Advancing Pharmaceutical Intermediates: A Green Rhodium-Catalyzed Route to Isoindolinones
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly isoindolinones, which serve as critical cores in numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN115160211A, which discloses a novel green synthesis method for isoindolinone compounds. This technology leverages a rhodium-catalyzed cascade reaction that utilizes phenoxyacetonitrile as a stable one-carbon synthon. Unlike traditional approaches that often struggle with harsh conditions and unstable intermediates, this method achieves high atom economy and excellent functional group tolerance. By operating under mild air conditions without external oxidants, this process represents a paradigm shift towards sustainable manufacturing, offering a robust solution for producing high-purity pharmaceutical intermediates at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of isoindolinone skeletons has relied on multi-step synthetic routes that are fraught with inefficiencies and environmental concerns. Conventional methodologies frequently necessitate the use of highly reactive and unstable one-carbon synthons, such as diazo compounds, allenols, or ketene imines, which pose significant safety hazards and storage challenges. Furthermore, many of these traditional transformations require stoichiometric amounts of oxidants to drive the catalytic cycle, leading to increased waste generation and higher purification costs. The need for specialized ligands and Lewis acid additives further complicates the reaction setup, often resulting in lower overall yields and difficult downstream processing. These limitations create substantial bottlenecks for procurement teams aiming to secure reliable supplies of complex intermediates while maintaining cost-effectiveness.
The Novel Approach
In stark contrast, the method described in CN115160211A introduces a streamlined, one-step protocol that circumvents these historical hurdles. By employing phenoxyacetonitrile—a simple, stable, and commercially accessible nitrile—as the carbon source, the process eliminates the dependency on hazardous reagents. The reaction proceeds via a direct nucleophilic addition of an inert C-H bond to the cyano group, followed by a tandem cyclization, all facilitated by a rhodium catalyst. Crucially, this transformation occurs under air atmosphere without the need for additional oxidants, ligands, or Lewis acids. This simplification not only enhances operational safety but also drastically reduces the chemical load required for the reaction. As illustrated in the general reaction scheme below, the convergence of N-methoxybenzamide and phenoxyacetonitrile leads directly to the target isoindolinone structure with impressive efficiency.
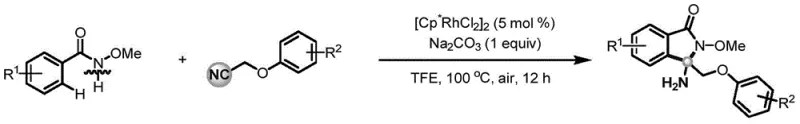
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated mechanism of transition metal-catalyzed C-H bond functionalization. The rhodium catalyst, specifically the dimer of pentamethylcyclopentadiene rhodium dichloride, activates the inert ortho-C-H bond of the N-methoxybenzamide directing group. This activation generates a reactive metallacycle intermediate that is poised for nucleophilic attack. Unlike oxidative coupling reactions that require external oxidants to regenerate the active catalyst species, this system utilizes the inherent reactivity of the nitrile group. The nucleophilic addition to the cyano moiety of the phenoxyacetonitrile creates a new carbon-carbon bond, setting the stage for the subsequent ring closure. This mechanistic pathway is highly selective, minimizing side reactions and ensuring that the majority of the starting material is converted into the desired heterocyclic product.
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on product quality. The absence of strong oxidants prevents the formation of over-oxidized byproducts that are common in traditional C-H activation protocols. Additionally, the mild reaction conditions (80-110°C) preserve sensitive functional groups that might otherwise degrade under harsher thermal or chemical stress. The high chemoselectivity observed across a wide range of substrates, including those with electron-withdrawing or electron-donating substituents, ensures a clean impurity profile. This purity is critical for pharmaceutical applications, where stringent regulatory standards demand minimal levels of unknown impurities. The ability to tolerate diverse substituents on both the benzamide and the phenoxyacetonitrile rings underscores the versatility of this catalytic system for generating diverse chemical libraries.
How to Synthesize Isoindolinone Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward, requiring standard equipment and readily available reagents. The process begins with the precise weighing of the N-methoxybenzamide derivative and the phenoxyacetonitrile substrate, typically in a molar ratio that favors the nitrile component to drive the equilibrium forward. These solids are combined with the rhodium catalyst and a base, such as sodium carbonate, in a reaction vessel containing trifluoroethanol as the solvent. The mixture is then heated to the specified temperature range under an air atmosphere, allowing the catalytic cycle to proceed over a defined period. Detailed standardized synthesis steps follow below to guide technical teams in replicating these results.
- Combine N-methoxybenzamide, phenoxyacetonitrile, rhodium catalyst, and sodium carbonate in trifluoroethanol solvent.
- Heat the mixture to 80-110°C under air atmosphere and react for 12 hours.
- Perform post-treatment including extraction and column chromatography to isolate the isoindolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant reduction of raw material complexity and cost. By replacing unstable, expensive, or hazardous coupling reagents with stable phenoxyacetonitrile, the supply chain becomes more resilient and less prone to disruptions caused by the scarcity of specialized chemicals. Furthermore, the elimination of oxidants and ligands reduces the total number of SKUs required for production, simplifying inventory management and lowering procurement overheads. The simplified workup procedure, involving basic extraction and chromatography, also reduces the consumption of solvents and silica gel, contributing to overall cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of costly additives and the improvement in atom economy. Traditional methods often suffer from low atom efficiency due to the loss of mass from oxidants and leaving groups; this new route incorporates the nitrile carbon directly into the final skeleton. Additionally, the use of air as the reaction atmosphere removes the need for expensive inert gas systems or rigorous degassing procedures, lowering utility costs. The high yields reported across various substrates mean that less starting material is wasted, directly improving the cost-per-kilogram of the final API intermediate. These factors combine to create a highly competitive cost structure for large-scale production.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any manufacturing operation, and this method excels by utilizing commodity chemicals. Phenoxyacetonitrile and substituted benzamides are widely available from multiple global suppliers, reducing the risk of single-source dependency. The stability of these starting materials allows for long-term storage without significant degradation, enabling companies to maintain strategic stockpiles without fear of spoilage. Moreover, the robustness of the reaction conditions means that production can be maintained even if minor fluctuations in environmental controls occur, ensuring consistent output and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for expansion. The reaction does not generate large volumes of hazardous waste associated with heavy metal oxidants or toxic byproducts, simplifying waste treatment and disposal compliance. The use of trifluoroethanol, while requiring proper handling, is manageable within standard solvent recovery systems. The absence of exothermic risks associated with unstable diazo compounds makes the scale-up from grams to tons safer and more predictable. This aligns with modern green chemistry principles, helping companies meet increasingly strict environmental regulations while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the proven capabilities of the method as demonstrated in the provided examples.
Q: What are the key advantages of using phenoxyacetonitrile in this synthesis?
A: Phenoxyacetonitrile serves as a stable and easily prepared one-carbon synthon, eliminating the need for unstable reagents like diazo compounds or allenols often required in conventional methods.
Q: Does this process require expensive oxidants or ligands?
A: No, the method operates efficiently under air conditions without the need for additional oxidants, ligands, or Lewis acid additives, significantly simplifying the reaction system.
Q: What is the substrate scope for this rhodium-catalyzed reaction?
A: The reaction demonstrates broad substrate compatibility, tolerating various functional groups such as halogens, alkyls, alkoxy, and nitro groups on both the benzamide and phenoxyacetonitrile components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology for the production of high-value isoindolinone derivatives. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of rhodium catalysts and the efficient recovery of solvents. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest international standards. Our commitment to quality assurance guarantees that the isoindolinone intermediates supplied to you are free from critical impurities and ready for subsequent synthetic steps.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. Please contact our technical procurement team to request specific COA data for relevant analogues and to discuss route feasibility assessments for your custom molecules. Together, we can optimize your supply chain and accelerate your drug development timelines with reliable, high-quality chemical solutions.
