduces the risk of processing errors and delays. This reliability strengthens the supply chain resilience, ensuring that downstream drug substance manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: The method is inherently scalable, having been demonstrated to work efficiently across a wide range of substrate concentrations. The mild conditions reduce the safety risks associated with exothermic runaways, making it safer to operate on a large scale. From an environmental standpoint, the metal-free nature of the catalyst reduces the ecological footprint of the synthesis. While carbon tetrachloride usage requires careful handling and recycling, the overall green chemistry metrics are favorable due to the high yields and reduced waste generation. This alignment with sustainability goals is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this synthesis technology. These answers are derived directly from the experimental data and findings presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of incorporating these derivatives into their drug discovery pipelines.
Q: What is the primary advantage of using chiral phosphoric acid in this synthesis?
A: The use of chiral phosphoric acid, particularly the spiro-backbone derivative 6j, enables extremely high enantioselectivity (up to 96% ee) under mild room temperature conditions, eliminating the need for harsh reagents or transition metals.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological testing indicates that these derivatives possess strong cytotoxic activity against QGP-1 pancreatic tumor cells, suggesting significant potential for development as anticancer therapeutic agents.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes conventional reaction conditions, simple operation, and readily available substrates with high yields, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral phosphoric acid catalyzed synthesis for the development of novel anticancer therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to global supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess required for chiral pharmaceutical intermediates. We are committed to delivering high-quality N-N axis chiral pyrrole derivatives that meet the exacting standards of the international pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug discovery program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate the development of life-saving medicines by overcoming the synthetic challenges of complex chiral architectures.
