Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as the backbone for potent bioactive molecules. Patent CN113045489B introduces a groundbreaking methodology for the preparation of 3-arylquinolin-2(1H)one derivatives, a class of compounds renowned for their diverse biological activities ranging from antibiotic to antitumor properties. This innovation leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm of quinolinone synthesis by utilizing benzisoxazole as a dual-purpose reagent. Unlike traditional methods that often rely on hazardous gases or multi-step sequences, this approach integrates nitrogen insertion and carbonylation into a single, streamlined operation. The significance of this technology lies in its ability to access complex pharmacophores with high atom economy and operational simplicity. As depicted in the structural diversity of known bioactive quinolinones, the demand for efficient access to these cores is critical for drug discovery pipelines.
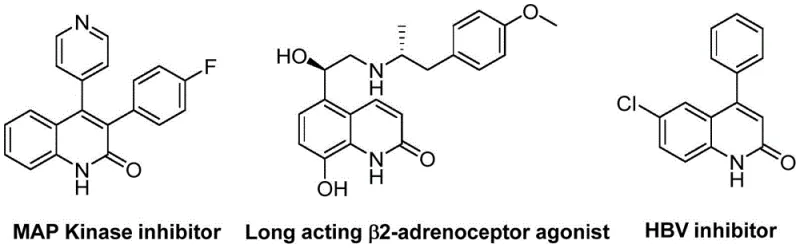
The strategic value of this patent extends beyond mere academic interest; it offers a tangible solution for reliable pharmaceutical intermediate supplier networks aiming to reduce dependency on volatile supply chains. By establishing a pathway that tolerates a wide array of functional groups, the method ensures that medicinal chemists can rapidly iterate on lead compounds without being constrained by synthetic limitations. The use of readily available benzyl chlorides further democratizes access to these valuable intermediates, positioning this technology as a cornerstone for cost reduction in API manufacturing. For procurement and R&D teams alike, understanding the mechanistic underpinnings and commercial viability of this process is essential for integrating it into future production schedules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one scaffold has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, and Friedlander syntheses. While these methods are well-documented, they suffer from significant drawbacks that hinder their applicability in modern, green chemistry-focused manufacturing environments. The Vilsmeier-Haack reaction, for instance, typically necessitates the use of phosphorus oxychloride (POCl3), a highly corrosive and moisture-sensitive reagent that generates substantial amounts of acidic waste, complicating downstream processing and environmental compliance. Furthermore, these traditional routes often require harsh thermal conditions and strong acids or bases, which can lead to the degradation of sensitive functional groups present on the substrate. Such limitations restrict the chemical space that can be explored, forcing researchers to protect and deprotect groups, thereby increasing step counts and reducing overall yield. Additionally, the reliance on specific aniline precursors can create bottlenecks in the supply chain if those specific starting materials are not commodity chemicals.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113045489B circumvents these historical inefficiencies by employing a transition metal-catalyzed carbonylation strategy. This method utilizes benzisoxazole not merely as a reactant but as a clever surrogate for both nitrogen and carbon monoxide equivalents. By reacting benzisoxazole with benzyl chloride derivatives in the presence of a palladium catalyst system, the process achieves the formation of the quinolinone core under relatively mild conditions. The elimination of gaseous carbon monoxide, which is toxic and requires specialized high-pressure equipment, represents a major safety and logistical advancement. Instead, the carbonyl group is sourced from molybdenum hexacarbonyl or potentially derived in situ, allowing the reaction to proceed in standard sealed vessels. This shift from corrosive condensations to catalytic coupling significantly enhances the functional group tolerance, enabling the direct synthesis of derivatives with halogens, ethers, and nitriles without the need for extensive protection strategies. The result is a cleaner, safer, and more versatile synthetic route that aligns perfectly with the goals of sustainable chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate dance of the palladium catalytic cycle, which facilitates the simultaneous cleavage of the benzisoxazole ring and the insertion of the carbonyl moiety. The reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step forms a benzyl-palladium(II) complex, which is then poised for carbonyl insertion. Uniquely, the benzisoxazole acts as a masked isocyanate or equivalent species; under the reaction conditions, the N-O bond of the benzisoxazole is cleaved, likely facilitated by the metal center or the basic environment provided by triethylamine. This cleavage releases the necessary nitrogen atom and the formyl group required to close the ring. The subsequent migratory insertion of the carbonyl group into the palladium-carbon bond, followed by nucleophilic attack by the nitrogen species, leads to the formation of the cyclic amide structure. Water plays a crucial role in this mechanism, potentially assisting in the hydrolysis of intermediate imines or stabilizing transition states, ensuring the reaction proceeds to the thermodynamically stable quinolinone product.
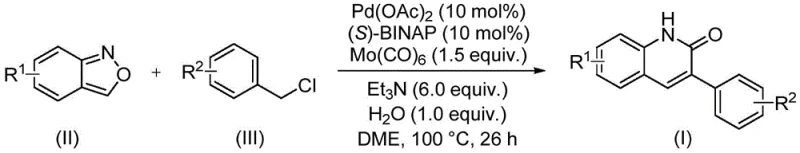
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensations. The specificity of the palladium catalyst minimizes side reactions such as polymerization or non-selective alkylation that often plague Friedlander-type reactions. The use of (S)-BINAP as a ligand not only stabilizes the palladium center but also imposes steric constraints that favor the desired coupling pathway over homocoupling of the benzyl chloride. Furthermore, the reaction conditions—specifically the temperature of 100°C and the use of DME as a solvent—are optimized to balance reaction kinetics with selectivity. The data indicates that extending the reaction time beyond 26 hours does not significantly improve conversion but may increase costs, suggesting a well-defined kinetic window. This precise control over the reaction parameters ensures a high-purity crude profile, reducing the burden on downstream purification units and enhancing the overall mass balance of the process. For R&D directors, this level of mechanistic clarity translates to predictable scale-up behavior and robust quality control.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to maximize yield and reproducibility. The process is designed to be operationally simple, involving the mixing of solid and liquid reagents in a common organic solvent followed by heating. The key to success lies in the precise stoichiometric balance of the catalyst system and the careful management of the reaction atmosphere to prevent catalyst deactivation. While the general procedure is straightforward, attention to detail regarding the quality of the benzisoxazole and the dryness of the solvent can influence the initiation of the catalytic cycle. The following guide summarizes the standardized steps derived from the experimental examples, providing a clear roadmap for technical teams to replicate the high efficiencies reported in the intellectual property.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The primary driver for cost optimization is the selection of starting materials; benzyl chlorides and benzisoxazoles are commodity chemicals available from multiple global suppliers, ensuring a competitive pricing landscape and reducing the risk of single-source dependency. Unlike processes that require custom-synthesized anilines or unstable diazonium salts, the inputs for this reaction are shelf-stable and easy to transport. This stability simplifies inventory management and allows for just-in-time purchasing strategies, which are critical for maintaining lean operations in a volatile market. Furthermore, the elimination of hazardous reagents like phosphorus oxychloride removes the need for specialized corrosion-resistant reactors and extensive scrubbing systems for acidic off-gases. This reduction in infrastructure requirements directly lowers the capital expenditure (CAPEX) needed for production lines and decreases the operational expenditure (OPEX) associated with waste treatment and regulatory compliance.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy and the avoidance of expensive protecting group chemistry. By tolerating a wide range of functional groups directly, the synthesis eliminates multiple synthetic steps that would otherwise be required to mask reactive sites. Each skipped step represents a saving in labor, solvents, energy, and time, cumulatively leading to substantial cost savings per kilogram of the final API intermediate. Additionally, the catalyst loading is kept relatively low (10 mol%), and the use of palladium acetate, while a precious metal, is justified by the high turnover and yield, allowing for potential recovery and recycling of the metal from the waste stream. The simplified workup procedure, which involves filtration and standard chromatography rather than complex distillations or crystallizations from difficult solvent systems, further reduces the utility costs associated with solvent recovery and drying.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the reaction conditions and the availability of raw materials. The protocol operates at a moderate temperature of 100°C, which is easily achievable with standard steam heating systems found in most multipurpose chemical plants, removing the need for specialized high-temperature oil baths or cryogenic cooling. This thermal flexibility ensures that production can be maintained consistently across different manufacturing sites without requiring extensive re-engineering of equipment. Moreover, the broad substrate scope means that if a specific benzyl chloride derivative faces supply constraints, the platform technology is flexible enough to accommodate alternative substitution patterns or analogues without re-optimizing the entire process. This adaptability is crucial for maintaining continuity of supply for downstream drug products, especially in the face of global raw material shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with strong acid additions. The reaction profile is smooth, allowing for safe scale-up in batch reactors with standard agitation and temperature control. From an environmental standpoint, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of stoichiometric amounts of corrosive acids and the generation of fewer inorganic salts simplifies wastewater treatment protocols. The use of DME as a solvent, while requiring recovery, is manageable within standard solvent recycling loops. Overall, the process presents a lower environmental footprint compared to traditional methods, aiding companies in meeting increasingly stringent sustainability targets and regulatory standards for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific portfolio of compounds. The answers reflect the practical realities of running this chemistry in a production environment, focusing on yield consistency, purification needs, and raw material handling.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (C1) source, eliminating the need for separate carbon monoxide gas or toxic formylating agents, thereby simplifying the operational safety profile.
Q: What is the typical yield range for this palladium-catalyzed protocol?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 68% to 97% across various substrates, indicating robust performance even with electron-withdrawing or donating groups.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and avoids hazardous gaseous reagents, making it highly amenable to scale-up in standard stainless steel reactors with appropriate pressure ratings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN113045489B for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling palladium-catalyzed reactions under controlled atmospheres, and our rigorous QC labs enforce stringent purity specifications to meet the exacting demands of the global pharmaceutical market. We understand that consistency is key, and our process engineering team is dedicated to optimizing every parameter, from catalyst recovery to solvent recycling, to deliver a product that exceeds quality expectations while maintaining cost competitiveness.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to deep chemical expertise and a supply chain built on reliability and transparency. Please contact us today to request specific COA data for our quinolinone intermediates and to discuss route feasibility assessments for your proprietary molecules. Let us help you turn complex synthetic challenges into commercial successes.
