Scalable Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Applications
Scalable Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Applications
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in the production of these valuable pharmacophores. The incorporation of the trifluoromethyl group is strategically significant, as it markedly enhances the metabolic stability, lipophilicity, and bioavailability of the parent heterocyclic molecule, making these compounds highly desirable candidates for anticancer, antifungal, and antihypertensive drug development. This novel methodology distinguishes itself by operating under exceptionally mild conditions, utilizing inexpensive inorganic salts rather than precious metal catalysts, thereby offering a robust platform for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied heavily on condensation reactions between amidrazones and 1,2-diketones or alkynes, often necessitating multicomponent reactions involving hydrazides and ammonium acetate. These traditional pathways are frequently plagued by significant operational drawbacks, including the requirement for harsh reaction conditions, prolonged heating, and the generation of complex impurity profiles that complicate downstream purification. Furthermore, the structural diversity achievable through these classical methods is often limited, restricting the ability of R&D teams to rapidly explore structure-activity relationships (SAR) around the triazine scaffold. The reliance on specific, sometimes unstable substrates further exacerbates supply chain vulnerabilities, making the consistent production of high-purity intermediates a challenging endeavor for large-scale manufacturers.
The Novel Approach
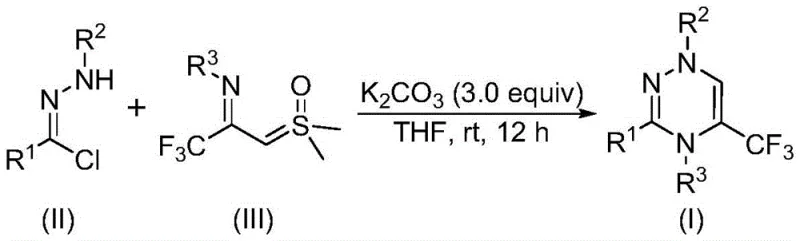
In stark contrast to legacy techniques, the disclosed invention leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl thio ylides, catalyzed by the ubiquitous and non-toxic base potassium carbonate. This approach fundamentally shifts the paradigm by enabling the reaction to proceed at room temperature in an open air atmosphere, completely eliminating the need for expensive inert gas protection systems or energy-intensive heating protocols. The use of readily available starting materials, such as acyl chlorides and hydrazines, ensures a stable and cost-effective supply chain, while the absence of heavy metals removes the regulatory burden associated with residual metal limits in active pharmaceutical ingredients. This streamlined process not only accelerates the timeline from bench to pilot plant but also significantly reduces the environmental footprint of the synthesis.
Mechanistic Insights into Potassium Carbonate-Promoted Cycloaddition
The mechanistic elegance of this transformation lies in the in situ generation of a highly reactive nitrile imine intermediate, which serves as the 1,3-dipole in the subsequent cycloaddition event. Upon treatment with potassium carbonate, the chlorohydrazone precursor undergoes a dehydrohalogenation reaction, eliminating hydrogen chloride to form the transient nitrile imine species. This intermediate then engages in a concerted [3+3] cycloaddition with the trifluoroacetyl thio ylide, a process that constructs the six-membered triazine ring with high regioselectivity. The final step involves the elimination of dimethyl sulfoxide (DMSO) from the adduct, driving the equilibrium towards the formation of the aromatic trifluoromethyl substituted 1,2,4-triazine product. This cascade sequence is remarkably efficient, minimizing the formation of side products and ensuring a clean reaction profile that is ideal for industrial application.
From an impurity control perspective, the choice of potassium carbonate as the promoter is critical, as stronger bases might induce decomposition of the sensitive thio ylide or promote unwanted side reactions on the electron-deficient triazine ring. The mild basicity ensures that functional groups such as esters, halides, and ethers remain intact throughout the synthesis, preserving the integrity of complex molecular architectures. Additionally, the reaction's tolerance to moisture and oxygen suggests that the nitrile imine intermediate is consumed rapidly by the ylide, preventing hydrolysis or oxidation pathways that typically degrade yield in similar dipolar cycloadditions. This robustness allows for the synthesis of a wide array of derivatives, including those with sensitive substituents, without compromising the overall purity of the final isolated material.
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators should adhere to the optimized protocol outlined in the patent, which balances reagent stoichiometry with practical handling considerations. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl thio ylide, typically in a molar ratio favoring the ylide to drive the reaction to completion, alongside three equivalents of potassium carbonate to ensure full conversion. These solids are suspended in an aprotic organic solvent such as tetrahydrofuran (THF), which provides excellent solubility for the reactants while facilitating the necessary ionization of the base. The detailed standardized synthesis steps see the guide below.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl thio ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this metal-free synthesis protocol represents a strategic opportunity to enhance operational resilience and reduce total cost of ownership. By eliminating the dependency on transition metal catalysts such as palladium or copper, manufacturers can bypass the costly and time-consuming processes associated with metal scavenging and validation of residual metal levels, which are strictly regulated under ICH Q3D guidelines. This simplification of the purification workflow translates directly into reduced processing time and lower consumption of specialized adsorbents, resulting in substantial cost savings in API manufacturing without sacrificing product quality or yield.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with commodity-grade potassium carbonate drastically lowers the raw material costs per kilogram of product. Furthermore, the ability to run the reaction at room temperature eliminates the energy expenditures associated with heating or cooling large-scale reactors, contributing to a more sustainable and economically viable production model. The simplified workup procedure, which often requires only filtration and standard chromatography, reduces solvent usage and waste disposal costs, enhancing the overall green chemistry profile of the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted acyl chlorides, hydrazines, and trifluoroacetic acid derivatives, are widely available from global chemical suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which do not require stringent exclusion of air or moisture, allows for greater flexibility in manufacturing scheduling and reduces the likelihood of batch failures due to equipment leaks or atmospheric contamination. This reliability ensures a consistent flow of high-purity intermediates to downstream formulation teams, supporting uninterrupted drug development timelines.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration on gram scales with consistent yields, indicating a smooth path to kilogram and tonne-level production. The absence of toxic heavy metals and the use of relatively benign solvents align with increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous waste management. This eco-friendly profile not only facilitates faster regulatory approval but also enhances the corporate sustainability metrics of the manufacturing organization, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this trifluoromethyl triazine synthesis route. These insights are derived directly from the experimental data and scope limitations defined within the patent literature, providing a realistic framework for process development teams evaluating this technology for adoption.
Q: Is a heavy metal catalyst required for this triazine synthesis?
A: No, the patented process explicitly avoids the use of heavy metal catalysts, utilizing potassium carbonate as a benign promoter instead, which simplifies purification and regulatory compliance.
Q: What are the optimal reaction conditions for this cycloaddition?
A: The reaction proceeds efficiently at room temperature (20-40°C) under an air atmosphere for 10-14 hours, eliminating the need for inert gas protection or cryogenic cooling.
Q: Can this method accommodate diverse substrate functional groups?
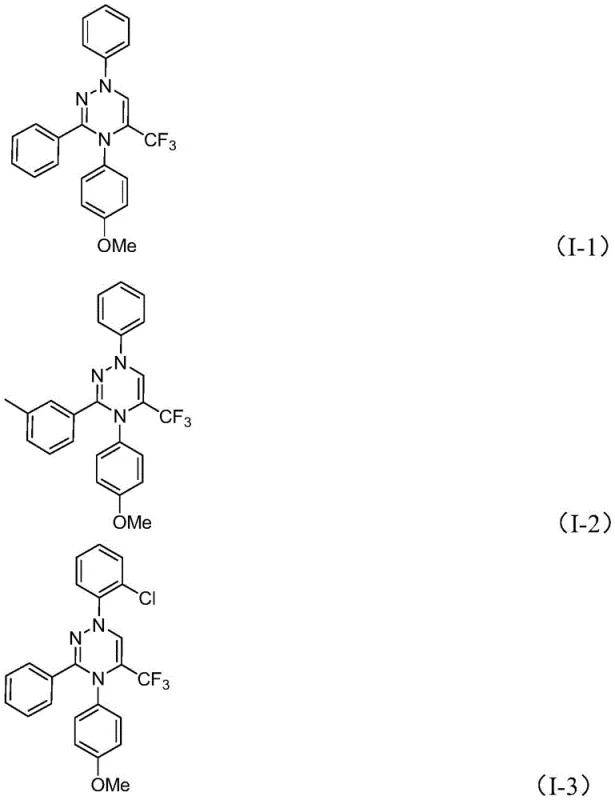
A: Yes, the method demonstrates high tolerance for various substituents including alkyl, alkoxy, halogens, and nitro groups on the phenyl rings, allowing for broad structural diversity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic building blocks play in the discovery and development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to market launch. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles according to international pharmacopoeia standards.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific pipeline needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines while optimizing your budget.
