Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
The rapid evolution of medicinal chemistry demands robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in the synthesis of these pharmacologically privileged structures. This innovation leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate in an air atmosphere. For R&D directors and procurement specialists alike, this technology represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates. The ability to generate these core structures without heavy metal catalysts not only streamlines purification but also aligns with stringent global regulatory standards for residual impurities in active pharmaceutical ingredients.
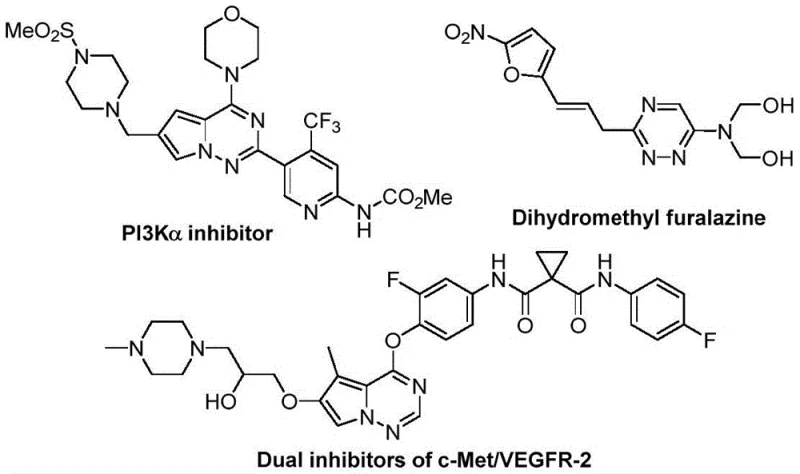
The strategic incorporation of the trifluoromethyl group into the 1,2,4-triazine core significantly modulates the electronic properties and lipophilicity of the resulting molecules, which is crucial for optimizing drug-receptor interactions. As illustrated in the provided chemical structures, these scaffolds are foundational to a wide array of therapeutic agents, including PI3K inhibitors and dual c-Met/VEGFR-2 inhibitors, underscoring their immense commercial potential. The disclosed method offers a direct pathway to these valuable motifs, bypassing the multi-step sequences often required by conventional approaches. By utilizing readily available starting materials and avoiding sensitive reaction conditions, this process enhances the reliability of the supply chain for critical drug candidates, ensuring that development timelines are not compromised by synthetic complexity or raw material scarcity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazine rings has relied heavily on condensation reactions between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. These traditional pathways frequently suffer from significant drawbacks, including the necessity for harsh reaction conditions, prolonged heating, and the use of stoichiometric amounts of dehydrating agents or strong acids. Furthermore, the structural diversity achievable through these methods is often limited, restricting the ability of chemists to rapidly explore structure-activity relationships (SAR) during lead optimization phases. The reliance on sensitive substrates that require rigorous exclusion of moisture and oxygen further complicates scale-up efforts, leading to inconsistent batch-to-batch reproducibility and increased operational costs due to the need for specialized inert atmosphere equipment.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a mild, metal-free catalytic system that operates efficiently at room temperature under ambient air conditions. The core transformation involves the reaction of a chlorohydrazone derivative with a trifluoroacetyl sulfur ylide in the presence of potassium carbonate, as depicted in the general reaction scheme below. This approach not only simplifies the operational procedure by eliminating the need for nitrogen protection but also drastically reduces the energy consumption associated with heating or cooling the reaction vessel. The use of tetrahydrofuran as a solvent ensures excellent solubility of the reactants, facilitating high conversion rates and minimizing the formation of side products. This streamlined process exemplifies how modern synthetic design can overcome the inefficiencies of legacy chemistry to deliver superior outcomes in both yield and purity.
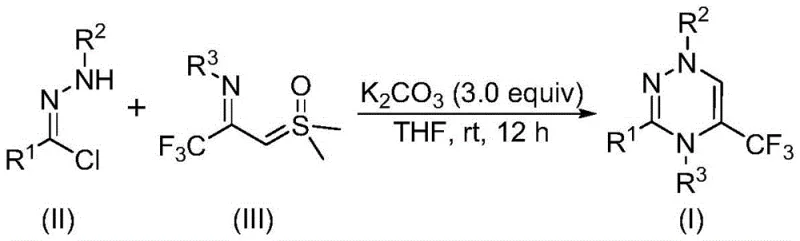
Mechanistic Insights into Potassium Carbonate-Promoted Cycloaddition
The mechanistic elegance of this transformation lies in the in situ generation of a reactive nitrile imine intermediate from the chlorohydrazone precursor. Under the promotion of potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to release hydrogen chloride, generating the highly electrophilic nitrile imine species. This intermediate then engages in a concerted [3+3] cycloaddition with the nucleophilic trifluoroacetyl sulfur ylide. The reaction proceeds through a transition state that allows for the simultaneous formation of two new carbon-nitrogen bonds, constructing the six-membered triazine ring with high regioselectivity. The subsequent elimination of dimethyl sulfoxide drives the reaction forward, yielding the final aromatic 1,2,4-triazine product. This mechanism avoids the formation of unstable intermediates that often plague other cyclization strategies, thereby ensuring a clean reaction profile that is amenable to large-scale production.
From an impurity control perspective, the absence of transition metal catalysts is a decisive advantage for pharmaceutical manufacturing. Traditional metal-catalyzed couplings often leave behind trace amounts of palladium, copper, or nickel, which require extensive and costly purification steps such as scavenging or recrystallization to meet ICH Q3D guidelines. In this metal-free protocol, the only inorganic byproduct is potassium chloride, which is easily removed during the aqueous workup or filtration steps. The structural diversity of the products is further evidenced by the successful synthesis of various derivatives, including those with electron-donating methoxy groups and electron-withdrawing halogens, as shown in the specific examples. This broad substrate scope confirms the robustness of the mechanistic pathway, allowing for the facile introduction of diverse functional handles for downstream derivatization without compromising the integrity of the triazine core.
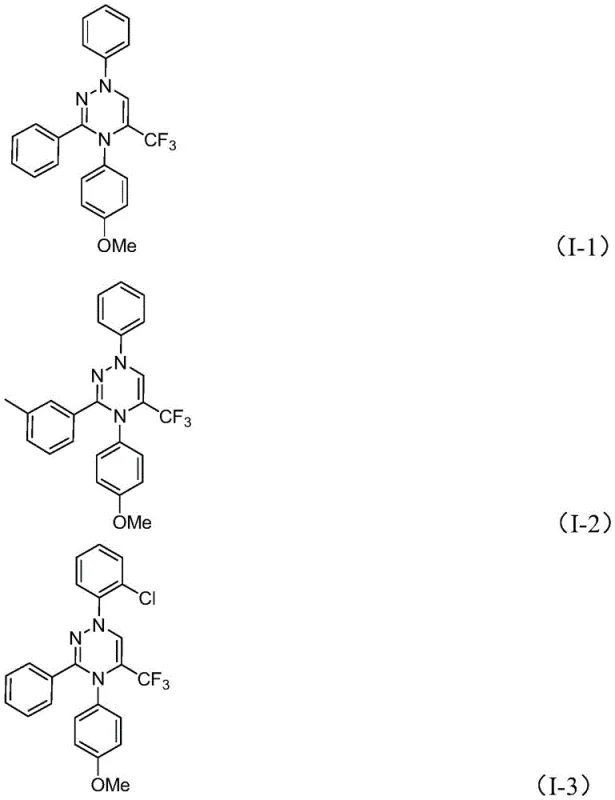
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
The practical implementation of this synthesis is straightforward and designed for immediate adoption in process chemistry laboratories. The protocol requires the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, which are mixed with three equivalents of potassium carbonate in tetrahydrofuran. The reaction mixture is stirred at ambient temperature for a period of 10 to 14 hours, after which completion is monitored via standard analytical techniques such as TLC or HPLC. Upon confirmation of full conversion, the solid inorganic salts are removed by filtration, and the filtrate is concentrated. The crude product is then purified using standard silica gel column chromatography to afford the target compound in high purity. For detailed standardized operating procedures and safety data sheets regarding the handling of sulfur ylides and chlorohydrazones, please refer to the specific technical documentation provided by your chemical supplier.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as tetrahydrofuran.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts directly translates to a reduction in raw material costs, as there is no longer a need to purchase expensive palladium or rhodium complexes. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during the cleanup phase, leading to substantial cost savings in waste disposal and material usage. The ability to run the reaction under air atmosphere removes the dependency on nitrogen or argon gas supplies and the associated infrastructure for maintaining inert environments, thereby lowering the overhead costs of production facilities. These factors combined create a leaner, more efficient manufacturing process that enhances the overall margin profile of the final API intermediate.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive, commodity-grade potassium carbonate results in a drastic reduction in catalyst costs. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating or cryogenic cooling, significantly lowering utility expenses. The simplified workup procedure reduces the labor hours required for purification and minimizes solvent consumption, contributing to a lower cost of goods sold (COGS) for the final product.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and sulfur ylides, are derived from commercially available precursors such as acyl chlorides, hydrazines, and trifluoroacetic acid derivatives, ensuring a stable and diversified supply base. The robustness of the reaction against moisture and oxygen means that production is less susceptible to delays caused by equipment failure or environmental fluctuations. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risk of stockouts in critical drug development pipelines.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the regulatory approval process for new drug applications, as there is no need to validate complex metal removal steps. The process generates minimal hazardous waste, primarily consisting of benign inorganic salts and organic solvents that can be readily recycled. This alignment with green chemistry principles facilitates easier permitting for manufacturing sites and supports corporate sustainability goals, making it an attractive option for environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and application of this trifluoromethyl triazine synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for process engineers and quality assurance teams. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows and for ensuring compliance with internal quality standards.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild room temperature and air conditions, and utilizes inexpensive inorganic bases like potassium carbonate, significantly simplifying post-treatment and reducing environmental impact.
Q: What is the typical reaction time and temperature for this process?
A: The reaction proceeds efficiently at temperatures between 20°C and 40°C, typically requiring 10 to 14 hours to reach completion, making it highly suitable for standard batch processing without specialized heating or cooling equipment.
Q: Can this method accommodate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully synthesizing derivatives with substituents such as methyl, methoxy, chloro, bromo, and trifluoromethyl groups on phenyl and naphthyl rings, ensuring broad applicability in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug discovery programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethyl 1,2,4-triazine delivered meets the highest industry standards. Our commitment to excellence extends beyond mere supply; we act as a strategic partner, offering technical support to optimize your synthesis routes and resolve any process challenges that may arise during scale-up.
We invite you to contact our technical procurement team to discuss your specific project needs and to request a Customized Cost-Saving Analysis tailored to your production volumes. By leveraging our expertise in metal-free heterocycle synthesis, we can help you accelerate your timeline to market while optimizing your budget. Please reach out to us today to obtain specific COA data and route feasibility assessments for your target compounds, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
