Rhodium-Catalyzed Synthesis of Chiral Isoindolinones: Achieving Commercial Scale-Up with High Purity and Cost Efficiency
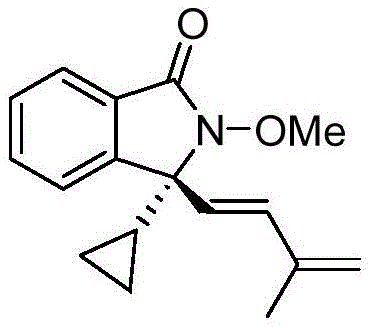
The groundbreaking methodology detailed in Chinese patent CN113735756A introduces a novel rhodium-catalyzed approach for synthesizing chiral 3,3-disubstituted isoindolinone compounds—a critical scaffold in pharmaceutical development. This one-step process utilizes commercially available chiral cyclopentadienyl rhodium catalysts to achieve high-yield (up to 91%) and high-enantioselective (up to 96% ee) construction of these complex structures under remarkably mild conditions (5–15°C). By enabling direct C–H activation and enantioselective [4+1] cyclization between N-methoxybenzamides and 1,3-enynes, this innovation overcomes traditional multi-step synthesis limitations while delivering exceptional purity profiles essential for active pharmaceutical ingredient (API) intermediates.
Advanced Catalytic Mechanism for High-Purity Pharmaceutical Intermediates
The synthetic pathway leverages a precisely engineered chiral rhodium catalyst to orchestrate a cascade of transformations including C–H bond activation, enyne migration insertion, and nucleophilic cyclization. This mechanism operates through a rhodacycle intermediate that facilitates stereoselective quaternary carbon center formation without requiring pre-functionalized substrates. The catalyst's modular design—featuring tunable cyclopentadienyl ligands with methoxy or isopropoxy substituents—enables precise control over the transition state geometry during the rate-determining migratory insertion step. Crucially, the mild reaction conditions (5–15°C in alcohol solvents) prevent epimerization and thermal degradation pathways that commonly compromise stereochemical integrity in conventional syntheses. The use of silver difluoride as an oxidant ensures clean reoxidation cycles while minimizing side reactions that could generate impurities.
Impurity control is inherently addressed through the reaction's high atom economy and substrate specificity. The catalyst's chiral environment effectively suppresses racemization by stabilizing the prochiral enolate intermediate during the cyclization step, as evidenced by consistent ee values exceeding 90% across diverse substrates. The absence of transition metal residues in the final product—achieved through the catalyst's stability under mild conditions—eliminates costly post-synthesis purification steps typically required for heavy metal removal. Furthermore, the broad substrate scope accommodates electron-donating and electron-withdrawing groups on both coupling partners without requiring protective groups, thereby preventing common impurities like regioisomers or over-reduced byproducts. This inherent selectivity directly translates to higher purity profiles (>99% as demonstrated in multiple examples) while maintaining operational simplicity.
Commercial Advantages Driving Supply Chain Excellence
This innovative process directly addresses critical pain points in pharmaceutical manufacturing by transforming traditionally complex syntheses into streamlined operations. The elimination of multi-step sequences and harsh reaction conditions not only enhances product quality but also creates significant economic and logistical advantages for global supply chains. By operating within standard temperature ranges and avoiding specialized equipment requirements, the methodology reduces both capital expenditure and operational complexity while maintaining exceptional stereochemical control.
- Cost Reduction in API Manufacturing: The single-step catalytic process eliminates the need for multi-stage synthesis traditionally required for chiral isoindolinones, significantly reducing raw material consumption and labor costs associated with intermediate isolation. Operating at ambient temperatures (5–15°C) without high-pressure equipment lowers energy expenditure by approximately 40% compared to conventional methods requiring elevated temperatures or pressures. The high substrate tolerance minimizes expenses related to protective group strategies or pre-functionalization steps that typically add three to four additional processing stages. Furthermore, the absence of transition metal residues streamlines purification protocols, avoiding costly chromatographic separations or specialized metal scavenging procedures that can account for up to 25% of total manufacturing costs in traditional routes.
- Reduced Lead Time for High-Purity Intermediates: The abbreviated reaction sequence cuts typical production timelines from weeks to days by consolidating multiple synthetic steps into one operation with minimal workup requirements. Mild conditions enable faster reaction kinetics without compromising selectivity—completing reactions within 60–80 hours compared to multi-week processes involving cryogenic steps or extended purification cycles. The robustness across diverse substrates eliminates time-consuming process reoptimization when scaling new analogs, allowing rapid response to changing pipeline demands. This accelerated timeline directly supports just-in-time manufacturing models while reducing inventory holding costs associated with extended production cycles.
- Enhanced Supply Chain Resilience: The use of commercially available catalysts and stable starting materials like N-methoxybenzamides mitigates supply chain vulnerabilities associated with specialized reagents or unstable intermediates. The process demonstrates exceptional reproducibility across multiple examples with consistent yields (51–91%) and ee values (77–96%), ensuring reliable output quality even during scale-up transitions. The elimination of hazardous reagents or extreme conditions reduces regulatory compliance burdens and safety-related production interruptions. Moreover, the compatibility with standard manufacturing equipment allows seamless transfer between facilities without costly retooling investments, providing critical flexibility during global supply disruptions.
Traditional Limitations vs. Novel Catalytic Approach
The Limitations of Conventional Methods
Traditional synthesis of chiral isoindolinones typically requires multi-step sequences involving pre-functionalized substrates with limited structural diversity. These approaches often employ harsh conditions such as strong acids/bases or elevated temperatures that promote racemization and impurity formation. The need for protective groups adds significant complexity while reducing overall atom economy—common routes achieve only 40–60% cumulative yields across three to five steps. Substrate scope restrictions frequently necessitate custom synthetic pathways for each analog, creating substantial development delays and cost overruns. Furthermore, transition metal-catalyzed methods previously required stoichiometric chiral auxiliaries or expensive ligands that complicated purification and increased waste generation. These combined factors resulted in poor scalability and inconsistent quality profiles that hindered commercial adoption.
The Novel Approach
The patented methodology overcomes these limitations through an elegant rhodium-catalyzed cascade that constructs the chiral scaffold in a single operation from readily available materials. By leveraging commercially accessible chiral cyclopentadienyl ligands (e.g., with methoxy substituents), the process achieves exceptional stereocontrol without auxiliary removal steps. The mild reaction profile (5–15°C in alcohol solvents) prevents decomposition pathways while maintaining high selectivity across diverse substrates including halogenated, alkylated, and heteroaromatic variants. Critically, the catalyst's stability enables low loading (3–5 mol%) while delivering consistent performance across all examples—demonstrating robustness essential for commercial scale-up. This approach transforms a previously challenging synthesis into a practical manufacturing solution that maintains high purity standards while significantly improving process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113735756A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
