Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
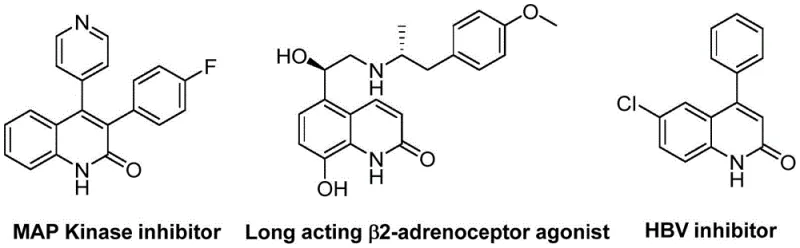
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing privileged heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in Chinese Patent CN113045489B, which discloses a highly efficient preparation method for 3-arylquinolin-2(1H)-one derivatives. This specific class of compounds is not merely an academic curiosity; it represents a critical structural motif found in a vast array of bioactive molecules, including potent antibiotics, antiplatelet agents, antitumor drugs, and endothelin receptor antagonists. The strategic value of this scaffold is underscored by its presence in complex drug candidates targeting critical pathways, as illustrated by the diverse biological applications ranging from MAP Kinase inhibition to HBV inhibition. By leveraging a novel palladium-catalyzed aminocarbonylation strategy, this technology offers a streamlined pathway to access these high-value intermediates, addressing the growing demand for reliable pharmaceutical intermediate suppliers who can deliver complex heterocycles with consistent quality and efficiency.
The significance of this patent extends beyond simple molecule construction; it fundamentally reimagines the atom economy and operational simplicity required for modern API intermediate manufacturing. Traditional approaches to synthesizing quinolin-2(1H)-one cores often rely on classical condensation reactions that may suffer from harsh conditions, limited substrate scope, or the generation of substantial chemical waste. In contrast, the methodology described in CN113045489B utilizes benzisoxazole as a unique dual-function reagent, acting simultaneously as the nitrogen source and the formyl source. This innovative approach eliminates the need for pre-functionalized formylating agents, thereby reducing the step count and minimizing the environmental footprint associated with the synthesis. For procurement managers and supply chain heads, this translates into a more resilient supply chain where the reliance on exotic or hazardous reagents is drastically reduced, ensuring greater continuity in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)-one ring system has been achieved through well-established protocols such as the Vilsmeier-Haack reaction, the Knorr synthesis, or the Friedlander condensation. While these methods have served the industry for decades, they are increasingly viewed through the lens of modern green chemistry and process efficiency as being suboptimal for large-scale commercialization. The Vilsmeier-Haack reaction, for instance, typically requires the use of phosphorus oxychloride (POCl3), a highly corrosive and moisture-sensitive reagent that poses significant safety hazards and waste disposal challenges in a manufacturing setting. Furthermore, classical methods often struggle with regioselectivity issues when dealing with substituted anilines or require multi-step sequences to install the necessary carbonyl functionality at the C2 position. These inefficiencies accumulate, leading to higher production costs, longer lead times, and a broader impurity profile that necessitates rigorous and costly purification steps to meet the stringent purity specifications demanded by regulatory bodies for active pharmaceutical ingredients.
The Novel Approach
The methodology presented in patent CN113045489B offers a paradigm shift by employing a transition metal-catalyzed carbonylation strategy that bypasses the limitations of classical condensation chemistry. This novel approach utilizes a palladium catalytic system in conjunction with molybdenum hexacarbonyl as a solid carbon monoxide surrogate, allowing the reaction to proceed under relatively mild thermal conditions without the need for high-pressure CO gas cylinders. The core innovation lies in the use of benzisoxazole derivatives, which undergo cleavage to provide both the nitrogen atom and the carbonyl carbon required for ring closure. This tandem process effectively merges the amination and carbonylation steps into a single operational unit, significantly simplifying the reaction workflow. By starting from readily available benzisoxazoles and benzyl chlorides, this route opens up a vast chemical space for diversification, enabling the rapid synthesis of libraries of 3-arylquinolin-2(1H)-one derivatives with varied electronic and steric properties, which is invaluable for medicinal chemistry optimization campaigns.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
At the heart of this transformative synthesis is a sophisticated palladium-catalyzed cycle that orchestrates the coupling of the benzisoxazole nitrogen source with the benzyl chloride electrophile and the carbon monoxide source. The reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step is critical as it activates the benzylic carbon for subsequent nucleophilic attack. Concurrently, the benzisoxazole ring undergoes activation, likely facilitated by the basic conditions provided by triethylamine and the presence of water, leading to the release of the nitrogen nucleophile. The insertion of carbon monoxide, supplied by the thermal decomposition of Mo(CO)6, into the palladium-carbon bond forms an acyl-palladium intermediate. This acyl species then undergoes nucleophilic attack by the nitrogen derived from the benzisoxazole, followed by reductive elimination to forge the new C-N bond and close the quinolinone ring, regenerating the Pd(0) catalyst to continue the cycle.
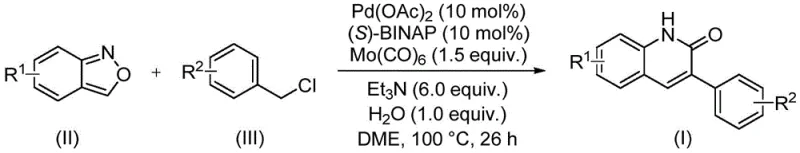
Beyond the primary catalytic cycle, the robustness of this method is attributed to its exceptional tolerance for diverse functional groups, a feature that is often a bottleneck in transition metal catalysis. The patent data explicitly demonstrates that substituents such as electron-withdrawing groups (e.g., cyano, trifluoromethyl, halogens) and electron-donating groups (e.g., methoxy, tert-butyl) on both the benzisoxazole and the benzyl chloride components are well-tolerated. This broad substrate scope suggests that the catalytic system is resistant to poisoning by common heteroatoms and is capable of navigating steric hindrance effectively. From an impurity control perspective, the use of a well-defined ligand system like (S)-BINAP helps to stabilize the palladium center, minimizing the formation of palladium black and other off-cycle species that could lead to metal contamination in the final product. This mechanistic elegance ensures that the resulting 3-arylquinolin-2(1H)-one derivatives are obtained with high purity, reducing the burden on downstream purification processes and aligning with the rigorous quality standards required for cost reduction in pharmaceutical intermediate manufacturing.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-scale production. The protocol involves charging a sealed reaction vessel with the requisite amounts of palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, the specific benzisoxazole derivative, and the benzyl chloride substrate in a solvent system such as dimethoxyethane (DME). The mixture is then heated to a temperature range of 90 to 110°C, with 100°C being the optimal setpoint identified in the examples, and maintained for approximately 26 hours to drive the reaction to completion. Following the reaction period, the workup procedure is remarkably straightforward, involving simple filtration to remove insoluble salts and catalyst residues, followed by silica gel treatment and standard column chromatography to isolate the pure product. For a detailed, step-by-step guide on executing this synthesis with precise stoichiometric ratios and safety precautions, please refer to the standardized protocol below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and supply chain logistics, the adoption of this patented methodology offers compelling strategic advantages that go beyond mere technical feasibility. The primary driver for adoption is the significant potential for cost reduction in pharmaceutical intermediate manufacturing, achieved through the utilization of commodity-grade starting materials. Benzisoxazoles and benzyl chlorides are widely available in the global chemical market, ensuring a stable supply base that is less susceptible to the volatility often seen with specialized reagents. Furthermore, the elimination of gaseous carbon monoxide cylinders in favor of solid molybdenum hexacarbonyl simplifies the infrastructure requirements for the reaction, removing the need for high-pressure gas handling systems and associated safety audits. This simplification directly translates to lower capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing facilities, allowing for more agile production scheduling and faster response times to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the high atom efficiency and the avoidance of expensive, hazardous reagents like phosphorus oxychloride or pre-formed acid chlorides. By using benzisoxazole as a dual nitrogen and formyl source, the synthesis effectively combines two synthetic transformations into one pot, reducing solvent consumption, energy usage, and labor hours associated with isolation and purification between steps. Additionally, the catalyst loading is optimized to be effective at relatively low concentrations, minimizing the cost contribution of the precious metal palladium. These factors cumulatively result in a substantially lower cost of goods sold (COGS) for the final quinolinone derivatives, providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. Since the key inputs for this reaction—benzisoxazoles and benzyl chlorides—are produced on a massive scale for various industrial applications, the risk of supply disruption is inherently lower compared to routes relying on niche custom synthons. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. This reliability ensures that production timelines can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and preventing costly delays in the drug development pipeline. The ability to source materials from multiple vendors adds an additional layer of security against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing practices, this method aligns well with environmental, social, and governance (ESG) goals. The use of a solid CO surrogate eliminates the safety risks associated with toxic CO gas, making the process safer for operators and easier to permit in regulated jurisdictions. The workup procedure, which relies on filtration and chromatography rather than complex aqueous extractions involving strong acids or bases, generates a simpler waste stream that is easier to treat and dispose of. This environmental compatibility facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase batch sizes from kilograms to tons without encountering the exponential increase in waste management costs that often plagues traditional synthetic routes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the technical disclosures within patent CN113045489B. These questions address common concerns regarding reaction scope, scalability, and product quality, providing a clear overview of what can be expected when implementing this palladium-catalyzed aminocarbonylation strategy. Understanding these nuances is essential for making informed decisions about process adoption and for anticipating any potential challenges during technology transfer. The answers provided are grounded in the experimental data and beneficial effects reported in the patent documentation.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate formylation reagents and simplifying the synthetic route significantly.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates wide functional group tolerance, successfully accommodating substituents such as halogens, methoxy groups, cyano groups, and trifluoromethyl groups without compromising yield.
Q: Is this process suitable for large-scale manufacturing?
A: The method utilizes commercially available and inexpensive starting materials with a straightforward workup procedure involving filtration and chromatography, making it highly amenable to scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
As the global demand for complex heterocyclic building blocks continues to rise, partnering with a contract development and manufacturing organization (CDMO) that possesses deep technical expertise in advanced catalytic methodologies is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging cutting-edge technologies like the Pd-catalyzed aminocarbonylation described in CN113045489B to deliver superior results for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from bench-scale discovery to full-scale commercial manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-arylquinolin-2(1H)-one derivatives meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, allowing you to quantify the economic benefits of switching to this more efficient manufacturing process. We encourage you to reach out today to discuss your specific requirements, obtain specific COA data for our catalog compounds, and receive comprehensive route feasibility assessments that will accelerate your path to market. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
