Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharma Production
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharma Production
The pharmaceutical industry is constantly seeking robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112209947A introduces a groundbreaking synthetic pathway for chiral indoxazinone compounds, a class of molecules exhibiting significant cytotoxic activity against PC-3 human prostate cancer cells. This innovation addresses the longstanding challenges associated with synthesizing chiral indole-fused ring systems, which have historically suffered from low enantioselectivity and harsh reaction conditions. By leveraging a dual catalytic system involving chiral phosphoric acids and hexafluoroisopropanol, this technology enables the efficient assembly of these complex architectures under remarkably mild thermal conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more sustainable, and highly selective manufacturing processes for high-purity pharmaceutical intermediates.
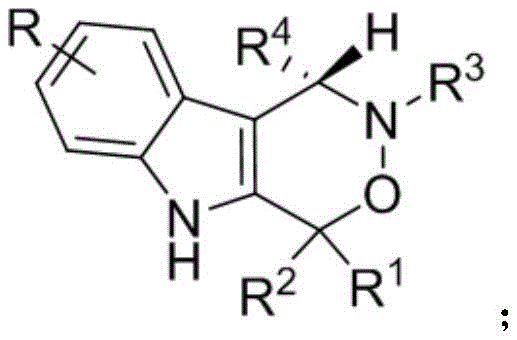
The core innovation lies in the structural versatility and biological potential of the resulting indoxazinone scaffold. As illustrated in the general formula, the method accommodates a wide range of substituents, allowing for the generation of diverse libraries of compounds essential for structure-activity relationship (SAR) studies. The ability to introduce various alkyl, alkoxy, halogen, and aryl groups at multiple positions on the indole and oxazinone rings provides medicinal chemists with unprecedented flexibility. This structural diversity is crucial for optimizing pharmacokinetic properties and enhancing binding affinity to biological targets such as the S1P1 receptor or androgen receptors. Consequently, this synthesis method not only delivers a specific compound but offers a platform technology for the rapid discovery of novel anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral indole-fused heterocycles has been plagued by significant operational and economic inefficiencies that hinder commercial viability. Traditional routes often rely on stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts, which necessitate rigorous and costly purification steps to remove trace metal residues to meet regulatory standards for API manufacturing. Furthermore, many existing protocols require cryogenic temperatures or highly reactive reagents that pose substantial safety risks in a production environment. These harsh conditions frequently lead to side reactions, resulting in complex impurity profiles that are difficult to separate, thereby reducing overall yield and increasing waste generation. For supply chain managers, these factors translate into longer lead times, higher raw material costs, and increased liability regarding hazardous waste disposal.
The Novel Approach
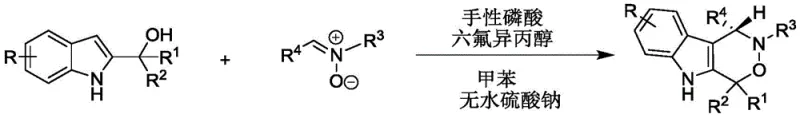
In stark contrast, the methodology disclosed in CN112209947A employs an elegant organocatalytic strategy that circumvents the drawbacks of metal-mediated chemistry. By utilizing a chiral phosphoric acid in conjunction with hexafluoroisopropanol (HFIP) as a co-catalyst, the reaction proceeds with exceptional stereocontrol at near-ambient temperatures ranging from 20-30°C. This mild thermal profile drastically reduces energy consumption and eliminates the need for specialized cryogenic equipment, making the process inherently safer and more energy-efficient. The use of toluene as a solvent and anhydrous sodium sulfate as an additive further simplifies the reaction setup, ensuring high atom economy and operational simplicity. This approach not only achieves high yields but also ensures excellent enantiomeric excess, directly addressing the purity requirements demanded by modern regulatory bodies for chiral drug substances.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
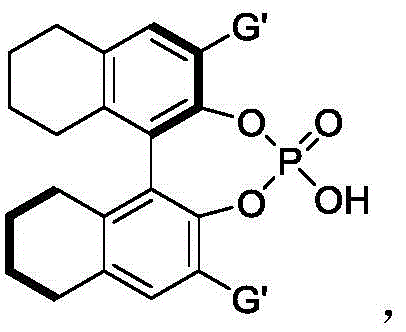
The success of this transformation is rooted in the sophisticated interplay between the chiral phosphoric acid catalyst and the hydrogen-bonding donor capability of hexafluoroisopropanol. The chiral phosphoric acid acts as a Brønsted acid, activating the electrophilic nitrone species through hydrogen bonding interactions, while simultaneously organizing the nucleophilic 2-indolemethanol within a well-defined chiral pocket. This dual activation mode lowers the activation energy of the cyclization step, facilitating the formation of the new C-N and C-O bonds with high fidelity. The bulky substituents on the binaphthyl or spiro backbone of the catalyst, such as the 9-phenanthryl group shown in preferred embodiments, create a steric environment that effectively discriminates between the prochiral faces of the substrate. This precise spatial arrangement is critical for achieving the observed high levels of enantioselectivity, ensuring that the desired enantiomer is produced predominantly over its mirror image.
Furthermore, the inclusion of hexafluoroisopropanol plays a synergistic role in stabilizing the transition state and enhancing the acidity of the phosphoric acid catalyst through hydrogen bonding networks. This cooperative catalysis accelerates the reaction kinetics without compromising selectivity, allowing the process to reach completion within reasonable timeframes even at lower temperatures. From an impurity control perspective, this mechanism minimizes the formation of racemic byproducts and oligomeric species that are common in less controlled radical or metal-catalyzed pathways. The result is a cleaner crude reaction mixture, which significantly reduces the burden on downstream purification processes such as column chromatography or crystallization. For quality control teams, this translates to a more consistent product profile and reduced risk of batch-to-batch variability in critical quality attributes.
How to Synthesize Chiral Indoxazinone Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the reaction mixture by combining the 2-indolemethanol substrate and the nitrone coupling partner in toluene, followed by the addition of anhydrous sodium sulfate to manage water content. The catalytic system is then introduced, and the mixture is stirred under controlled thermal conditions until TLC analysis indicates complete consumption of the starting materials. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are outlined in the technical guide below to ensure reproducibility and optimal performance.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone in toluene with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol as co-catalysts to initiate the asymmetric cyclization.
- Stir the reaction at 20-30°C until completion, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant cost driver associated with both the initial purchase of reagents and the subsequent validation of metal clearance in the final product. Additionally, the use of commodity solvents like toluene and the avoidance of extreme temperatures simplify the engineering controls required for production, leading to substantial capital expenditure savings when scaling up from pilot to commercial manufacturing. The robustness of the reaction across a wide range of substrates ensures supply continuity, as alternative raw materials can be sourced without necessitating a complete re-optimization of the process parameters.
- Cost Reduction in Manufacturing: The transition from metal-based catalysis to organocatalysis fundamentally alters the cost structure of API intermediate production by removing the need for expensive ligands and metal scavengers. This shift not only lowers the direct material costs but also reduces the operational expenses related to waste treatment and environmental compliance, as the process generates less hazardous effluent. Furthermore, the high atom economy of the reaction means that a greater proportion of the input mass is converted into valuable product, minimizing raw material waste and maximizing overall process efficiency.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as chiral phosphoric acids and hexafluoroisopropanol mitigates the risk of supply disruptions often associated with specialized organometallic complexes. The mild reaction conditions also reduce the wear and tear on production equipment, extending asset life and minimizing unplanned downtime due to maintenance. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical customers who depend on just-in-time inventory models for their clinical and commercial programs.
- Scalability and Environmental Compliance: The inherent safety of operating at 20-30°C significantly lowers the risk of thermal runaway incidents, making the process easier to scale from kilogram to multi-ton quantities without extensive safety modifications. The reduced environmental footprint, characterized by lower energy usage and the absence of toxic heavy metals, aligns perfectly with the increasingly stringent sustainability goals of global pharmaceutical companies. This alignment facilitates faster regulatory approvals and enhances the marketability of the final drug product as a green chemistry achievement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral indoxazinone compounds. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing development pipelines.
Q: What represents the primary advantage of this synthesis method over traditional routes?
A: The primary advantage is the use of mild reaction conditions (20-30°C) and organocatalysis, which eliminates the need for toxic transition metals and harsh thermal inputs, resulting in higher safety and lower environmental impact.
Q: What level of enantioselectivity can be achieved with this protocol?
A: The protocol utilizes a specific chiral phosphoric acid catalyst combined with hexafluoroisopropanol to achieve extremely high enantioselectivity, with experimental data showing ee values reaching up to 96%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for scalability, utilizing common solvents like toluene and avoiding cryogenic temperatures, which simplifies reactor requirements and facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of novel anticancer therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest international standards for pharmaceutical ingredients. Our facility is equipped to handle complex organocatalytic reactions, providing a seamless transition from process development to full-scale manufacturing.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your timeline to market while optimizing your overall production costs.
