Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical research, particularly for scaffolds exhibiting potent biological activity. The tetrahydro-beta-carbolinone skeleton is a privileged structure found in numerous natural products and drug candidates, such as the antiviral agent bauerine C and the anxiolytic candidate SL651498. However, traditional synthetic approaches often rely on expensive transition metals or harsh conditions that limit their industrial viability. A significant breakthrough in this domain is detailed in Chinese Patent CN115260188A, which discloses a novel preparation method utilizing a cobalt-catalyzed C-H activation carbonylation strategy. This technology represents a paradigm shift from precious metal catalysis to base metal systems, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By leveraging earth-abundant cobalt catalysts and solid carbon monoxide surrogates, this method addresses critical pain points in process chemistry, including cost, safety, and environmental impact.
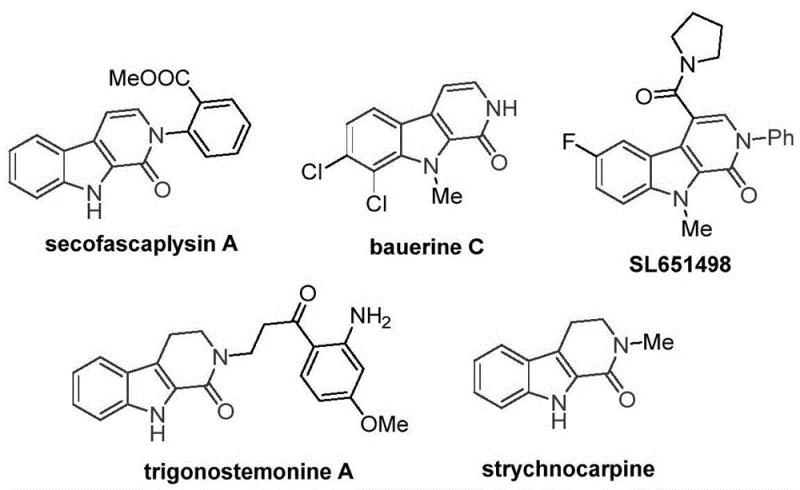
The strategic importance of this scaffold cannot be overstated, as it serves as a core motif in a wide array of bioactive molecules targeting the central nervous system and viral replication pathways. The ability to construct this ring system efficiently is therefore of paramount interest to R&D teams focused on pipeline acceleration. The patent highlights that while previous methods existed, they were often constrained by the necessity of palladium catalysis, which introduces significant cost burdens and regulatory hurdles regarding residual metal limits in final drug substances. The new methodology described in CN115260188A circumvents these issues by employing a cobalt system that operates under relatively mild thermal conditions (120-140°C) with high atom economy. For a reliable pharmaceutical intermediate supplier, adopting such a technology translates directly into enhanced supply chain resilience and the ability to offer high-purity tetrahydro-beta-carbolinones at a competitive price point.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydro-beta-carbolinone core via carbonylation reactions has been predominantly dependent on palladium catalysts. While palladium is renowned for its versatility in cross-coupling reactions, its application in large-scale manufacturing is fraught with challenges. The primary limitation is economic; palladium is a precious metal with volatile pricing, making the cost of goods sold (COGS) for processes relying on it unpredictable and often prohibitive for generic drug production. Furthermore, palladium-catalyzed reactions frequently require specialized ligands to achieve high turnover numbers, adding another layer of expense and complexity to the supply chain. From a regulatory perspective, the presence of palladium residues in Active Pharmaceutical Ingredients (APIs) is strictly controlled, necessitating additional purification steps such as scavenging or recrystallization, which inevitably reduce overall yield and increase waste generation. Additionally, many conventional carbonylation protocols utilize gaseous carbon monoxide, a highly toxic and flammable gas that requires specialized high-pressure equipment and rigorous safety protocols, thereby limiting the feasibility of these reactions in standard multipurpose chemical plants.
The Novel Approach
The methodology presented in patent CN115260188A offers a transformative solution by replacing the palladium system with a cobalt-based catalytic cycle. This approach utilizes cobalt acetate tetrahydrate, an inexpensive and readily available salt, as the precatalyst. Crucially, the reaction employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute. This innovation eliminates the safety hazards associated with handling CO gas, allowing the reaction to proceed in standard sealed vessels like Schlenk tubes or autoclaves without the need for high-pressure gas lines. The reaction conditions are optimized to operate in dioxane at temperatures between 120°C and 140°C for 16 to 24 hours, utilizing silver carbonate as an oxidant and triethylamine as a base. This combination of reagents facilitates a smooth C-H activation and subsequent cyclization to form the target lactam ring. The result is a process that is not only safer and cheaper but also demonstrates exceptional functional group tolerance, accommodating substrates with electron-donating and electron-withdrawing groups alike.

Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
Understanding the mechanistic underpinnings of this transformation is vital for process optimization and troubleshooting during scale-up. The proposed catalytic cycle begins with the oxidation of the cobalt(II) catalyst by silver carbonate to generate a reactive cobalt(III) species. This high-valent cobalt intermediate then coordinates with the tryptamine derivative substrate, specifically directing the activation of the C-H bond at the 2-position of the indole ring. This C-H activation step is the key selectivity-determining event, forming a stable cobalt(III) metallacycle. Following this activation, the carbon monoxide molecule, which is released in situ from the thermal decomposition of the 1,3,5-tricarboxylic acid phenol ester, inserts into the cobalt-carbon bond. This insertion generates an acyl-cobalt(III) intermediate, effectively building the carbonyl functionality required for the lactam ring. The cycle concludes with a reductive elimination step, which releases the cyclized tetrahydro-beta-carbolinone product and regenerates the lower-valent cobalt species to re-enter the catalytic cycle. Hydrolysis during the workup phase ensures the final isolation of the neutral ketone product. This mechanism highlights the elegance of using a solid CO source, as it provides a steady, controlled release of the gas directly within the reaction medium, maximizing utilization and minimizing waste.
From an impurity control perspective, this mechanism offers distinct advantages. The use of a specific directing group inherent in the tryptamine substrate ensures regioselective C-H activation, minimizing the formation of regioisomers that are common in non-directed functionalization strategies. Furthermore, the choice of silver carbonate as the terminal oxidant helps to drive the reaction to completion by re-oxidizing the cobalt center, preventing catalyst deactivation which often leads to incomplete conversion and difficult-to-remove starting materials. The compatibility of the system with various additives, such as pivalic acid, further stabilizes the catalytic species and enhances turnover frequency. For quality control teams, this means a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final high-purity pharmaceutical intermediates meet strict specifications for residual metals and organic impurities.
How to Synthesize Tetrahydro-beta-carbolinone Efficiently
The practical execution of this synthesis is designed to be straightforward, making it accessible for both laboratory discovery and pilot plant operations. The protocol involves a one-pot procedure where all reagents are combined in a single vessel, reducing unit operations and handling time. The reaction relies on the precise stoichiometric balance of the cobalt catalyst (30 mol%), oxidant (1.5 equiv), and CO source (3.0 equiv) to drive the equilibrium towards the product. The use of dioxane as the solvent is critical, as it provides the necessary solubility for both the organic substrates and the inorganic salts while withstanding the elevated reaction temperatures. Detailed standardized operating procedures for this synthesis, including specific mixing orders and quenching protocols, are essential for ensuring batch-to-batch consistency.
- Combine cobalt acetate tetrahydrate, silver carbonate, 1,3,5-tricarboxylic acid phenol ester, pivalic acid, triethylamine, and the tryptamine derivative substrate in dioxane solvent.
- Heat the reaction mixture to a temperature between 120°C and 140°C and maintain stirring for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the residue with silica gel, and purify the crude product via column chromatography to isolate the target tetrahydro-beta-carbolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology presents a compelling value proposition centered on cost stability and operational simplicity. The shift from precious metals to base metals fundamentally alters the cost structure of the synthesis, removing exposure to the volatile market prices of palladium and rhodium. Moreover, the use of a solid CO surrogate removes the logistical burden of sourcing and storing hazardous compressed gases, which often require specialized vendor contracts and safety audits. This streamlined supply chain allows for faster procurement cycles and reduced inventory holding costs for dangerous goods. The robustness of the reaction across a wide range of substrates means that a single manufacturing platform can be used to produce a library of analogues, maximizing asset utilization and reducing the need for campaign-specific equipment changes.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with cobalt acetate represents a drastic reduction in raw material costs, as cobalt is orders of magnitude cheaper than precious metals. Additionally, the elimination of expensive phosphine ligands, which are often air-sensitive and costly, further drives down the bill of materials. The simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of silica gel and solvents during purification, leading to substantial cost savings in waste disposal and material usage. These cumulative efficiencies translate directly into a lower cost of goods sold, enabling more competitive pricing for the final API or intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including cobalt acetate, silver carbonate, and triethylamine, are commodity chemicals available from multiple global suppliers. This multi-sourcing capability mitigates the risk of supply disruptions that can occur with specialized catalysts or ligands sourced from a single vendor. The stability of the solid CO source also ensures that the key carbonylating agent can be stored safely on-site for extended periods without degradation, providing a buffer against logistics delays. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, having been demonstrated to work effectively on gram scales with high conversion rates. The absence of high-pressure gas equipment lowers the barrier for scaling this reaction in existing multipurpose reactors, avoiding the need for significant capital expenditure on new infrastructure. From an environmental standpoint, the use of a base metal catalyst aligns with green chemistry principles by reducing the toxicity of the process. The simpler purification train results in less solvent waste and lower energy consumption, supporting corporate sustainability goals and ensuring compliance with increasingly stringent environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development teams evaluating this technology for adoption.
Q: Why is cobalt catalysis preferred over palladium for this synthesis?
A: Cobalt is an earth-abundant base metal that is significantly cheaper and less toxic than precious metals like palladium. This switch drastically reduces raw material costs and simplifies the removal of heavy metal residues, which is critical for meeting stringent pharmaceutical purity standards.
Q: What is the carbon monoxide source used in this reaction?
A: The process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute. This eliminates the need for handling hazardous high-pressure CO gas cylinders, greatly enhancing operational safety and ease of scale-up in standard laboratory or plant equipment.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the patented method demonstrates excellent substrate compatibility. It successfully accommodates various substituents including methyl, methoxy, halogens (Cl, Br), and different N-substituents (benzyl, allyl, phenyl), yielding products with high efficiency (up to 95% yield in model studies).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing manufacturing costs. Our technical team has thoroughly analyzed the potential of the cobalt-catalyzed route described in CN115260188A and is prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetrahydro-beta-carbolinone intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to evaluate the feasibility of this cost-effective route for your specific pipeline candidates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that compares this cobalt-mediated process against your current synthetic strategy. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our reference standards, and receive comprehensive route feasibility assessments tailored to your production volume needs. Let us help you secure a sustainable and economical supply of these vital pharmaceutical building blocks.
