Advanced Silver-Catalyzed Acylation of 2-Methylquinolines for Scalable Pharmaceutical Intermediate Production
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for greener, more efficient synthetic routes for high-value pharmaceutical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN115108980B, which details a novel preparation method for 4-position acylated derivatives of 2-methylquinoline compounds. This technology represents a paradigm shift from traditional radical functionalization methods, leveraging a unique combination of silver nitrate catalysis and Selectfluor oxidation within an aqueous medium. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenges of regioselectivity and environmental compliance in quinoline functionalization. By utilizing water as the primary solvent, the process not only enhances the solubility of polar substrates but also drastically reduces the reliance on volatile organic compounds (VOCs), aligning perfectly with modern green chemistry principles. The robustness of this method is evidenced by its ability to tolerate a wide range of functional groups, making it a versatile tool for the synthesis of complex drug candidates and agrochemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acylated quinoline derivatives has relied heavily on Minisci-type reactions or transition-metal catalyzed C-H activation strategies that often suffer from significant operational drawbacks. Traditional approaches frequently necessitate the use of hazardous organic solvents, high reaction temperatures, and expensive or toxic radical initiators such as di-tert-butyl peroxide (DTBP) or persulfates. As noted in the background art of the patent, previous methods developed by researchers like Sutherland utilized iron catalysts with triethyl chloroformate and TBHP, yet these processes were plagued by lower yields and the generation of substantial by-products due to the harsh thermal conditions required. Furthermore, the use of stoichiometric amounts of strong oxidants in organic media often leads to difficult work-up procedures and complex impurity profiles that complicate downstream purification. These limitations pose severe bottlenecks for procurement managers and supply chain heads, as the cost of waste disposal and the risk of batch-to-batch variability increase significantly when scaling up these conventional protocols.
The Novel Approach
In stark contrast, the methodology disclosed in CN115108980B introduces a streamlined, environmentally benign pathway that operates efficiently in water. The core innovation lies in the synergistic use of silver nitrate as a catalyst and Selectfluor as a mild yet effective oxidant. This combination facilitates the generation of acyl radicals from simple aldehydes under remarkably mild conditions, typically between 30°C and 50°C. The aqueous environment not only serves as a green solvent but also plays a crucial role in stabilizing the ionic intermediates involved in the catalytic cycle. This approach eliminates the need for flammable organic solvents during the reaction phase, thereby enhancing process safety and reducing fire hazards in manufacturing facilities. The versatility of this system is demonstrated by its compatibility with various aldehydes, including n-butyraldehyde, isobutyraldehyde, and benzaldehyde, allowing for the rapid diversification of the quinoline scaffold.
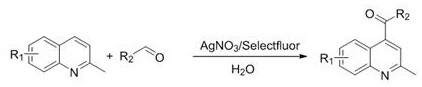
Mechanistic Insights into AgNO3-Catalyzed Radical Acylation
From a mechanistic perspective, the reaction proceeds through a sophisticated radical pathway mediated by the silver catalyst. The process initiates with the interaction between Selectfluor and the aldehyde, likely facilitated by the silver species, to generate an acyl radical species. This radical then attacks the electron-deficient quinoline ring. The presence of the silver ion is critical, as it likely coordinates with the nitrogen atom of the quinoline, thereby activating the ring towards nucleophilic radical attack specifically at the 4-position. This coordination effect explains the high regioselectivity observed in the patent examples, where substitution occurs exclusively at the C4 position despite the presence of other potential reactive sites. The subsequent oxidation and deprotonation steps restore the aromaticity of the quinoline system, yielding the desired 4-acylated product. Understanding this mechanism is vital for R&D teams aiming to optimize the process further, as it highlights the importance of maintaining the correct stoichiometry between the silver catalyst and the oxidant to prevent over-oxidation or catalyst deactivation.
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional high-temperature radical reactions, non-selective hydrogen abstraction often leads to a myriad of side products, including oligomers and over-oxidized species. However, the mild thermal profile of the silver-catalyzed aqueous system minimizes these parasitic pathways. The patent data indicates that side reactions are significantly suppressed, resulting in a cleaner crude reaction mixture. This purity profile is particularly beneficial for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metals are enforced. By avoiding the use of transition metals like copper or iron which can be difficult to remove to ppm levels, this silver-based method simplifies the purification train. Although silver is a precious metal, its usage in catalytic amounts (0.2 to 1.0 equivalents relative to substrate, optimized at 0.25) combined with the high yield ensures that the overall cost of goods remains competitive while delivering a superior quality product.
How to Synthesize 4-Acylated 2-Methylquinolines Efficiently
Implementing this synthesis route requires careful attention to reagent quality and mixing parameters to ensure reproducibility on a larger scale. The protocol involves dissolving the 2-methylquinoline substrate and the chosen aldehyde in water, followed by the addition of the silver nitrate catalyst and Selectfluor oxidant. The reaction mixture is then stirred at a controlled temperature, typically around 30°C, for a period of 20 to 24 hours to ensure complete conversion. Post-reaction processing involves a standard extraction workflow using ethyl acetate, followed by washing with sodium bicarbonate to neutralize any acidic by-products. The detailed standardized synthesis steps for laboratory and pilot-scale execution are outlined below.
- Mix the 2-methylquinoline substrate with the desired aldehyde (e.g., n-butyraldehyde or benzaldehyde) in an aqueous solution containing silver nitrate catalyst.
- Add Selectfluor as the oxidant to the reaction mixture and maintain the temperature between 30°C and 50°C for approximately 20 to 24 hours.
- Upon completion, extract the product using ethyl acetate, dry the organic layer, and purify the final 4-acylated derivative via column chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this technology offers tangible benefits that extend beyond mere chemical elegance. The shift to an aqueous-based system fundamentally alters the cost structure of manufacturing these valuable intermediates. By replacing expensive and regulated organic solvents with water, the direct material costs are significantly reduced, and the logistical burden of handling hazardous liquids is minimized. Furthermore, the high selectivity of the reaction means that less raw material is wasted on forming by-products, leading to improved atom economy. This efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the yield losses associated with purification are curtailed. The simplicity of the reagent list—requiring only commercially available aldehydes, silver nitrate, and Selectfluor—ensures a robust and resilient supply chain that is less susceptible to disruptions caused by specialty chemical shortages.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents as the reaction medium drastically cuts down on solvent purchase and recovery costs. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to substantial cost savings in utility expenses. The high yield reported in the patent examples suggests that the process is highly efficient, minimizing the cost per kilogram of the final active ingredient precursor.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as silver nitrate and common aldehydes, are commodity chemicals with stable global supply chains. This contrasts with specialized radical initiators or exotic ligands that may have long lead times. By relying on widely available materials, manufacturers can secure long-term supply contracts and reduce the risk of production stoppages due to raw material unavailability.
- Scalability and Environmental Compliance: Scaling up exothermic radical reactions in organic solvents often presents significant safety challenges. Conducting the reaction in water provides a natural heat sink, making the process inherently safer and easier to scale from grams to tons. Moreover, the reduction in organic waste generation simplifies compliance with increasingly stringent environmental regulations, avoiding costly fines and permitting delays associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed acylation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations for potential licensees or manufacturing partners.
Q: What are the optimal reaction conditions for this acylation process?
A: According to patent CN115108980B, the optimal conditions involve using water as the solvent, a reaction temperature of 30°C, and a molar ratio of substrate to aldehyde of 1:3.0 with silver nitrate (0.25 eq) and Selectfluor (4 eq).
Q: Does this method support diverse substrate scopes?
A: Yes, the method demonstrates broad applicability, successfully accommodating various substituents on the quinoline ring such as halogens (F, Cl, Br), nitro groups, and trifluoromethyl groups, as well as different aliphatic and aromatic aldehydes.
Q: How does this aqueous method compare to traditional organic synthesis?
A: Unlike traditional Minisci reactions that often require harsh temperatures and organic solvents, this silver-catalyzed aqueous method operates under mild conditions (30-50°C), significantly reducing environmental impact and improving safety profiles for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN115108980B for the production of high-value heterocyclic building blocks. As a leading CDMO partner, we possess the technical expertise to translate these laboratory-scale innovations into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We understand that the transition from bench to plant requires rigorous optimization, and our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. Whether you require custom synthesis of complex quinoline derivatives or process development services to optimize this specific acylation route, our infrastructure is designed to support your growth.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data for similar quinoline intermediates and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner committed to delivering quality, efficiency, and innovation in every batch.
